OBJECTIVE: The immobility common to patients after acute stroke has the potential to increase negative mood symptoms. We evaluated the effect of very early mobilization after stroke on levels of depression, anxiety and irritability.
DESIGN: AVERT is a randomized controlled trial; patients in the very early mobilization group receive mobilization earlier (within 24 h of stroke) and more frequently than patients in the standard care group.
Patients: Seventy-one patients with confirmed stroke were included.
METHODS: Patients were assessed on the Irritability, Depression and Anxiety (IDA) scale at multiple time-points.
RESULTS: At 7 days, very early mobilization patients were less depressed (z = 2.51, p = 0.012) and marginally less anxious (z = 1.79, p = 0.073) than standard care patients (Mann- Whitney test). Classifying IDA scores as normal or depressed, and using backward stepwise multivariable logistic regression, very early mobilization was associated with a reduced likelihood of depression at 7 days (odds ratio 0.14, 95% confidence interval 0.03–0.61; p = 0.009).
CONCLUSION: Very early mobilization may reduce depressive symptoms in stroke patients at 7 days post-stroke.
Key words: stroke, rehabilitation, depression, anxiety.
J Rehabil Med 2008; 40: 609–614
Correspondence address: Toby Cumming, Level 2, Neurosciences Building, Heidelberg Repatriation Hospital, 300 Waterdale Rd, Heidelberg Heights, Victoria, 3081 Australia. E-mail: tcumming@nsri.org.au
Submitted November 22, 2007; accepted March 26, 2008
Introduction
In the early period following stroke, bed rest is an accepted form of management (1). Observations of patients after acute stroke indicate that they are relatively immobile, spending more than half the day resting in bed (2). Yet published trials across a wide range of conditions provide little evidence for bed rest as an effective treatment (3). Mobilization within 24 h of stroke is associated with prompt discharge home and contributes to improved outcomes in patients managed in a stroke unit (4, 5). Furthermore, Indredavik et al. (5) suggest that very early mobilization (VEM) may have important psychological effects.
After stroke, patients are particularly vulnerable to mood disorders. Whilst 11% of older people are depressed (6), 33% of all patients who suffer stroke experience depressive symptoms (7). Even after 5 years, symptoms of depression are present in approximately 17% of stroke survivors (8). Anxiety after stroke is also common. Fure et al. (9) found that more patients after stroke had anxiety (26%) than depressive (14%) symptoms in the acute phase (3–7 days post-stroke).
Several plausible mechanisms exist by which VEM might improve the psychological state of stroke survivors. The first is indirect: exposure to early mobilization may reduce physical disability, which consequently leads to a reduction in depression and anxiety. Secondly, earlier and more physical activity may have physiological effects on mood, such as changes in endorphin and monoamine concentrations (10). Thirdly, starting mobilization interventions early may improve self-efficacy (judgements about one’s own capabilities in a specific situation; 11), particularly if gains are perceived by the patient. Self- efficacy is strongly related to depression (12).
The aim of the current study was to determine whether VEM (commenced within 24 h of stroke onset and continued for the first 14 days) had a positive impact on psychological well-being in the first 12 months after stroke compared with standard care (SC). We hypothesized that patients exposed to VEM would be less depressed and less anxious than SC controls, particularly at the 7-day time-point.
Methods
The data for this study were acquired as part of phase II of A Very Early Rehabilitation Trial (AVERT). This was a prospective randomized controlled trial using blinded outcome assessment and intention-to-treat analysis. The primary focus of the phase II study was to test trial safety and feasibility. Methods are reported in detail elsewhere (13) and in brief below.
Patient sample
Patients were included if they were older than 18 years, had first or recurrent stroke, and were admitted within 24 h of symptom onset. Patients were excluded if they had a pre-morbid modified Rankin Scale (mRS) score > 3 (disability), deterioration within the first hour of admission to the stroke unit or direct admission to intensive care, a concurrent progressive neurological disorder, an acute coronary syndrome, severe heart failure, a lower limb fracture that prevented mobilization, or a need for palliative care. Of all stroke patients screened, 18% were recruited, 57% failed to meet the inclusion criteria (of these patients, 55% reached hospital more than 24 h after stroke symptom onset, 34% presented after working hours or at the weekend, 11% were recruited to other trials) and the remaining 25% were ineligible based on the exclusion criteria.
