OBJECTIVE: To assess the effectiveness of pulsed electromagnetic fields compared with placebo in the management of osteoarthritis of the knee.
DATA SOURCES: A systematic review of PubMed, EMBASE, and the Cochrane Controlled Trials Register.
METHODS: Randomized, controlled trials reporting on the blinded comparison of pulsed electromagnetic fields with placebo were included. Validity was tested according to the Jadad Scale. Studies were pooled using fixed-effects and random-effects models after exclusion of publication bias and assessment of heterogeneity. Sensitivity analyses and meta-regression were performed to test the stability of our findings.
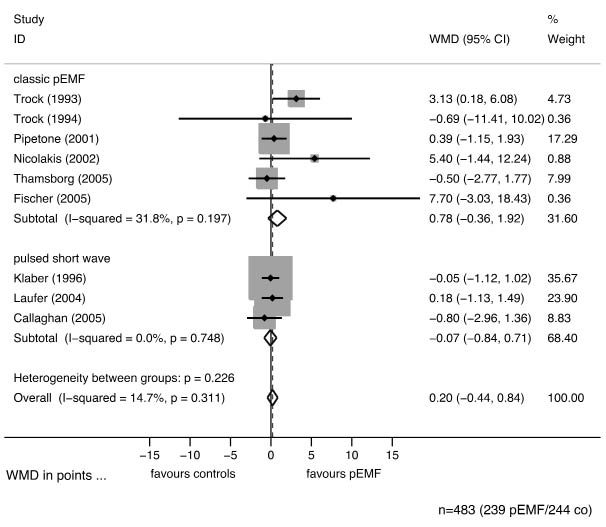
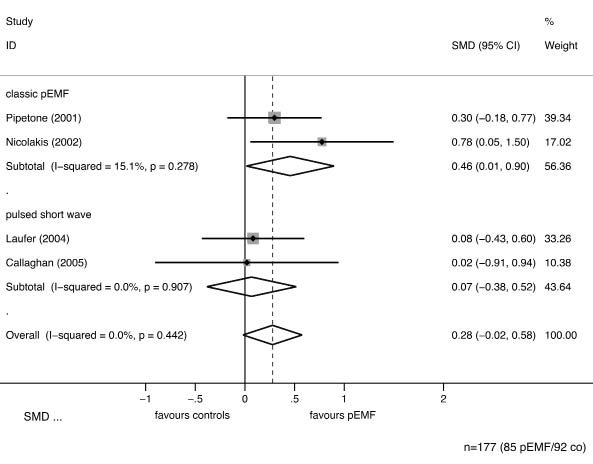
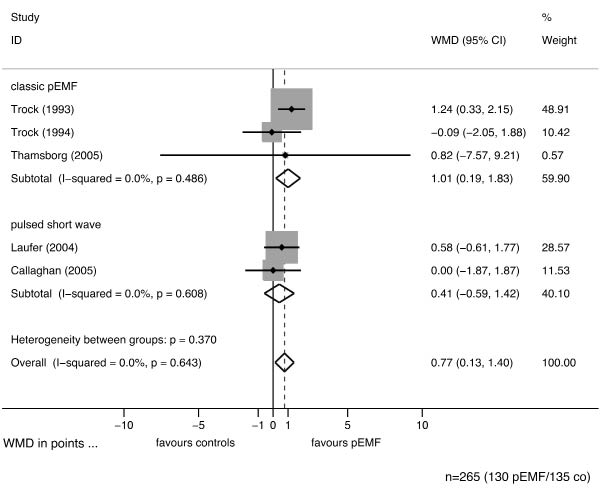
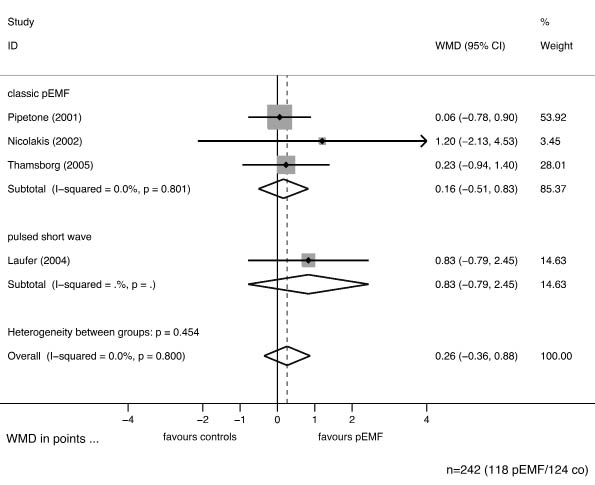
RESULTS: Nine studies, including 483 patients, were pooled. No significant difference could be shown for pain (weighted mean difference 0.2 patients; 95% confidence interval (CI): –0.4 to 0.8) or stiffness (weighted mean difference 0.3; 95% CI: –0.3 to 0.9). There was a significant effect on activities of daily living (weighted mean difference 0.8; 95% CI 0.2–1.4, p = 0.014) and scores (standardized mean difference 0.4; 95% CI: 0.05–0.8, p = 0.029). We saw only statistically insignificant differences between studies with different treatment protocols.
