OBJECTIVE: The aim of this study was to evaluate whether community-dwelling chronic stroke patients wearing an ankle-foot orthosis would benefit from changing to functional electrical stimulation of the peroneal nerve.
METHODS: In 26 community-dwelling chronic (> 6 months post-onset) patients after stroke, their ankle-foot orthosis was replaced by a surface-based functional electrical stimulation device (NESS L300®). Comfortable walking speed over 10 m was measured at baseline with the ankle-foot orthosis and after 2 and 8 weeks with both ankle-foot orthosis and functional electrical stimulation. The level of physical activity was assessed with a pedometer, and patients’ satisfaction was assessed with a questionnaire at baseline and at week 8 regarding ankle-foot orthosis and functional electrical stimulation, respectively.
RESULTS: Ankle-foot orthosis and functional electrical stimulation were equally effective with regard to walking speed and activity level. The participants were more satisfied with functional electrical stimulation than with their ankle-foot orthosis regarding the effort and stability of walking, quality of the gait pattern, walking distance, comfort of wearing and appearance of the device.
CONCLUSION: The patients judged functional electrical stimulation superior to their ankle-foot orthosis, but measurements of walking speed and physical activity could not objectify the experienced benefits of functional electrical stimulation. Other outcome measures focusing on the stability and effort of ambulation may objectify the perceived benefits of functional electrical stimulation in community-dwelling chronic stroke patients.
Key words: electric stimulation; rehabilitation; stroke; walking; foot; peroneal nerve.
Correspondence address: A. C. H. Geurts, Department of Rehabilitation, Radboud University Nijmegen Medical Centre,, PO Box 9101, 6500 HB Nijmegen, The Netherlands. E-mail: A.Geurts@reval.umcn.nl
J Rehabil Med 2010; 42: 117–121
Submitted April 7, 2009; accepted September 29, 2009
INTRODUCTION
The standard of care for stroke patients with a paresis of the ankle dorsiflexor muscles (a so-called drop foot) is the prescription of an ankle-foot orthosis (AFO). Although an AFO provides a stable support of the ankle joint, its disadvantage is that it limits normal ankle mobility and reduces the adjustments of the foot and ankle to the walking surface.
Functional electrical stimulation (FES) of the peroneal nerve is an alternative to an AFO. Muscles that dorsiflex and evert the ankle are activated during the swing phase of gait. As early as 1961, Liberson et al. (1) reported the use of peroneal FES, but, for a long time afterwards, its use in clinical settings was limited due to technical and ergonomic problems. With recent technological advances, a renewed interest has grown in the application of peroneal FES in patients with stroke. The question now arises as to whether, in clinical practice, peroneal FES is beneficial to patients who regularly use an AFO.
To justify the replacement of an AFO, FES should be at least as effective as an AFO. There is convincing evidence for beneficial effects of FES on the gait speed in stroke patients with a drop foot (2–8). However, most of these studies investigated the effects of FES compared with walking without any ankle device, whereas stroke patients with a drop foot are usually provided with an AFO or raised orthopaedic footwear.
The number of studies comparing FES with an AFO is limited and the results are not conclusive as to whether FES has additional value over an AFO with regard to walking speed (5, 9–12). Two studies found positive effects (5, 12), but in one of these studies, FES was not strictly compared with an AFO, as a number of patients used orthopaedic footwear or no device at all (5). In the other study, not only stroke patients, but also patients with other central neurological diseases were included (12). Furthermore, both Kottink et al. (5) and Waters et al. (12) investigated the effects of an implanted FES device. Such studies typically select patients who have responded well to transcutaneous stimulation. As a result, these studies suffer from selection bias concerning the question as to whether peroneal FES should generally be preferred over an AFO in chronic patients after stroke.