Procedure
The study was conducted in acute stroke units at 2 large hospitals in Melbourne, Australia. Ethics approval was granted by both hospitals, and informed consent was obtained from all patients or their representative. Computer-generated blocked randomization procedures (block size 6) using opaque envelopes, with stratification by stroke severity and clinical site, were used to allocate patients to VEM or SC. We aimed to recruit equal numbers of patients across the stroke severity spectrum using the National Institute of Health Stroke Scale (NIHSS; 14): mild (0–7), moderate (8–16) and severe (> 16).
Intervention
Both groups received SC from ward and therapy staff. Patients randomized to the VEM group began mobilization as soon as practical after recruitment, with the aim of first mobilization within 24 h of stroke symptom onset. VEM was delivered by a nurse and physiotherapist team for the first 14 days post-stroke or until discharge (whichever was sooner). The emphasis of VEM was to assist the patient to be upright and out of bed at least twice per day, thereby doubling the standard dose previously identified (2, 15). This level of mobilization was comparable to a stroke unit with well-established VEM protocols (16). Usual hospital occupational health and safety procedures for manual handling of patients were maintained. The VEM protocol included physiological monitoring of blood pressure, heart rate, oxygen saturation and temperature prior to each mobilization in the first 3 days post-stroke (13).
Outcome measures
Patient characteristics collected at baseline included gender, age, stroke severity (NIHSS) and previous stroke history. Pre-stroke history of depression or anxiety was established from medical records. Retrospective pre-morbid disability was scored using the mRS (17), with lower scores (0–2) representing less disability and a higher score (3) representing moderate disability. For the current study, the primary outcome of interest was psychological well-being, measured using the Irritability, Depression and Anxiety (IDA) scale (18) at 7 and 14 days, and 3, 6 and 12 months post-stroke. Our focus was on depression and anxiety in the acute phase (7 days) and in the longer term (12 months). The validity of the IDA has been established (19), and it has been used in stroke populations because it avoids somatic symptoms that may be present in stroke survivors irrespective of mood disorder (8). The scale includes 4 components: depression, anxiety, inward irritability and outward irritability. Scores on each component indicate 1 of 3 clinical classifications (19). The scores for depression are normal (0–3), borderline (4–6) and morbid (7–15), while for anxiety they are normal (0–5), borderline (6–8) and morbid (9–15). For analyses requiring a binary outcome measure, scores were reclassified as normal (not depressed, 0–3) or depressed (4–15) and normal (not anxious, 0–5) or anxious (6–15). All patients in the borderline and morbid categories, therefore, were classified together as symptomatic. Other outcome measures included whether the patient could walk 50 m unassisted at 7 days and 12 months post-stroke, and disability on the mRS at 12 months.
Blinding
Patients were blind to group allocation. They consented to be randomized to one of 2 rehabilitation styles: A or B. Although trial therapists and nursing staff could not be blinded to intervention group, the importance of maintaining blinding was emphasized to them. Mobilization sessions were conducted behind closed curtains wherever possible and were not documented in the medical record. There was no evidence of contamination (i.e. increased physical activity or decreased bed rest) among other stroke patients not involved in the trial (13). All outcome measures were recorded by a blinded assessor located off-site. When the patient was still in hospital at the 7- and 14-day assessments, trial staff were informed of an impending blinded assessor visit so that the assessor would not inadvertently view a mobilization session.
Statistical analyses
Between-group differences were computed on an intention-to-treat basis. Group differences on total IDA score and scores on the 4 IDA subscales were assessed using a 2-sample Wilcoxon rank-sum (Mann-Whitney U) test. To assess differences between groups, categorical variables were analysed using a χ2 test. Univariable logistic regression analyses were performed to determine differences between groups for these same factors. Statistical significance was set at p < 0.05. A multivariable logistic regression model was then constructed that included depression or anxiety, and other potentially contributing factors (age, gender, stroke severity (NIHSS > 7), pre-morbid mRS denoted as good (0–2) or poor (3), and ability to walk unassisted (yes/no) at 7 days). Least significant variables were serially removed until only variables with p < 0.10 remained. Finally, each omitted variable was then added into the final model 1 at a time to determine its overall contribution to the model.
