Takuya Takeichi1, Kazumitsu Sugiura1, Yuki Nakamura2, Yumi Fujio2, Izumi Konohana2 and Masashi Akiyama1*
1Department of Dermatology, Nagoya University Graduate School of Medicine, 65 Tsurumai-cho, Showa-ku, Nagoya 466-8550, and 2Department of Dermatology, Hiratsuka Municipal Hospital, Hiratsuka, Japan. *E-mail: makiyama@med.nagoya-u.ac.jp
Accepted Mar 24, 2016; Epub ahead of print Mar 29, 2016
Darier’s disease (DD, OMIM#124200) is an autosomal dominant skin disorder characterized by multiple dark-brown keratotic plaques and warty papules covered by thick crusts. Most cases of DD are caused by mutations in ATP2A2, which is expressed both in the skin and the brain (1). ATP2A2 encodes the cardiac muscle SERCA2a protein (997 amino acids) and the ubiquitously expressed SERCA2b (1042 amino acids), but no cardiac manifestations have been observed in patients with DD (2). DD sometimes shows neuropsychiatric manifestations, including schizophrenia (3). In a recent Swedish cohort study, 6 of 770 patients with DD (0.8%) were found to have schizophrenia (4). It was determined that individuals with DD had a 2.3 times higher risk of being diagnosed with schizophrenia than matched individuals from the general population, although the genotypes of ATP2A2 in the patients were not mentioned (4). Thus far, no genotype/phenotype correlations between ATP2A2 mutations and schizophrenia have been demonstrated. In this report, we describe a novel mutation in ATP2A2, p.Cys318Tyr, in a Japanese male with DD complicated by schizophrenia.
CASE REPORT
The proband is a 66-year-old Japanese man, with no apparent family history, who presented with multiple brown papules with itching on his entire body surface from one year earlier. Examination revealed multiple brown papules with crusts on the seborrhoeic areas of the head, face, neck, abdomen, back and legs (Fig. 1A and B). Partially exudative, hyperkeratotic lesions with odour were seen on the forehead, the nasal wings (Fig. 1A) and the intertriginous areas. His hair and nails were normal. He had been diagnosed with schizophrenia and intellectual disability, and had been treated with oral olanzapine since the age of 63 years. Light microscopy of a lesional skin biopsy from his neck revealed hyperkeratosis, acantholysis, cleft, grains and corps ronds in the epidermis (Fig. 1C). Treatment with etretinate (0.2 mg/kg/day) effectively resolved the eruptions.
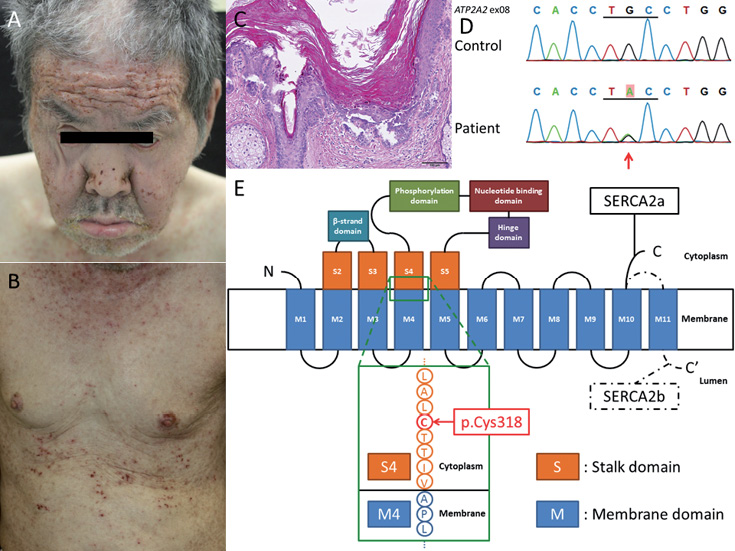
Fig. 1. Clinical and molecular features of the proband with Darier’s disease. Multiple brown papules with crusts are present on the (A) face, and (B) abdomen.(C) Haematoxylin-eosin staining shows marked hyperkeratosis, acantholysis, and clefts and dyskeratosis in the suprabasal layers. Scale bar: 100 μm. (D) Sanger sequencing reveals a heterozygous mutation within ATP2A2, c.953G>A (p.Cys318Tyr), in the proband. (E) A simple schematic of the SERCA2 domain structure. Blue areas: transmembrane domains. Orange areas: stalk domains. SERCA2a and SERCA2b isoforms are identical in the first 993 amino acid residues, but differ in the C terminus. Red arrow: mutation in the present patient with DD. Written informed consent was provided by the patient’s legal guardian.
