Song Youn Park1, Sohee Oh2, Eun Jee Kim1, So Young Yoon1, Hyun Sun Park1, Hyun-Sun Yoon1 and Soyun Cho1*
Departments of 1Dermatology and 2Biostatistics, SMG-SNU Boramae Medical Center, Boramae-Ro 5-Gil, Dongjak-Gu, Seoul, 156-707, Korea. *E-mail: sycho@snu.ac.kr
Accepted Aug 6, 2013; Epub ahead of print Oct 15, 2013
Atopic dermatitis (AD) is a chronic-intermittent eczematous dermatitis that starts in early childhood (1). AD can be subdivided into extrinsic (ADe) and intrinsic (ADi) subtypes (2). Although 10–45% of patients with AD have been reported to have ADi, defined as AD with low serum immunoglobulin E (IgE) and no allergen-specific IgE, there are few laboratory markers to diagnose or evaluate the severity of ADi. Moreover, because AD is diagnosed based on its characteristic clinical features, it is difficult to diagnose when a patient is too young or in the early stage of the disease with subtle clinical features of AD. It is now widely accepted that AD is associated with elevated levels of eosinophil cationic protein (ECP) (3–5). ECP is a ribonuclease produced by eosinophils, which is elevated in many allergic diseases (3). However, reports are scarce and controversial regarding the age-related changes and diagnostic utility of ECP in AD. Moreover, with regards to evaluating ADi, only a few studies have been published (6–10).
The aim of the present study was to compare the clinical and laboratory parameters between ADi and ADe and to assess the usefulness of ECP in diagnosing AD, including ADi.
MATERIALS AND METHODS
Retrospective chart review was carried out on subjects with AD whose total IgE, ECP and allergen-specific IgE were checked from January 2005 to August 2012 in our hospital. Disease severity was classified into 3 categories (mild, moderate and severe), according to the prescription at the patient’s visit. The subjects were classified into the ADi group if their IgE levels were ≤ 183 U/ml and allergen-specific IgE was absent. Subgroup analysis was carried out for very young patients under the age of 2 years, the age range at which AD most commonly presents (11).
Statistical analysis was performed with the SPSS 18.0 program. Fisher’s exact test and Student’s t-test were used to compare the mean values of the 2 groups. To analyse the correlation of categorical data, multivariate regression analysis was performed.
RESULTS
A total of 178 patients were enrolled in the study. The sex ratio was 1.5:1 (108 males and 70 females). Mean ± standard deviation (SD) age was 9.5 ± 10.5 years (males 8.3 ± 0.9, females 11.3 ± 1.4 years, p = 0.06), age range 0.2–41.9 years.
Among the subjects, 30 (16.9%) had ADi and 148 (83.1%) had ADe (Table I). Total IgE level was lower in the ADi group than in the ADe group (p = 0.004). The ECP level was also lower in the ADi group, albeit non-significant (p = 0.10).
Table I. Some characteristics of the studied paediatric patients with intrinsic (ADi) and extrinsic (ADe) atopic dermatitis
|
ADi |
ADe |
Significance level |
|
|
Patients, n |
30 |
148 |
|
|
Age, years, mean ± SD |
5.2 ± 7.1 |
10.3 ± 10.9 |
p = 0.014 |
|
IgE, IU/ml, mean ± SD |
49.4 ± 49.6 |
793 ± 1,397 |
p = 0.004 |
|
ECP, µg/ml, mean ± SD |
23.9 ± 17.0 |
56.2 ± 108 |
Not significant |
ECP: eosinophil cationic protein.
A significant correlation between age and total IgE was noted for both the ADe (r = 0.489, p < 0.0001) and the ADi group (r = 0.541, p = 0.002). In a subgroup analysis of ADe subjects ≤ 2 years, the positive correlation of age with total IgE still held (r = 0.271, p = 0.050), whereas, the ECP level demonstrated a negative correlation with age (r = –0.298, p = 0.030). In subjects with ADi ≤ 2 years, the total IgE level did not show any correlation with age (r = 0.28, p = 0.354), but ECP showed a negative correlation (r = –0.785, p = 0.001) (Fig. 1).