CONCLUSION: Pulsed electromagnetic fields improve clinical scores and function in patients with osteoarthritis of the knee and should be considered as adjuvant therapies in their management. There is still equipoise of evidence for an effect on pain in the current literature.
Key words: pulsed electromagnetic fields, osteoarthritis, meta-analysis, evidence-based medicine.
J Rehabil Med 2009; 41: 406–411
Correspondence address: Patrick Vavken, Department of Orthopedic Surgery, Children’s Hospital Boston, 300 Longwood Avenue, Enders 1016, Boston, MA 02115, USA. E-mail: Patrick.vavken@childrens.harvard.edu
Submitted November 10, 2008; accepted March 2, 2009
INTRODUCTION
Osteoarthritis (OA) is a major burden on our society, and there is a dire need for effective treatments. In the conservative management of OA, especially, a variety of therapy options has been developed, and among physical treatment options, pulsed electromagnetic fields (pEMF) has received a good deal of attention in both clinical and basic research during recent years (1–3). This therapy has been employed successfully in numerous clinical settings concerning the musculoskeletal apparatus, ranging from fracture healing to chronic pain (1, 4). A number of studies shed light on the cellular processes involved in the response of chondrocytes to pEMF, and showed positive effects on bioactivity and glycosaminoglycan metabolism (5, 6). The precise mechanisms of pEMF and even its major biological effect(s) are still elusive, however. Randomized, controlled studies investigating the clinical effect of pEMF in the management of osteoarthritis have produced somewhat contradictory results (7–15). One of the reasons for this, and probably the major problem in this field, is the dearth of clearly and unequivocally described algorithms. The reports on the methods used vary considerably in details such as duration of treatment, energy levels, and other parameters. The question remains as to whether pEMF is a therapy by itself or rather an umbrella term. The latter, if true, would mean that we must expect different effects and need to address this fact accordingly in study design.
In 2006, McCarthy et al. (16) presented a concise systematic review and meta-analysis that refuted the effectiveness of pEMF and was in conflict with a number of sufficiently sized, appropriately designed and conducted studies. We decided to repeat this meta-analysis for 2 reasons. Firstly, new evidence from randomized, controlled trials is now available that has not been included previously. Secondly, we believe McCarthy et al. might possibly have biased their own results through study selection. These researchers excluded non-English studies and 2 randomized, controlled trials (14, 15) because of a potential bias, but without actually testing for its existence. Thus, among inconsistent randomized trials and a potentially biased meta-analysis, the clinical problem as to whether pEMF therapy is effective in the management of OA of the knee, remains unanswered.
The primary objective of this study was to assess the effectiveness of pEMF in the management of OA of the knee, as measured by a reduction in pain, clinical scores, activities of daily living (ADL), and stiffness. The secondary objective was to assess whether there is evidence for differences in effects due to the existing differences in treatment protocols described in published studies.
MATERIAL AND METHODS
This study follows the guidelines of the QUOROM (Quality Of Reporting Of Meta-analyses) Statement.
Search strategy and study selection
A systematic online review of PubMed, EMBASE, and the Cochrane Controlled Trial Register was performed using exploded MeSH terms. The search terms used were “electromagnetic”, “pulsed electromagnetic”, “osteoarthritis”, and “gonarthrosis”. To avoid bias, searches were not restricted by publication date or language. Additionally, experts in the field were interviewed to identify further important information from meetings or not yet published or rejected papers. We included full text papers reporting on randomized, controlled trials comparing pEMF with placebo in the management of arthritis of the knee in humans. An expert in the field (OS) assessed the methods from all relevant papers. All searches were concluded by January 2008.