Factors associated with depression or anxiety at both 7 days and 12 months post-stroke were assessed for all patients (VEM and SC grouped together). Univariable and multivariable logistic regression was conducted, with depression and anxiety as the outcome measures, using the same procedure as the between-group analysis outlined above. Additional variables inserted into these regressions were intervention group (VEM or SC) and previous stroke (yes/no). For the 7-day analysis, ability to walk unassisted at 7 days (yes/no) and pre-morbid mRS (good (0–2) vs poor (3)) were entered. For the 12-month analysis, ability to walk unassisted at 12 months (yes/no) and 12-month mRS (good (0–2) vs poor (3–5)) were entered. All analyses were performed using SPSS version 14.0.
Results
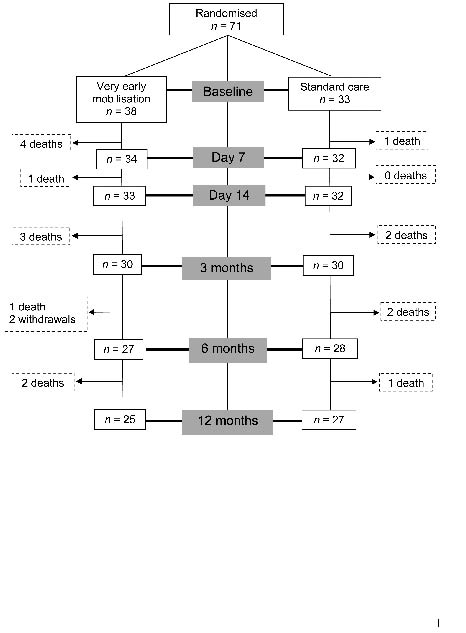
A total of 71 patients was recruited: 60 from the Austin Hospital between March 2004 and February 2006 and 11 from St Vincent’s Hospital between May 2005 and February 2006. Enrolment and retention of patients throughout the trial is outlined in Fig. 1. Patient demographics, including baseline measures of impairment and disability, are presented in Table I. The VEM and SC groups were comparable on all baseline measures.
Fig. 1. Patient enrolment and retention flow chart.
| Table I. Patient demographics |
| | VEM n = 38 n (%) | SC n = 33 n (%) |
| Age, years, mean (SD) | 74.6 (14.6) | 74.9 (9.8) |
| Gender, male | 22 (58) | 16 (48) |
| Previous stroke | 11 (29) | 7 (21) |
| Pre-morbid mRS, median (IQR) | 1 (0, 2) | 0 (0, 1) |
| NIHSS, median (IQR) Mild, 0–7 Moderate, 8–16 Severe, > 16 | 9.5 (5, 18) 15 (39) 13 (34) 10 (26) | 8 (4, 12) 15 (45) 11 (33) 7 (21) |
| Stroke type (Oxfordshire)† TACI PACI POCI LACI ICH | 10 (26) 13 (34) 7 (18) 5 (13) 3 (8) | 6 (18) 10 (30) 5 (15) 6 (18) 6 (18) |
| Lesion side Left Right | 22 (58) 16 (42) | 14 (42) 19 (58) |
| †Oxfordshire classifications (27): Total Anterior Circulation Infarct (TACI), Partial Anterior Circulation Infarct (PACI), Posterior Circulation Infarct (POCI), Lacunar Infarct (LACI), Intracerebral Haemorrhage (ICH). VEM: very early mobilization; SC: standard care; SD: standard deviation; IQR: interquartile range (denoted by 25th and 75th percentile); NIHSS: National Institute of Health Stroke Scale. |
The intended intervention was delivered (13): median time to first mobilization was significantly shorter in the VEM group (18.1 vs 30.8 h in SC) and median total dose of mobilization was significantly greater in the VEM group (167 vs 69 min in SC).