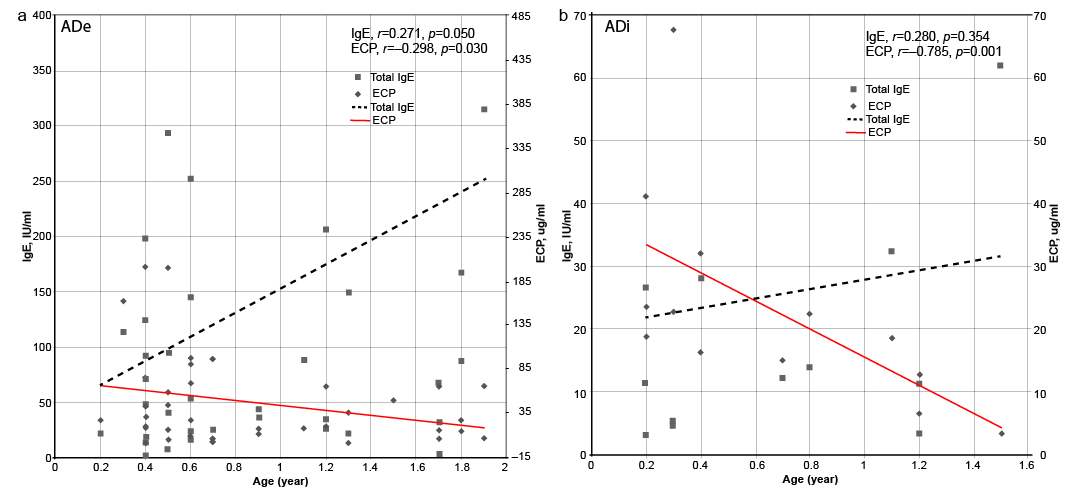
Fig. 1. Trends in total immunoglobulin E (IgE) and eosinophil cationic protein (ECP) in relation to age in subjects under 2 years. (a) Analysis of the extrinsic atopic dermatitis (ADe) group. (b) Analysis of the intrinsic atopic dermatitis (ADi) group. Only the correlation between ECP and age is statistically significant. Note the difference in absolute values of IgE and ECP in ADe and ADi.
DISCUSSION
Although the pathomechanisms of AD are not fully elucidated, the clinical features of ADi include a relatively milder severity and more intact skin barrier function than for ADe (1). In this study, the severity was also milder in the ADi group, which supports our current knowledge. Also not shown is that we did not find any correlation of clinical severity to ECP level in either groups (although we did not include the effect of therapy in our study).
It is known that AD is generally more common in females (1). However, a Korean national survey (11) and the Jeju Island study (12) revealed a male predominance at younger ages. In the present study, the sex ratio was 1.5:1, with a male predominance. This can probably be attributed to the high proportion of younger children.
In agreement with previous reports, subjects in the present study had significantly higher IgE and ECP values compared with normal reference values (3). The mean ECP value was twice as high in ADe as in ADi; however, the difference was not significant. Very few studies of laboratory values in relation to AD subtypes and age have been published, and they usually do not categorize the subjects into ADi and ADe (6, 7). The present study reveals that the total IgE level increases significantly with age. Although ECP did not show a correlation in the whole age range, a tendency to negative correlation was revealed in subjects under the age of 2 years, and this correlation was significant especially in the ADi group.
Since ECP and IgE levels demonstrated opposite trends across the 0–2 year age range, we speculate that ECP might reflect a mostly constitutive baseline level in genetically predisposed individuals, whereas IgE changes occur later in life in response to various environmental factors. As there are few laboratory parameters to diagnose ADi, ECP can be a useful diagnostic tool, especially in infants < 2 years whose skin lesions are too subtle to be definitely diagnosed as AD. But it cannot per se distinguish ADi from ADe.
The authors declare no conflicts of interest.
REFERENCES