Validity assessment
The internal validity of the included trials was assessed using a modified Jadad scale (17). This score attributes 1 point each for randomization, blinding, and description of attrition.
Data abstraction
We extracted data for the end-points pain, and clinical scores. Additionally we extracted data on stiffness and activity of daily living to complement information given by clinical scores, and avoid erroneous interpretation due to overlap or conflicting results in isolated components and score totals. All end-points were abstracted for a time-point as close as possible to 6 weeks of follow-up from the included studies. Data abstraction was performed in duplicate using digital spreadsheets (PV, FA) and cross-checked. In case of disagreeing values the original data of the items in question were reviewed by both investigators (PV, FA) for errors during abstraction. If this process did not resolve disagreement the senior author (RD) was consulted to arbitrate or, if necessary, to provide a decision.
Statistical analysis and quantitative data synthesis
Publication bias was assessed graphically and mathematically using funnel plots and Egger’s regression. Extracted data were assessed for statistical heterogeneity using Cochrane’s Q-test and the I2-index. The latter reveals the percentage of total variability that is truly to be attributed to in between study heterogeneity, whereas the Q-test simply produces a binary outcome and has rather low power. The threshold of significance for the Q-test was set at p = 0.10 to account for this. Individual trial estimates were pooled using random-effects or fixed-effects models, depending on results from the heterogeneity tests.
The fixed-effects models were built using weighted averages of the differences in means of outcomes, using the inverse variance as weights, for weighted mean differences (WMD) of outcomes reported on the same scale to give the reader a quantifiable outcome. For outcomes reported on different scales, a pooled effect size was calculated using Cohen’s d and given as standardized mean difference (SMD). The DerSimonian-Laird method was used for random-effects models.
Beyond merely testing for heterogeneity we also wanted to explore its possible sources, and their influence on the pooled estimates. Thus, we reviewed the methods of the included studies and stratified them into those with a clear description of pEMF therapy and those describing methods inconsistent with classic pEMF but rather pulsed short wave units. Sensitivity analyses were then done based on this stratification, and the amount of heterogeneity between groups was assessed. A common effect was assumed if no heterogeneity could be found. Furthermore, the influence of individual studies was tested by excluding one study at a time and calculating pooled estimates from all other studies. The estimates from the individual studies were then compared with their pooled counterparts, as well as the pooled estimates amongst each other, searching for significant changes. To control for potential confounding, meta-regressions were performed for patient age and percentage of female patients (18). Unfortunately, there was insufficient information to control for body mass index.
All results are presented as summary estimates of weighted mean differences or standardized mean differences with 95% confidence intervals (CI). A p-value ≤ 0.05 was considered significant. All calculations were performed using intercooled Stata 10 (StataCorp LP, College Station, TX, USA).
RESULTS
Trial flow and characteristics of included studies
Our search produced 55 studies, all of which were obtained and reviewed for inclusion in duplicate. Ten randomized, controlled studies were identified and their methods were assessed specifically. One study was excluded because its methods were not consistent with pEMF in the study’s own description (19). Finally, 9 studies, published between 1993 and 2005 in English and German and reporting on a total of 483 patients (239 pEMF vs 244 controls), were included into the analysis (7, 9–16) (Table I). All included studies reached the maximum score on the modified Jadad Scale.