Primary outcome – Irritability, Depression and Anxiety (IDA) scale
Total IDA scores were compared between groups at the 5 time-points (Table II). There was a small difference in median total IDA scores at 7 days in favour of VEM (z = 1.827, p = 0.068), but no significant differences were identified.
| Table II. Median and interquartile range (IQR, denoted by 25th and 75th percentile) for total Irritability, Depression and Anxiety score in very early mobilization and standard care groups at each time period |
| Time period | VEM Median (IQR) | SC Median (IQR) | z* | p* | Missing data and reason |
| 7 days (n = 48) | 9 (5, 17) | 15 (10.5, 20.5) | 1.827 | 0.068 | Dead (5); Communication problem (10); Unable† (5); No assessment† (3) |
| 14 days (n = 51) | 11 (5, 14) | 11.5 (8, 17.5) | 0.718 | 0.473 | Dead (6); Communication problem (9); Unable (4); No assessment (1) |
| 3 months (n = 45) | 10 (5, 17) | 12 (4, 15) | –0.057 | 0.755 | Dead (12); Communication problem (7); No assessment (6); Dropped out (1) |
| 6 months (n = 44) | 10.5 (6, 15) | 10.5 (7, 15) | 0.223 | 0.823 | Dead (14); Communication problem (5); No assessment (6); Dropped out (2) |
| 12 months (n = 44) | 10 (8, 13) | 14 (7, 16) | 0.636 | 0.525 | Dead (17); Communication problem (6); No assessment (2); Dropped out (2) |
| *Differences between groups assessed using Wilcoxon rank-sum (Mann-Whitney U) test. †Unable: e.g. adverse event, palliated, disorientated, poor consciousness, coma, poor concentration. No assessment: e.g. refused, not available. VEM: very early mobilization; SC: standard care. |
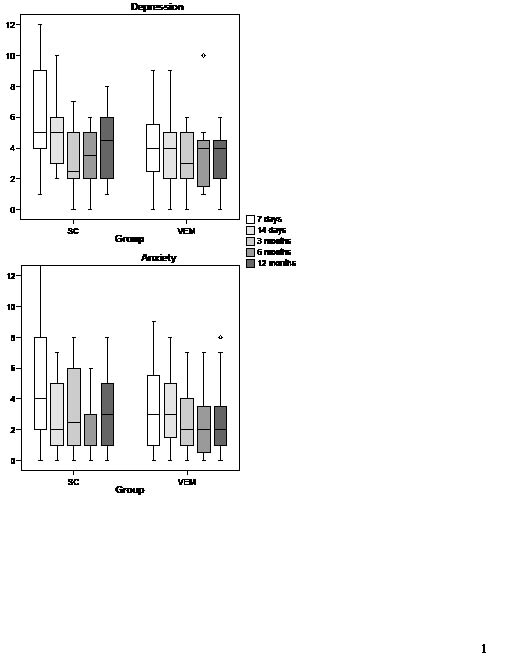
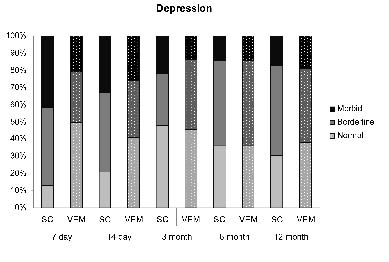
Scores on the depression and anxiety subscales of the IDA over the 12-month period following stroke are shown in Fig. 2. Group differences were identified at 7 days, where VEM patients were less depressed (z = 2.511, p = 0.012; Mann- Whitney U-test) and marginally less anxious (z = 1.791, p = 0.073; Mann-Whitney U-test) than SC patients. At day 7, depressive symptoms were significantly more common in SC (3 normal, 11 borderline, 10 morbid) than in VEM (12 normal, 7 borderline, 5 morbid) patients (χ2 = 7.96, p = 0.019). Fig. 3 illustrates the depression classifications at all time periods. Using multivariable backward stepwise logistic regression, VEM was associated with less depression at 7 days (odds ratio (OR) 0.14, 95% confidence interval (CI) 0.03 to 0.61, p = 0.009). The group difference in depression was not attributable to pre-stroke mood disorder: pre-morbid depression or anxiety was noted in 3/36 VEM patients (2 histories not available) and 5/33 SC patients. Inclusion of pre-stroke mood disorder as a factor in the multivariable regression did not alter the results.
