Wei Gao#, Hao Chen#, Haiqing Jiang, Qiulin Wang, Meiyu Tang and Hong-Sheng Wang*
Institute of Dermatology, Chinese Academy of Medical Sciences. Jiangsu Key Laboratory of Molecular Biology for Skin Diseases and STIs, 12, Jiangwangmiao Road, Nanjing 210042, China. *E-mail: whs33@vip.sina.com
#These authors contributed equally to this work and should be considered as first authors.
Accepted Feb 20, 2014: Epub ahead of print Feb 27, 2014
Mycobacterium colombiense belongs to the M. avium complex (MAC), a type of slow growing mycobacterium (1) which infects humans, both immunocompetent and incompetent patients.
CASE REPORT
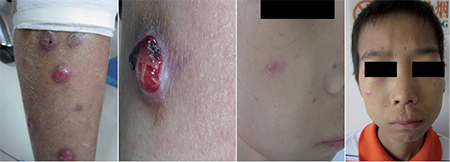
We wish to report a case of an apparently immunocompetent 17-year-old boy with disseminated cutaneous infection caused by M. colombiense. The patient presented with deep asymptomatic erythematous nodules on his face and extremities for 4 months without any preceding history of trauma or surgery. Some of the nodules on his extremities developed into discharging ulcers, others featured slightly scaly erythematous infiltrated plaques (Fig. 1). Two months earlier, he was admitted to a local hospital on account of irregular bowel movements and an occasional slight pain in the right lower quadrant. He was diagnosed with intestinal tuberculosis according to clinical features and biopsy of the intestinal mucosa, which indicated infectious granuloma. The patient received anti-tuberculosis chemotherapy for 2 weeks but ceased taking the drugs because of severe drug-induced liver hepatitis. The past medical history of the patient only revealed chronic hepatitis B with normal liver function. Routine blood test showed mild anaemia with haemoglobin of 101 g/l. Blood culture, purified protein derivative and human immunodeficiency virus antibody tests were negative. Systemic examinations of chest X-ray, type-B ultrasonography and CT scan indicated no abnormal lesions.
A skin biopsy of a nodule on the patient’s left leg displayed deep dermis showing nodular lesions with clear boundaries, collagen oedema, mucinous degeneration, blood vessel hyperplasia, infiltrated histiocytes and the absence of caseation necrosis, without special manifestation. Moderate growth of smooth, creamy, yolk yellow colonies was observed after 3 weeks of incubation only at 32 and 37°C on Löwenstein–Jensen (L–J) medium (Fig. 2A). Ziehl–Neelsen staining confirmed that the cultured organisms were acid-fast bacilli (AFB) (Fig. 2B). Pigment production testing showed that the isolated AFB was non-chromogenic. Fungal and other standard bacterial cultures were negative. Sequence analysis of 16S rDNA genes showed 99% similarity with M. colombiense strain CIP108962, and hsp65 genes indicated 99% homology with M. colombiense strain InDRE 9m. Gene sequences were analysed using the BLAST V2.0 software available at http://www.ncbi.nlm.nih.gov/BLAST/. Gene sequencing results indicated that the strain was most similar to M. colombiense.
As indicated by drug susceptibility analysis, we administered an oral regimen of clarithromycin (500 mg/day), rifabutin (750 mg/day) and moxifloxacin (300 mg/day). After 3 months of therapy, the patient’s skin lesions greatly improved. However, his bowel symptoms did not completely improve and he continues to receive antibiotics.
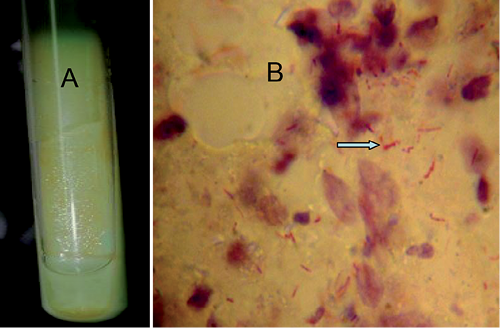
Fig. 2. Smooth, creamy, yolk yellow colonies grew after 3 weeks of incubation (37 °C) on Löwenstein–Jensen medium (A). Acid-fast bacilli were positive (arrow) in the tissue fluid of skin lesion by Ziehl-Neelsen staining (B).
DISCUSSION
M. colombiense, a Runyon group III, non-chromogenic, urease-positive, non-tuberculous mycobacterium (NTM), was initially isolated from blood specimens of 4 HIV-positive patients in Colombia. These isolates form a special species because of sequence comparisons with closely related Mycobacterium species (1). Following the initial series, M. colombiense has been isolated from other clinical scenarios (2–4). Diagnosing NTM infections requires a series of laboratory tests to identify and exclude suspicious diagnoses. Tissue cultures are important for the diagnosis of NTM infections. Approximately 90% of all known cases show positive culture results (5). However, diagnosis is always frequently delayed for weeks or even longer.
The development of molecular tools can be used to describe emerging MAC species previously diagnosed as M. avium subsp. avium (6). M. colombiense is most closely related to M. avium based on 16S rRNA sequence analysis and DNA–DNA hybridisation experiments (1). RpoB sequencing has also proven useful in identifying Mycobacterium isolates, particularly for assemblages (such as MAC) (7).
The most recommended treatment for MAC includes a combination of ethambutol, clarithromycin/azithromycin and rifampicin/rifabutin for 3–6 months or longer (8). Furthermore, in vitro drug susceptibility testing can help doctors choose drugs with the proper sensitivity. Diagnosis of this rare cutaneous infection should be considered both in immunocompromised and immunocompetent patients. Accurate and rapid identification of the causative Mycobacterium species is necessary since different infections have varying indications, drug selection, duration and follow-up protocols.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (No. 81371751), and the fund for Key Clinical Program of the Ministry of Health of China (No. 2010-2012-125).
The authors declare no conflict of interest.
References