| Table I. Characteristics of the individual included studies |
| Study (reference) | Total | Age | BMI | Female |
| pEMF n | Control n | pEMF Mean (SD) | Control Mean (SD) | pEMF Mean (SD) | Control Mean (SD) | pEMF % | Control % |
| Laufer et al. 2005 (9) | 27 | 31 | 72.7 (6.7) | 73.3 (6.9) | – | – | 91 | 66 |
| Thamsborg et al. 2005 (13) | 42 | 41 | 60.4 (8.7) | 59.6 (8.6) | 27 (4) | 27.5 (5.7) | 61 | 46.5 |
| Fischer et al. 2005 (8) | 34 | 35 | 58.1 (11.079) | 62.1 (8.7) | 29.2 (5.889) | 29.4 (4.32) | 71 | 72 |
| Callaghan et al. 2005 (7) | 9 | 9 | 63.5 (7.9) | 58.3 (7.3) | 27.2 (4.5) | 26.8 (3.9) | 44 | 56 |
| Nicolakis et al. 2002 (11) | 15 | 17 | 69 (5) | 67 (7) | – | – | 73 | 47 |
| Pipetone & Scott 2001 (12) | 34 | 35 | 62 | 64 | | | 35 | 20 |
| Klaber Moffet et al. 1996 (10) | 26 | 22 | 62.7 (8.69) | 63.5 (10.53) | – | – | 63 | 63 |
| Trock et al. 1994 (15) | 42 | 44 | 69.24 (11.48) | 65.82 (11.66) | – | – | 69 | 70 |
| Trock et al. 1993 (14)* | 10 | 10 | – | – | – | – | – | – |
| *Author reports no significant differences in gender, mean age, mean body weight. BMI: body mass index; SD: standard deviation; pEMF: pulsed electromagnetic fields. |
Thamsborg et al. (13) reported in 2005 on the results of a randomized, placebo-controlled, double-blinded study of 83 patients, treating 42 with pEMF, and following them for 6 weeks. Fischer et al. (8) reported on a randomized, controlled, blinded study of 69 patients with 10 weeks’ follow-up. Laufer et al. (9) compared 31 sham treatments with 37 low and 27 high-energy treatments in a double-blind trial and followed them for 3 months. The article by Callaghan et al. (7) reported on 18 patients in a randomized, controlled, blinded study. Pipetone & Scott (12) included 69 patients in his study and followed them for 6 weeks. Klaber Moffet et al. (10) had the longest follow-up with 12 weeks in their randomized, controlled, blinded study of 48 patients. Nicolakis et al. (11) reported in his randomized, controlled, blinded study on 32 patients. Trock et al. (15) published 2 studies on this subject. In 1994 they reported on the largest study in this analysis with 86 patients, after a previously published study of 20 patients (14). Both studies were randomized, controlled, and blinded and had 4 weeks’ follow-up. The parameters of the methods (pulsed short wave) used by Klaber Moffet et al., Callaghan et al., and Laufer et al. are not consistent with pEMF as employed by the other studies. Pulsed short wave units apply electromagnetic fields of high frequencies (27 MHz), whilst the other pEMF-units apply electromagnetic fields of not more than 3000 Hz. The possible therapeutic mechanism of both methods is till now speculative, but might be different between both methods. Sensitivity analyses were subsequently carried out to account for this circumstance (Table II).
| Table II. Characteristics of the treatments groups in the individual studies |
| Study (reference) | Frequency | Intensity | Treatment regimen | Duration of treatment |
| Laufer et al. 2005 (9) | 27 MHz pulsed | 18W | 20 min 3 times a day | 3 weeks |
| Thamsborg et al. 2005 (13) | 50 Hz | 10 mV/cm | 2 h daily | 6 weeks |
| Fischer et al. 2005 (8) | 10–300 Hz | 3.4–13.6 uT | 16 min daily | 6 weeks |
| Callaghan et al. 2005 (7) | 27 MHz pulsed | 10–20 W | 20 min | 6 sessions in 2 weeks |
| Nicolakis et al. 2002 (11) | 1–3000 Hz | 40 uT | 30 min twice a day | 6 weeks |
| Pipetone & Scott 2001 (12) | 3–7.8 Hz | 50 uT | 10 min 3 times a day | 6 weeks |
| Klaber Moffet et al. 1996 (10) | 27 MHz pulsed | 23 W | 15 min 3 times a week | 3 weeks |
| Trock et al. 1994 (15) | 5–12 Hz | 1–2.5 mT | 30 min | 18 sessions |
| Trock et al. 1993 (14)* | 30 Hz | 1–2 mT | 30 min | 8 weeks |
| *Author reports no significant differences in gender, mean age, mean body weight. |
Statistical analysis and quantitative data synthesis
There was no indication for publication bias in funnel plots or Egger’s regression. Assessment of between study heterogeneity produced no evidence for heterogeneous effects (Figs 1–4), thus a fixed-effects model was created for data synthesis. Figs 1–4 give the forest plots for the end-points pain, clinical scores, ADL, and stiffness pooled for all studies and with subgroups by type of treatment, together with the p-values for the Q-test and values for I2. Stratification by different described treatments did not show evidence for differences in treatment effects for pain (p = 0.237), Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores (p = 0.100), ADL (p = 0.416), or stiffness (p = 0.299). There was no evidence for confounding of treatment effects by age or gender. Table III provides p-values for pooled estimates and potential confounders. There was no indication of overly or unduly high influence of any study among the pooled articles.