Fig. 2. Box-plots of medians, interquartile ranges (25th–75th percentile) and ranges for the depression and anxiety components of the Irritability, Depression and Anxiety scale in VEM (very early mobilization) and SC (standard care) groups at each time period.
Fig. 3. Percentage of patients classified as normal, borderline or morbid on the depression component of the Irritability, Depression and Anxiety scale in VEM (very early mobilization) and SC (standard care) groups at each time period.
Again using multivariable logistic regression, there was no association between anxiety at 7 days, or any other covariate, and VEM group. Anxiety symptoms were similar for SC (13 normal, 6 borderline, 5 morbid) and VEM (17 normal, 5 borderline, 2 morbid) patients at 7 days (χ2 = 1.91, p = 0.385).
No significant differences between groups were identified for either inward or outward irritability at any time-point. Cases of irritability were relatively rare, both at 7 days (inward irritability: 4/48 morbid (3 VEM, 1 SC); outward irritability: 1/48 morbid (0 VEM, 1 SC)) and 12 months (inward irritability: 2/44 morbid (2 VEM, 0 SC); outward irritability: 2/44 morbid (1 VEM, 1 SC)).
Factors associated with depression and anxiety
Table III outlines the univariable and multivariable associates of depression. At 7 days, assignment to the SC group and female gender were independently associated with depression. At 12 months, female gender and poor 12-month mRS were the factors associated with depression. Patients with a good outcome on the mRS at 12 months were less likely to be depressed than those with a poor outcome (10/22 (45%) vs 19/22 (86%); χ2 = 8.19, p = 0.004).
| Table III. Univariable and multivariable logistic regression of factors associated with depression at 7 days and 12 months post-stroke |
| | 7 days | 12 months |
| Univariable | Multivariable† | Univariable | Multivariable† |
| β | SE | p | β | SE | p | β | SE | p | β | SE | p |
| VEM group | –1.95 | 0.74 | 0.009 | –2.13 | 0.80 | 0.008 | –0.34 | 0.64 | 0.593 | | | |
| Age | –0.02 | 0.03 | 0.491 | | | | 0.01 | 0.02 | 0.630 | | | |
| Gender | –1.57 | 0.73 | 0.033 | –1.79 | 0.81 | 0.027 | –2.08 | 0.85 | 0.014 | –1.81 | 0.89 | 0.043 |
| High impairment | 0.51 | 0.65 | 0.432 | | | | 0.06 | 0.69 | 0.934 | | | |
| Previous stroke | –0.12 | 0.79 | 0.881 | | | | 1.07 | 0.86 | 0.212 | | | |
| Poor mRS* | 0.02 | 0.90 | 0.869 | | | | 2.03 | 0.75 | 0.007 | 1.79 | 0.80 | 0.025 |
| Walking status* | –1.60 | 0.85 | 0.059 | | | | – | – | 1.000 | | | |
| *Poor mRS – pre-morbid/12 months; Walking status – 7 days/12 months. †Multivariable analysis is adjusted for all other variables in the column. High impairment refers to a score of 8 or more on the National Institute of Health Stroke Scale; poor mRS is a score of 3–5 on the modified Rankin Scale (mRS). β: β coefficient; SE: standard error; VEM: very early mobilization. |
Variables related to anxiety are shown in Table IV. Female gender and not walking unassisted by 7 days were the factors most strongly associated with anxiety at 7 days. Note the difference between the borderline significant influence of group on anxiety score at 7 days (p = 0.073; Mann-Whitney U-test) reported previously and the reduced effect of group when anxiety is a dichotomized outcome variable (p = 0.236; univariable regression). Female gender and prior stroke were independently associated with anxiety at 12 months post-stroke; poor outcome on 12-month mRS was not related to anxiety at this time-point.