A second consideration in interpreting the literature on FES concerns the outcome measures. The effects of FES are usually expressed in terms of walking speed. Indeed, in the evaluation of gait, walking speed is a valuable outcome measure, in which many other gait parameters are captured (13, 14). Another measure that has been used is the level of daily physical activity, but the results with regard to the effects of FES are inconclusive (5, 15). There may be other outcomes, such as patients’ preference and satisfaction, which are relevant in the clinical decision as to whether to replace an AFO with peroneal FES (16). Although some investigators have already described that patients were generally satisfied with a peroneal FES device (4, 15, 17, 18), this has never been contrasted with AFO use.
Hence, the aim of this study was to determine whether patients with chronic stroke who regularly use an AFO would benefit from changing to transcutaneous peroneal FES. In a within-subjects design, we compared a transcutaneous FES device with a custom-made AFO with regard to walking speed, activity level, and patients’ satisfaction. We included community walkers only, because these patients are most likely to benefit from, and make extensive use of, the FES device in daily life.
METHODS
Participants
Twenty-six patients with chronic stroke who were referred to an academic hospital participated in this study. All patients suffered from a drop foot due to a stroke at least 6 months prior to recruitment. As an inclusion criterion, they had to regularly use a (polypropylene) AFO, with which they did not experience any problems. An additional inclusion criterion was independent walking ability without a walking aid for more than 10 min. Exclusion criteria related to the paretic body side were: a passive range of ankle motion less than 30 degrees, inability to load the heel while standing with an extended knee, severe hypertonia of the calf (Modified Ashworth Scale scores 4 and 5), inability to stimulate the superficial or deep peroneal nerves, and skin lesions at the electrode sites. Other exclusion criteria were: pregnancy, psychological disorders (depression or psychosis), and a demand-type pacemaker. The study protocol was approved by the medical ethics committee of the Arnhem-Nijmegen region. Participants gave their written informed consent before participation in the study.
Protocol and outcome measures
At inclusion, clinical assessments were conducted with regard to gait (Functional Ambulation Categories) (13), balance (Berg Balance Scale) (19), lower extremity hypertonia (Modified Ashworth Scale) (20), muscle strength (Motricity Index) (13), and motor selectivity (Fugl-Meyer Assessment) (21). Furthermore, sensation of the lower limb was evaluated by means of the Quantitative Vibration Threshold (22). Vibration was administered at the lateral malleolus and the first metatarsophalangeal joint of the paretic leg with a semi-quantitative tuning fork (Rydel Seiffer, Neurologicals®, Arno Barthelmes & Co, GmbH, Tuttingen, Germany). The lowest level of vibration that was perceived by the patient was recorded.
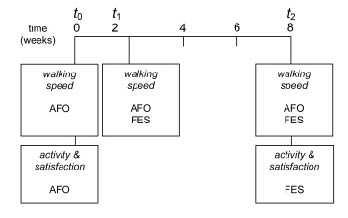
At baseline (t0), comfortable walking speed with the AFO was assessed (Fig. 1). The participants performed 8 walking trials on a wooden walkway. Length, width and height of the walkway were 10, 1.25 and 0.15 m, respectively. Mean speed for each participant was computed over the 8 trials. In the same session, the FES-device (NESS L300®) was adjusted to the patient. The NESS L300® is a transcutaneous, 2-channel system, stimulating the peroneal nerve and the anterior tibial muscle (pulse rate 30 Hz; phase duration 200 µs). A 2-week period of adaptation to this device followed, in which the participant increased the use of FES up to 6 h a day. After the 2-week adaptation period (t1), the comfortable walking speed was assessed both with the AFO and with the FES device. Subsequently, in weeks 3 to 8 of the study protocol, the patients followed a schedule to increase the daily use of FES to “whole day long”, although they were still required to use the AFO for 1 h a day (in order not to “unlearn” how to walk with the AFO). At week 8 of the study (t2), comfortable walking speed was assessed again with the AFO and with FES.
Fig. 1. Time line of the study, indicating the assessments of patients’ satisfaction, physical activity and walking speed at baseline (t0), at week 2 (t1) and at the final week of the study period (t2). AFO: ankle-foot orthosis; FES: functional electrical stimulation.
