OBJECTIVE: The aim of this observational study was to explore whether patients with traumatic peripheral nerve injury of the hand perceive referred sensations; sensations that are perceived to emanate from other areas of the body than the part being stimulated. Referred sensations have been reported following amputation, somatosensory deafferentation, local anaesthesia, stroke, brachial plexus avulsion injury, spinal cord injury and complex regional pain syndrome type 1.
DESIGN: Ten patients with ulnar or median nerve injuries underwent sensory testing of the face, upper body and legs, involving light touch with a cotton swab. Patients were asked to describe the location of the stimulated site, the sensations emanating from it and any other sensations experienced. Three patients with referred sensations were identified and followed over a period of time.
RESULTS: Clear and reproducible referred sensations were found in 3 out of 10 patients examined.
CONCLUSION: Referred sensations were found in traumatic nerve injury, providing evidence of reorganization of the central nervous system after peripheral injury.
Key words: referred sensation; arm injury; neuroplasticity.
J Rehabil Med 2010; 42: 357–361
Correspondence address: Ruud W. Selles, Erasmus MC, University Medical Center Rotterdam, Department of Rehabilitation Medicine, Room h.016, PO Box 2040, NL-3000 CA Rotterdam, The Netherlands. E-mail r.selles@erasmusmc.nl
Submitted September 30, 2009; accepted December 14, 2009
INTRODUCTION
In peripheral nerve injuries, acute deafferentation of the hand has immediate and long-standing influences on the corresponding hand areas in the brain cortex as well in adjacent cortical territories (1, 2). This cortical and sub-cortical functional reorganization is important, since it is considered to be a key factor in explaining the poor recovery of motor and sensory functions of the hand following peripheral nerve injury (3).
A phenomenon that has been reported repeatedly in deafferentation after upper limb amputation and which may be a clinical sign of cortical representation after deafferentation is “referred sensations”. In referred sensations, a sensation is felt in a place other than the site at which a stimulus is applied. The term referred sensation was first used by Rivers & Head (4) in 1908 to describe the changed representations within the area of innervation after a self-inflicted peripheral nerve injury. Since that time, referred sensations have been described in a wide range of patient groups, including patients with upper limb amputations (5), lower limb amputation (6), complex regional pain syndrome (CRPS) (7), stroke (8) and brachial plexus avulsion injury (9). As an example of these referred sensations, when some upper limb amputee patients are touched on the ear, cheek or shoulder with a cotton swab, they may also report feeling sensations in the amputated phantom hand. In patient populations such as CRPS and amputees it has been suggested that there is a correlation between pain, especially phantom pain, and referred sensations (7, 10, 11).
As described extensively by Ramachandran & Hirstein (5), referred sensations generally follow a somatotopic pattern of reorganization; that is, a pattern that can be predicted based on the classical homunculus representation. For example, in the above-mentioned amputee patients, the ear, cheek or shoulder are adjacent to the hand in the sensory cortex. However, Borsook et al. (12) and Grusser et al. (11) evoked referred sensations in arm amputees, not only in a somatotopical pattern, but also in a non-somatotopical pattern. In addition, Moore et al. (13) described referred sensations in a non-somatotopical reorganization in patients with spinal cord injury.
A nerve injury of the median and ulnar nerve represents a similar acute deafferentation of the hand to that of an amputation. This would suggest that, as in amputee patients, patients with peripheral nerve injuries should have similar referred sensations. However, to our knowledge, this has not been investigated. Therefore, the aim of this study was to explore whether patients with traumatic peripheral injury perceived referred sensations. We also determined whether the locations of any referred sensations followed the pattern predicted based on the somatotopical map, or whether referred sensations were also found at other locations (11–13). We report here 3 cases with evidence of referred sensations.
METHODS
Participants
Patients with a surgically repaired complete median and/or ulnar nerve injury of the hand were invited to participate. The reconstructive surgery had been performed in the first 48 h after injury in all patients. All patients received standard hand therapy treatment without special attention to sensory re-education. The study was approved by the institutional medical ethics committee and all patients gave their written informed consent.
Measurements
Ten patients were investigated while sitting or, whenever possible, were placed in a supine position. Sensory loss was tested with Semmes and Weinstein monofilaments at the injured extremity and at the contra-lateral extremity.
To test for the presence of referred sensations, all patients underwent sensory testing using touching with a cotton swab, first with their eyes closed and then with their eyes open, in random order at the face, arms, chest and legs. Each time the subject was touched, they were asked to describe exactly the location of the stimulated site, the sensations emanating from it and any other sensations experienced. If a patient reported referred sensations on the first examination with a cotton swab, a further examination was performed, testing for referred sensations using pinprick for a duration of approximately 1 s, vibration sense with a 256 Hz tuning fork, and airflow. The referred sensations were drawn on a schematic diagram of the body and noted down. Patients were told that the aim of this study was to evaluate the sensitivity of the body after peripheral injury. They were not informed beforehand of the possibility of experiencing referred sensations.
Pain was assessed using a visual analogue scale (VAS), which consisted of a 10-cm line anchored by 2 extremes of pain, with zero representing no pain and 10 representing unbearable pain. Pain was defined as every form of pain in the hand.