| Table IV. Univariable and multivariable logistic regression of factors associated with anxiety at 7 days and 12 months post-stroke |
| | 7 days | 12 months |
| Univariable | Multivariable† | Univariable | Multivariable† |
| β | SE | p | β | SE | p | β | SE | p | β | SE | p |
| VEM group | –0.72 | 0.61 | 0.236 | | | | –0.25 | 0.63 | 0.690 | | | |
| Age | –0.01 | 0.02 | 0.751 | | | | –0.03 | 0.01 | 0.195 | | | |
| Gender | –2.44 | 0.71 | 0.001 | –2.34 | 0.78 | 0.003 | –1.43 | 0.66 | 0.032 | –1.52 | 0.72 | 0.033 |
| High impairment | 0.55 | 0.60 | 0.366 | | | | –0.08 | 0.65 | 0.907 | | | |
| Previous stroke | –0.89 | 0.87 | 0.304 | | | | 1.54 | 0.74 | 0.037 | 1.66 | 0.80 | 0.038 |
| Poor mRS* | 0.94 | 0.83 | 0.256 | | | | 0.39 | 0.63 | 0.532 | | | |
| Walking status* | –1.40 | 0.67 | 0.036 | –1.53 | 0.79 | 0.052 | – | – | 1.000 | | | |
| *Poor mRS – pre-morbid/12 months; walking status – 7 days/12 months. †Multivariable analysis is adjusted for all other variables in the column. High impairment refers to a score of 8 or more on the National Institute of Health Stroke Scale; poor mRS is a score of 3–5 on the modified Rankin Scale (mRS). β: β coefficient; SE: standard error; VEM: very early mobilization. |
Discharge destination and living arrangements in the 12 months post-stroke were recorded. Twenty-eight percent of patients were discharged home, 55% were discharged to a rehabilitation hospital, and the remaining 17% were discharged elsewhere or died in hospital. At 7 days, 17 patients (24%) had returned home. These factors were not associated with depression or anxiety in this sample.
Adverse events
By the 12-month follow-up, 17 patients had died and 2 had dropped out (see Table II). More patients in the intervention group had died (VEM 11/38; SC 6/33), but this was not statistically different. All deaths in the first 3 months were due to stroke, and those who died were more severely affected (median baseline NIH score: 22) than those who survived (median baseline NIH score: 8). The number of falls and other serious adverse events was similar in the 2 groups (see Bernhardt et al. (13) for full details).
Discussion
Patients exposed to the VEM intervention had fewer depressive symptoms than controls at 7 days post-stroke. The association between intervention group and less depression at the 7-day time-point remained significant when multivariable logistic regression was used to adjust for other factors that may explain differences in psychological well-being among stroke patients. Initial analyses indicated a marginal effect of very early mobilization on reduced anxiety at 7 days, but multivariable logistic regression yielded no independent association between very early mobilization and anxiety at this time-point. No significant differences between groups were found for depression or anxiety beyond 7 days, and no reliable group differences in irritability were identified at any stage. Although the mobilization intervention could potentially last up to 14 days, it did not continue beyond hospital discharge, which was typically 6–7 days post-stroke (13). The beneficial effect of mobilization on depression, therefore, appeared to be confined to the intervention phase and did not persist beyond 7 days post-stroke. In healthy populations, it is accepted that physical activity reduces depression, but whether it reduces depression once exercise ceases is unclear (20, 21). The fact that initial “reactive” depression was less prevalent in the VEM group may indicate the potential to manage or treat this component of depression. Influencing the sustained depression that is present beyond the acute phase is also possible (22), but might require an intervention that is specifically targeted at reducing depressive symptoms.
Across the whole sample, approximately 30% of patients were classified as morbidly depressed in the first 14 days, falling to 15–20% at later time-points. These levels are consistent with earlier findings (7). Depression was more common than anxiety, which is in agreement with previous studies (23, 24), although Fure et al. (9) did report more anxiety than depression at 7 days post-stroke. It should be noted that scores on the IDA and its subscales do not correspond to clinical diagnoses. Also, completion of the IDA questionnaire requires a certain level of comprehension and communication, and as such our results may not be generalizable to aphasic patients. Aphasic patients were not excluded from the study, but failure to complete the IDA as a result of communication problems was an issue, particularly at the 7- and 14-day time-points (see Table II). In the absence of a gold standard for assessing post-stroke mood disorders, particularly in those with recent stroke, the IDA scale was chosen for its brevity and its avoidance of somatic symptoms that might be mimicked by stroke.