In the week preceding the baseline gait assessment (t0), the patient completed a questionnaire assessing the individual use of, and satisfaction with, the AFO. To this aim, a purpose-designed questionnaire was used, consisting of 9 aspects regarding the orthosis, which had to be evaluated on a 5-point scale (from very unsatisfied to very satisfied). The questionnaire was completed at home with no input from either the researchers or the clinicians involved with the study. During the same week, the patient also monitored his or her level of physical activity. The number of steps taken per day was counted by means of a pedometer (Yamax Digiwalker SW-650, Yamax USA, Inc, San Antonio, USA) and averaged over 7 days.
In the final week of the study period (t2), the patient completed the same questionnaire assessing the individual use of and satisfaction with the NESS L300®. During this week, the patient again monitored his or her level of physical activity, but now with FES, in order to complete the second assessment.
Statistical analysis
An analysis of variance (ANOVA) with repeated-measures was performed to compare walking speed between FES and AFO. Device (2 levels, AFO and FES) and time (2 levels, t1 and t2) were used as within-subjects factors. Differences in the number of steps taken per day between the AFO and FES were tested with a paired t-test. For each of the items of the questionnaire, a non-parametric test for 2 related samples was conducted to identify differences in satisfaction between the AFO and FES. The alpha level was set at 0.05.
RESULTS
Participants
The characteristics of the patients are shown in Table I. All patients had a Functional Ambulation Categories score of 5 (independent ability to walk over uneven terrain) and were used to walking with an AFO. The AFO was a custom-made polypropylene brace, which limited plantar flexion, but allowed dorsiflexion mobility at the ankle in all patients except for 2 patients who wore an AFO merely providing an external dorsiflexion moment about the ankle. Two patients dropped out of the study. Reasons for drop-out were discomfort from the electrical stimulation (n = 1) and allergic skin reaction at the electrode sites (n = 1).
| Table I. Characteristics of the participants |
| Variable | |
| Subjects, n | 26 (2)b |
| Age, years, mean (range) | 52.8 (21–68) |
| Time post-stroke, months, mean (range) | 38 (7–105) |
| Gender, male/female, n | 21 (2)b/5 |
| Body weight, kg, mean (range) | 84 (57–131) |
| Body height, m, mean (range) | 1.77 (1.53–1.89) |
| Hemisphere of stroke, left/right, n | 15 (1)/11 (1)b |
| Type of stroke, infarction/haemorrhage, n | 19 (2)b/7 |
| Modified Ashworth Score (0–5)a, median (range) Knee flexors/extensors Ankle plantar flexors/dorsiflexors | 0 (0–4) /0 (0–2) 0.5 (0–3)/0 (0–1) |
| Motricity Index (0–100), median (range)a | 64 (27–83) |
| Fugl-Meyer Assessment (% functional recovery), median (range)a | 66 (21–93) |
| Quantitative Vibration Thresholda (0 = no sensation; 8 = normal), median (range) First metatarsophalangeal joint Lateral malleolus | 4.5 (0–8) 5 (1–8) |
| Berg Balance Scale (0–56), median (range) | 53 (41–56) |
| Baseline comfortable walking speed with AFO, m/s, mean (SD) | 1.02 (0.05) |
| aScores of the paretic body side. bNumber of drop-outs (between brackets) are included in the total numbers. AFO: ankle-foot orthosis; SD: standard deviation. |
Walking speed
Walking speed was not different between the AFO and FES (1.00 (standard deviation (SD) 0.05) and 1.02 (SD 0.05) m/s for AFO; 0.98 (SD 0.05) and 1.03 (SD 0.05) m/s for FES, at t1 and t2, respectively) as indicated by the absence of a significant main effect of device (F(1,23) = 0.068; p = 0.796) or time × device interaction effect (F(1,23) = 1.049; p = 0.316). A significant main effect of time indicated that walking speed at t2 was, on average, 0.04 m/s higher than at t1 (F(1,23) = 6.30; p = 0.02).