Fig. 1. Forest plot of the meta-analysis of pain giving weighted mean differences in points on a visual analogue scale (VAS) with 95% confidence intervals (CI) graphically and numerically, stratified by the description of methods. The upper block summarizes all studies with clearly described methods consistent with pulsed electromagnetic fields (pEMF). The lower block summarizes studies with not clearly described methods that might be different from classical pEMF. The diamond shapes represent the 95% CI of the pooled estimates. If this estimate crosses the line of null effect (vertical line) then there is no significant effect at the 5% level. However, there is no indication of difference between their effects (p = 0.237). The shaded areas represent the weight of the study, the dashed line gives the pooled estimate.
Fig. 2. Forest plot of the meta-analysis of clinical scores, stratified by description of methods. The subgroup of studies clearly using pulsed electromagnetic fields (pEMF) shows a significant effect (p = 0.029). In order to pool results from studies reporting on different scores (Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) or Arthritis Impact Measurement Scale (AIMS)), standardized mean differences are given. These are the same as the effect size. The shaded areas represent the weight of the study, the dashed line gives the pooled estimate.
Fig. 3. Forest plot for activities of daily living (ADL) with weighted mean differences (WMD) and 95% confidence intervals (CI), stratified by potentially clinically heterogeneous methods. However, since the 95% CI of the pooled estimates in the subgroups (diamond shapes) overlap, there is no evidence for a difference in effects in classic pulsed electromagnetic fields (pEMF) and pulsed short wave. The pooled estimate is clearly significant (p = 0.014) and in favour of pEMF. The shaded areas represent the weight of the study, the dashed line gives the pooled estimate.
Fig. 4. Stratified forest plot for stiffness with weighted mean differences (WMD) and 95% confidence intervals (CI). Again, we stratified for potential clinically heterogeneity in methods, but found no evidence for such differences in effects. The overall pooled estimate shows no significant effect, represented by the diamond shape widely overlapping the line of null effect (vertical line), and the 95% CI, which contains null. The shaded areas represent the weight of the study, the dashed line gives the pooled estimate.
| Table III. Pooled estimates (overall and by type of treatment). Data were pooled overall and stratified by classic pulsed electromagnetic fields (pEMF) and pulsed short wave to account for possible differences in effectiveness. The potential confounders age and gender were assessed in a meta-regression, but showed no significant association with treatment effects |
| End-point | Scale | Grouping | Pooled estimates | 95% CI | p-value | Effect of age | Effect of gender |
| Pain (VAS) | WMD | Overall | 0.2 | –0.4–0.8 | 0.511 | 0.992 | 0.766 |
| | | pEMF | 0.8 | –0.4–1.9 | 0.178 | | |
| | | Pulsed short wave | –0.1 | –0.8–0.7 | 0.909 | | |
| Scores | SMD | Overall | 0.3 | –0.02–0.6 | 0.067 | 0.683 | 0.470 |
| | | pEMF | 0.5 | 0.01–0.9 | 0.043 | | |
| | | Pulsed short wave | 0.1 | –0.4–0.5 | 0.771 | | |
| ADL | WMD | Overall | 0.8 | 0.1–1.4 | 0.014 | 0.679 | 0.714 |
| | | pEMF | 1.0 | 0.2–1.8 | 0.016 | | |
| | | Pulsed short wave | 0.4 | –0.6–1.4 | 0.333 | | |
| Stiffness | WMD | Overall | 0.3 | –0.4–0.9 | 0.353 | 0.648 | 0.471 |
| | | pEMF | 0.2 | –0.5–0.8 | 0.636 | | |
| | | Pulsed short wave | 0.8 | –0.8–2.5 | 0.190 | | |
| CI: confidence interval; VAS: visual analogue scale; WMD: weighted mean difference; SMD: standardized mean difference; ADL: activities of daily living. |
DISCUSSION
The principal findings of the present study were a significant effect of pEMF on scores 6 weeks after treatment, which seems to be attributed mostly to ADL, with an average effect size of 0.4, i.e. a medium effect, in contrast to poorer results for stiffness. We did not detect a significant difference in pain between treatment and controls. However, it is important to note that the pooled estimate has a fairly wide confidence interval, and future studies might show a significant effect without disagreeing with our findings. This effect is likely to be close to 0.8 patients, but might be as high as 1.9. Such a difference in pain would be consistent with a better to much-better effect, according to the study by Wells et al. on experiences and expectations of patients with rheumatoid arthritis (20).