To determine the factors associated with depression and anxiety, logistic regression analyses were performed using the full sample. At 7 days, the factors independently associated with depression were intervention group and gender, indicating that SC patients and women were more likely to be depressed. At 12 months, the factors associated with depression were female gender and disability, as measured by the mRS at 12 months. The relationship between physical disability and post-stroke depression is well established (25). Factors associated with greater anxiety at 7 days post-stroke were female gender and not being able to walk unassisted at 7 days. Associates of anxiety at 12 months were female gender and having had a prior stroke. The finding that women were more anxious than men following stroke is consistent with previous research (26). Disability at 12 months was not significantly associated with anxiety at 12 months, even in univariable analysis. Although walking status early after stroke was an important factor in a patient’s anxiety levels, physical disability at 12 months post-stroke was related only to depression and not to anxiety.
The most striking and novel finding of this study was the significant and independent effect of very early mobilization on depression at 7 days post-stroke. There are several possible explanations for this effect. The first account relies on mediating factors: very early mobilization may reduce medical complications and disability, and as a result, patients may experience less depression. This is unlikely to provide a full explanation, however, as the effect of intervention group on depression remained when baseline stroke severity, pre-morbid disability and unassisted walking status at 7 days were adjusted for in the multivariable analysis. The second explanation is that an increase in movement and physical activity directly reduces the risk of depression. This has certainly been shown to be true in healthy populations (20, 21). It remains unclear whether the mechanisms behind an exercise effect are chiefly physiological (e.g. via changes in endorphin and monoamine concentrations; 10) or psychological (e.g. via the fostering of an optimistic outlook and enhancement of self-efficacy; 12). Finally, it is possible that reduced depression in the VEM patients can be attributed to the extra companionship and attention they received in the additional mobilization sessions. Refuting this possibility requires a follow-up study that includes a second control group, in which patients receive extra sessions of attention and conversation but no added mobilization.
It should be noted that multiple group comparisons were made in this study, and therefore we cannot avoid a heightened risk of false positive findings (Type I errors). To reduce this risk, depression and anxiety at 7 days and 12 months were set as primary outcome measures, with the other time-points and measures of irritability included only for peripheral interest. Furthermore, the strength of the relationship between exposure to VEM and depression at 7 days in multivariable analysis (p = 0.009) means it is unlikely to be a Type I error. Nevertheless, caution is required when interpreting the current results.
Depression in stroke survivors is a major concern. Post-stroke depression has been linked to a range of negative outcomes, including less participation in rehabilitation, poorer rate and extent of recovery, and increased mortality. Exactly how important the prevention of depression in the early period after stroke is to long-term outcome is not known. The current results suggest that depression in acute stroke patients can be influenced by a simple mobilization intervention, and the potential benefits of a reduction in depression at this early stage warrant further research.
Acknowledgements
We want to thank all the patients and clinicians who participated in this study. We are particularly grateful to Dorcas Quah, for her work as blinded assessor and impeccable recall of patient details, and Carolyn Luke, for her help in locating medical records. The Phase II trial was supported by grants from the National Heart Foundation of Australia (Grant Number G 04M 1571), Affinity Health and an equipment grant from the Austin Health Medical Research Fund. Dr Bernhardt was supported by a National Health and Medical Research Council (Australia) fellowship (157305).
References
1. Diserens K, Michel P, Bogousslavsky J. Early mobilisation after stroke: review of the literature. Cerebrovasc Dis 2006; 22: 183–190.
2. Bernhardt J, Dewey H, Thrift A, Donnan G. Inactive and alone: physical activity within the first 14 days of acute stroke unit care. Stroke 2004; 35: 1005–1009.
3. Allen C, Glasziou P, Del Mar C. Bed rest: a potentially harmful treatment needing more careful evaluation. Lancet 1999; 354: 1229–1233.
4. Indredavik B, Bakke F, Slordahl SA, Rokseth R, Haheim LL. Stroke unit treatment improves long-term quality of life: a randomised controlled trial. Stroke 1998; 29: 895–899.
5. Indredavik B, Bakke F, Slordahl SA, Rokseth R, Haheim LL. Treatment in a combined acute and rehabilitation stroke unit: which aspects are important? Stroke 1999; 30: 917–923.
6. Salaycik KJ, Kelly-Hayes M, Beiser A, Nguyen AH, Brady SM, Kase CS, et al. Depressive symptoms and risk of stroke: the Framingham Study. Stroke 2007; 38: 16–21.