Ethical approval was obtained and all research was performed in accordance with the principles of the Declaration of Helsinki. Genomic DNA from the patient’s peripheral blood leukocytes was used for Sanger sequencing analysis, as described previously (5). Analysis of the data revealed a novel heterozygous missense mutation in ATP2A2: c.953G>A (p.Cys318Tyr) (Fig. 1D). The mutation has not been described in the Human Genetic Variation Database, which includes 1,208 exome-sequencing data-sets of healthy Japanese controls (http://www.genome.med.kyoto-u.ac.jp/SnpDB/index.html). In silico analysis with PolyPhen-2 predicts the mutation to be “probably damaging” (6), and Sorting Intolerant From Tolerant (SIFT) predicts it to be “damaging” (7); thus, the mutation is thought to be functionally relevant.
DISCUSSION
In DD, 2 mutations at the corresponding cysteine residue, Cys318, within the ATP2A2, p.Cys318Arg (8) and p.Cys318Phe (9), have been reported previously, and we detected a novel mutation, p.Cys318Tyr, the third mutation in the residue Cys318 (Fig. 1E). Members of the DD pedigree associated with p.Cys318Arg substitution show severe phenotypes, with early onset, oral lesions and erosive eruptions. However, they have no neuropsychiatric manifestations (8). By contrast, members of the reported family with p.Cys318Phe alteration show typical phenotypes of DD. It is of note that the elder brother of this patient had mild mental retardation and psychiatric abnormality (9). Table I summarizes reported patients with missense mutations affecting the cysteine residue Cys318 and their clinical features. The mutation p.Cys318Arg changes a hydrophobic amino acid into a basic one in the stalk 4 domain of the molecule (8). As for cysteine, both phenylalanine and tyrosine are hydrophobic amino acids, but phenylalanine and tyrosine are aromatic acids, differing from cysteine. In addition, mutation of the same residue to an alanine was shown to have no effect on Ca2+ transport in vitro (10). Thus, the change to aromatic acids from the corresponding cysteine might determine the occurrence of neuropsychiatric disorders.
Table I. Reported missense mutations of the first cysteine in the stalk 4 domain of SERCA2
|
Pedigree |
Family 1 (ref. 8) |
Family 2 (ref. 9) |
|
Present case |
|
Substitution |
p.Cys318Arg |
p.Cys318Phe |
|
p.Cys318Tyr |
|
Family member |
2 patients |
Patient 1 |
Patient 2 |
No family history |
|
Age of onset |
< 10 years |
10 years |
Not stated |
65 years |
|
Eruptions |
Oral and flexural lesions |
Typical |
Typical |
Typical |
|
CNS manifestations |
Absent |
Mild psychiatric abnormality |
Absent |
Schizophrenia and intellectual disability |
Jacobsen et al. (11) reported that some missense mutations in ATP2A2 correlate with the presence of neuropsychiatric phenotypes and, more specifically, that missense mutations in the ATP-binding domain may have relevance in mood disorders. In contrast, Ruiz-Perez et al. (3) noted that neuropsychiatric features are not associated with a specific type of mutation in ATP2A2, but rather depend on concomitant genetic and environmental factors (3). In fact, there are several reports about neuropsychiatric complications in patients with DD having nonsense and frameshift mutations (8). With respect to schizophrenia, there have been few reports related to ATP2A2 genotypes. So far, there are no reported schizophrenia-associated mutation sites or domains within ATP2A2. A significant association between ATP2A2 and schizophrenia has been reported recently in a genome-wide association study (4). Further accumulation of schizophrenia cases with ATP2A2 mutations is needed to confirm the molecular pathogenesis of schizophrenia by ATP2A2 mutations.
ACKNOWLEDGEMENTS
The authors would like to thank Ms Haruka Ozeki and Ms Yuka Terashita for their technical help in analysing mutations of ATP2A2. This study was supported by JSPS KAKENHI Grant Numbers 15H04887, 15K15415, 15H04886, 15K15414, 15H06280. The work was also supported by the Japan Intractable Diseases Research Foundation.
REFERENCES