Physical activity level
There was no significant difference in the number of steps per day between the AFO and FES (t(23) = 0.609; p = 0.548; 5541 (SD 2900) and 5733 (SD 2516) steps, respectively).
Satisfaction
Table II summarizes the patients’ satisfaction with the AFO at baseline and with FES at the end of the study. The participants experienced benefits of FES over their conventional walking device with regard to comfort, appearance of the device, quality of the gait pattern, walking distance, effort of walking, and stability during gait (all p-values < 0.05). For all these items, the median score for FES was 4, whereas the median score for the AFO was 3, except for the stability during gait that was 3.5. Participants were not significantly more satisfied about FES compared with AFO regarding getting the device on and off, ease of use, and going up and down the stairs (median scores of 4 on all items with FES and 3–3.5 with AFO), although all scores tended to be higher for FES.
| Table II. Median scores (ranges) of patients’ satisfaction with walking device |
| | Baseline (AFO) | Week 8 (FES) |
| Ease of getting on and off | 3.5 (2–5) | 4.0 (2–5) |
| Comfort to wear | 3.0 (2–4) | 4.0 (2–5)* |
| Ease of use | 3.0 (2–5) | 4.0 (2–5) |
| Appearance | 3.0 (1–4) | 4.0 (3–5)* |
| Quality of the gait pattern | 3.0 (2–4) | 4.0 (2–5)* |
| Walking distance | 3.0 (2–4) | 4.0 (2–5)* |
| Effort of walking | 3.0 (2–4) | 4.0 (2–5)* |
| Stability during gait | 3.5 (2–5) | 4.0 (2–5)* |
| Going up/down the stairs | 3.0 (2–5) | 4.0 (2–5) |
| *Significantly better scores with functional electrical stimulation (FES) than with the ankle-foot orthosis (AFO). Satisfaction was scored on a 5-point scale (1 = very unsatisfied; 2 = unsatisfied; 3 = neutral; 4 = satisfied; 5 = very satisfied). |
Problems experienced with functional electrical stimulation
One patient dropped out of the study due to allergic reactions to the electrodes. An additional 3 participants experienced skin irritations under the electrodes for short periods of time. Other minor problems that were encountered using FES were: discomfort of the device below the knee (n = 7), tendovaginitis of the peroneal muscles at the level of the lateral malleolus (n = 1), and muscle soreness (n = 5). These problems were transient and did not prevent the patients from completing the study.
DISCUSSION
The aim of the present study was to investigate whether community-dwelling patients with chronic stroke who were regularly using an AFO would benefit from changing to transcutaneous peroneal FES. The results showed that patients judged FES superior to their AFO, without concomitant differences in walking speed or level of daily activity. The patients judged FES superior to some key features of walking ability, which may indeed not translate to a higher walking speed. The participants experienced more stability of gait with FES, which may be particularly relevant during activities such as walking over uneven terrain or on inclines. A second aspect of gait on which the patients experienced a benefit of FES was the effort of walking, as patients reported that walking with FES was “less fatiguing”. Several studies indeed demonstrated a reduction in energy expenditure with FES, although this was never compared with an AFO (2, 4, 6, 8, 23). In addition, our findings are in line with those from previous studies investigating patients’ satisfaction (4, 18). In these studies, patients were also satisfied about the stability and the effort of walking with FES, but the present study is the first to demonstrate that patients were more satisfied with FES than with their AFO.
The increase in stability and reduction in effort experienced may be explained by several properties of the NESS L300®. First, in contrast to most AFOs, this FES device does not limit ankle mobility, permitting easier balance reactions and plantar flexion movements during loading and push-off. Furthermore, peroneal FES may improve the stereotyped movement pattern of the paretic leg by reduction of spasticity (24–29) or triggering of the flexion reflex (30–33). The stability as well as the effort of gait may benefit from such an improved movement pattern.