7. Hackett ML, Yapa C, Parag V, Anderson CS. Frequency of depression after stroke: a systematic review of observational studies. Stroke 2005; 36: 1330–1340.
8. Paul SL, Dewey HM, Sturm JW, Macdonell RAL, Thrift AG. Prevalence of depression and use of antidepressant medication at 5-years poststroke in the North East Melbourne Stroke Incidence Study. Stroke 2006; 37: 2854–2855.
9. Fure B, Wyller TB, Engedal K, Thommessen B. Emotional symptoms in acute ischemic stroke. Int J Geriatr Psychiatry 2006; 21: 382–387.
10. Thornen P, Floras JS, Hoffman P, Seals DR. Endorphins and exercise: physiological mechanisms and clinical implications. Med Sci Sports Exerc 1990; 22: 417–428.
11. Bandura A, editor. Self-efficacy. New York: WH Freeman; 1997.
12. Robinson-Smith G, Johnston MV, Allen J. Self-care self-efficacy, quality of life, and depression after stroke. Arch Phys Med Rehabil 2000; 81: 460–464.
13. Bernhardt J, Dewey H, Thrift A, Collier J, Donnan G. A Very Early Rehabilitation Trial for Stroke (AVERT): phase II safety and feasibility. Stroke 2008; 39: 390–396.
14. Brott T, Adams HP, Olinger CP, Marler JR, Barson WG, Biller J, et al. Measurements of acute cerebral infarction: a clinical examination scale. Stroke 1989; 20: 864–870.
15. Bernhardt J, Chan J, Nicola I, Collier JM. Little therapy, little physical activity: rehabilitation within the first 14 days of organised stroke unit care. J Rehabil Med 2007; 39: 43–48.
16. Bernhardt J, Chitravas N, Meslo IL, Thrift AG, Indredavik B. Not all stroke units are the same: a comparison of physical activity patterns in Melbourne, Australia and Trondheim, Norway. Stroke, 2008; 39: 2059–2065.
17. de Haan R, Limburg M, Bossuyt P, van der Meulen J, Aaronson N. The clinical meaning of Rankin ‘handicap’ grades after stroke. Stroke 1995; 26: 2027–2030.
18. Snaith RP, Constantopoulos AA, Jardine MY, McGuffin P. A clinical scale for the self-assessment of irritability. Br J Psychiatry 1978; 132: 164–171.
19. Aylard PR, Gooding JH, McKenna PJ, Snaith RP. A validation study of three anxiety and depression self-assessment scales. J Psychosom Res 1987; 31: 261–268.
20. Strawbridge WJ, Deleger S, Roberts RE, Kaplan GA. Physical activity reduces the risk of subsequent depression for older adults. Am J Epidemiol 2002; 156: 328–334.
21. Kritz-Silverstein D, Barrett-Connor E, Corbeau C. Cross-sectional and prospective study of exercise and depressed mood in the elderly. Am J Epidemiol 2001; 153: 596–603.
22. Watkins CL, Auton MF, Deans CF, Dickinson HA, Jack CIA, Lightbody CE, et al. Motivational interviewing early after acute stroke. Stroke 2007; 38: 1004–1009.
23. Sturm JW, Donnan GA, Dewey HM, MacDonell RAL, Gilligan AK, Srikanth V, et al. Quality of life after stroke: the North East Melbourne Stroke Incidence Study (NEMESIS). Stroke 2004; 35: 2340–2345.
24. Angelelli P, Paolucci S, Bivona U, Piccardi L, Ciurli P, Cantagallo A, et al. Development of neuropsychiatric symptoms in poststroke patients: a cross-sectional study. Acta Psychiatr Scand 2004; 110: 55–63.
25. Hackett ML, Anderson CS. Predictors of depression after stroke: a systematic review of observational studies. Stroke 2005; 36: 2296–2301.
26. Morrison V, Pollard B, Johnston M, MacWalter R. Anxiety and depression 3 years following stroke: demographic, clinical, and psychological predictors. J Psychosom Res 2005; 59: 209–213.
27. Bamford J, Sandercock P, Dennis M, Burn J, Warlow C. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet 1991; 337: 1521–1526.