A previous meta-analysis produced somewhat different results and presented a quite different interpretation (16). Although this study was well done and reported, we decided to repeat it for 2 reasons. The first reason is that new evidence is available, and we were able to include 483 instead of 276 patients. The second reason is potential selection bias. One potential source of bias is language bias, since we found 2 studies published in German that were not included. Secondly, McCarthy et al. (16) decided to exclude the studies published by Trock et al. in 1993 and 1994 (14, 15) arguing that their results might have been deviated by the use of a non-validated instrument of outcome measurement. While we agree with McCarthy that this might be a possible source of bias, we think that this assumption should be tested before excluding studies. We found no evidence for biased results in these studies, thus we decided to include them. We did not, however, include studies of questionable quality. We used the Jadad score to test internal validity of all studies, and found results that were clearly higher than expected in wide ranges of clinical musculoskeletal research (21). In the light of these facts we were not surprised to find somewhat different results than McCarthy et al. (16).
Our findings suggest that pEMF have a clinical relevance as a successful adjuvant option in the management of OA rather than a stand-alone therapy. Results from meta-analyses on alternative options in the conservative management of OA of the knee can be used to construct a league table of therapies. Stronger effects compared with placebo have been shown for intra-articular corticosteroids at 6 weeks (WMD of 7.1 on a 0–100 mm visual analogue scale (VAS)), for tramadol with less than 8 weeks of follow-up (WMD of 7.6 on a 0–100 mm VAS), and for rofecoxib at 6 weeks (WMD of 2.83 on a 0–100 mm VAS) (22–24). The results for pain reduction with pEMF were higher than those reported for acetaminophen (WMD –0.25) or physical therapy (WMD 0.5) compared with placebo (25, 26). The treatment effect of pEMF on WOMAC scores (0.4), in turn, was fairly similar to those of intra-articular corticosteroids (0.3) or tramadol (0.34) (22, 23). Importantly, there have been no reports of side-effects of pEMF other than numbness during treatments, or of initial pain increase with treatment (12). Thus pEMF have a clearly superior safety profile. Finally, interpretation of such comparisons of treatments across different studies has to be done cautiously and potential bias must be considered, since there may be significant differences in the studied populations.
It should be considered that treatment algorithms for pEMF therapy are fairly heterogeneous clinically, which might reflect in treatment effects. We defined a secondary research question to account for and test this possibility. We focused on between-study heterogeneity and influence based on stratification by described methods. We could not find evidence for heterogeneity among the included studies. Our sensitivity analyses, however, suggest that there might be some difference in effects. Another potential shortcoming of our analysis is the fairly small number of included studies, as reflected by the considerably large standard errors and wide confidence intervals. It has to be remembered that a statistically not significant result with a wide confidence interval, i.e. the absence of evidence for a beneficial effect, must not be confused with evidence for the absence of any effect (27). Future studies or meta-analyses including more patients might narrow down the confidence intervals and show significant effects with the full support of our findings (28). Furthermore, this circumstance limits the possibility to study subgroups by energy levels or other parameters of treatment in order to produce recommendations for future studies.
The question remains as to whether new findings, even if they were significant, would be of clinical significance. To date the exact mechanisms of pEMF are still elusive and are ascribed to different events at the cellular and tissue level (5, 6). The time to onset and subsequent longevity of pEMF effects should be considered in future study design to achieve an accurate measurement of results. A clearer definition of the mechanisms might also help in choosing patients who are more likely to benefit from such a treatment.
In conclusion, our study provides evidence for a beneficial effect of pEMF on scores in general and ADL in specific for patients with OA of the knee. Our results showed no significant effect on pain, but left enough space for improvement to significant values in forthcoming, larger studies (28). These findings suggest that pEMF might be a useful and effective addition to the conservative management of OA of the knee. Future studies are needed to prove this assumption right or wrong.
Conflict of interest
None of the authors has conflict of interest to disclose.
REFERENCES