With respect to walking speed, no differences were observed between the AFO and FES. The small increase in walking speed at t2 compared with t1 was probably due to a slightly decreased walking ability shortly after the application of the FES device. Gait speed was restored to baseline level at t2 (Table I). A separate ANOVA of the 3 AFO measurements at t0, t1 and t2 showed that there was no significant time effect on walking with the AFO, indicating that there was no influence of FES on walking with the AFO. These findings are in agreement with previous studies, in which no differences were found between AFO and FES (9–11). In contrast, other studies found a significantly higher speed when walking with FES (5, 12). This discrepancy may be explained by the study populations. The participants in the current study had a mean comfortable walking speed of 1.02 m/s at baseline, whereas the patients in the studies of Kottink et al. (5) and Waters et al. (12) walked at a mean speed of 0.70 and 0.58 m/s, respectively. An already high baseline walking speed, as in the patients in our study, may yield a ceiling effect (3, 5). From this perspective, it is even more interesting that our patients experienced clear benefits from FES. The difference between our results and the results of previous studies (5, 12) may also be related to the fact that, in those studies, an implanted FES system was used, whereas the patients in our study used a transcutaneous device. Transcutaneous and implanted FES have not yet been compared directly, so it cannot be excluded that effects with respect to walking speed may differ between these types of devices. More importantly, as mentioned in the introduction, studies investigating the effects of implanted devices are bound to suffer from (positive) selection bias considering the general comparison between peroneal FES and AFO.
As for the level of physical activity, no change was found in the number of steps per day between the devices. The finding that physical activity did not increase with FES is in line with the observations of Kottink et al. (5). It suggests that the walking distance reflects a person’s functional aims, and that in patients with chronic conditions these have become adjusted to their walking ability over time. It may, therefore, be that an 8-week study protocol is too short for true behavioural changes. In contrast, Laufer et al. (7) did find improvements in self-reported physical functioning in activities of daily living and participation in community life after 8 weeks of using FES. The baseline level of physical functioning, however, was probably much lower in their patients than in ours, as indicated by the mean walking speed (0.65 m/s). Furthermore, our study sample was already quite active with ~5,600 steps per day compared with the average of only ~2,800 steps per day in individuals with chronic stroke (34). So it may well be that due to a ceiling effect our participants did not increase their activity level.
As a disadvantage of transcutaneous FES, skin problems are mentioned in the literature. In this study, 4 participants (15%) developed skin problems, one of whom dropped out of the study for this reason. Both higher and lower incidences of skin problems with FES have been reported in the literature (4, 18, 23). The larger numbers may be related to prolonged use of FES, but other factors may also be involved, such as the conductive substance used in the electrodes or genetic differences in skin types.
This study has some limitations. We used a purpose-designed questionnaire to assess patients’ satisfaction, which has face validity but which has not been formally validated. Furthermore, besides gait speed, no other gait variables were included to objectify possible benefits of FES. Particularly in the case of good walking ability, future studies should include other outcome measures, e.g. related to gait endurance or the ability to negotiate uneven surfaces.
In conclusion, this study aimed to compare the efficacy of transcutaneous peroneal FES with a conventional AFO in order to support clinicians in their decision whether to replace an AFO with FES in community-dwelling patients with chronic stroke who suffer from a drop foot. The patients judged FES to be superior to the AFO, but measurements of walking speed and level of physical activity could not objectify the perceived benefits of FES. The patients experienced greater stability of gait with FES, which may be related to a feeling of safety during transfers, walking on inclines or over uneven terrain. FES was also judged superior with respect to the effort of gait.
Conflict of interest: The authors have no conflict of interest to declare. They received the necessary L300® devices on loan during the study as well as a small unrestricted grant to conduct this study from Ness Netherlands BV (now Bioness®). In addition, this study was supported by a small unrestricted grant from Unu BV in the Netherlands. The authors have no other involvement with any organization with interest in the subject matter.
REFERENCES