RESULTS
During the inclusion period of 1 year 10 patients were included and examined. Subjects were tested at varying time intervals after injury. In 3 patients, clear and reproducible sensations were found that could be interpreted as referred sensations. The remaining 7 cases were examined with a cotton swab. If they did not present referred sensations during this examination we did not proceed to the examination with pinprick, vibration sense and airflow. We describe below the 3 patients with referred sensations.
Case 1
The first subject was a 23-year-old man who had sustained a glass injury of his right underarm. Due to this injury he had a lesion of the ulnar and median nerve, the ulnar, median and radial artery, and the flexor tendons. Within 24 h of injury the tendons, nerves and vascular system were micro-surgically repaired.
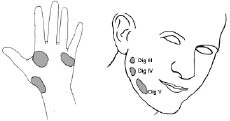
Five days after injury, referred sensations were detected during examination with a cotton swab. At this time, the patient had his lower arm in a cast. He had complete loss of sensation in all fingers of his right hand and at the palmar side of his hand and did not report any pain. The patient described a tingling sensation and pressure in his right injured hand when he was touched with a cotton swab in his contra-lateral hand (see left part of Fig. 1). The locations on the injured side corresponded exactly with the other hand. In addition, when touching his right cheek with a cotton swab, he sensed a pressing, tingling feeling in his third, fourth and fifth finger of his injured hand that could be evoked repeatedly (see right part of Fig. 1). After discharge from the hospital the patient did not attend any therapy sessions and could not be reached for further follow-up.
Fig. 1. Referred sensations in case 1, 5 days after median and ulnar nerve injury of his right arm. Left: the areas on the palmar side of the left (uninjured) hand where, when being touched, the patient described a tingling sensation and pressure at the exact same position at the injured hand. Right: the areas on the face where he sensed a pressing, tingling feeling on his third (III), fourth (IV) and fifth (V) finger of his injured hand when he was stimulated.
Case 2
The second subject was a 19-year-old man who had sustained a glass injury of his right lower arm, approximately 10-cm distal to the elbow crease. Due to this injury all of the flexor muscles, as well as the radial, median and ulnar nerves of his right lower arm were cut. Surgery was performed within 24 h of injury.
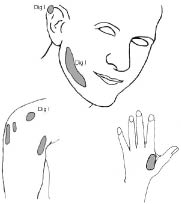
We saw him 5 weeks after injury. There was complete loss of sensation at the radial side of his lower arm and thumb. On the VAS he scored 1 out of 10. During the examination he reported referred sensations at 9 different locations on the ipsilateral (right) cheek, ear, and shoulder (see left part of Fig. 2). More specifically, when touched with a cotton swab on his ipsilateral cheek he described an itching sensation at the top of his ipsilateral index finger. On pinprick testing on his shoulder, he described a warm feeling in his thumb and an involuntary movement of the thumb. With air blowing on his ipsilateral shoulder he reported a cramping feeling at the top of his right thumb. Vibration on his ipsilateral shoulder produced an unconscious movement of the right thumb and perception of movement of the hand. In addition, he reported referred sensations from his contralateral ear.
Fig. 2. Left: the referred sensation locations in case 2, 5 weeks after a combined radial, median and ulnar nerve injury. When stimulated on the grey areas with a cotton swab, pinprick, vibration, or air, the patient described itching, warm sensations, cramping or perceived movement of the deafferented thumb. Right: the same subject 9 weeks after the initial nerve injury but 2 weeks after a new surgical procedure and immobilization. When testing with the cotton swab, vibration and pinprick at the dorsal side of his uninjured hand on the area illustrated, the patient reported similar sensations at the same location on his injured hand.
Seven weeks after the injury we saw him again. He used his hand more often and frequently rubbed his hand. He still had complete loss of sensation of his lower arm and thumb. At this time, on cotton swab testing, he had only one referral site at his ipsilateral cheek, where he reported experiencing a warm, tingling feeling in the palm of his hand. These sensations could be emanated with his eyes open and closed and the patient was capable of evoking these sensations himself. The sensations were not evoked when applying pinprick, vibration or airflow. Stimulation at the previous referral sites from the prior measurements and on the rest of his body did not lead to referred sensations.
Nine weeks after the injury the patient was again operated, undergoing a nerve transfer to improve motor outcome. After this surgery, he received a cast and his arm was immobilized for 11 days. Two weeks after this second surgery we saw him again while he was still wearing a protective cast. He could flex his thumb and index finger better than before the procedure, but still had a complete loss of sensation of his right lower arm with minimal pain. The patient reported a tingling sensation at the top of his index finger when he was touched with a cotton swab at his ipsilateral cheek, as if the cotton swab was brushing his hand. When testing with the cotton swab on his non-injured hand the patient reported the same sensation on his injured hand. The sensations corresponded with the exact location on his injured hand (see right part of Fig. 2). When testing with pinprick at the contralateral hand, a pressing sensation was felt in his injured hand on the same location. Vibration at the healthy hand evoked a perception of vibration at the same place at his injured hand.
Ten weeks after the injury we performed a final assessment. The sensory loss had not changed. At this time, no referred sensations were reported during any of the stimulations. The patient had no pain and used his hand increasingly during daily life.
Case 3
The third subject was a 26-year-old women with a glass injury of her left hand. She had an almost complete (90%) lesion of the ulnar nerve and a complete lesion of the median nerve, in addition to lesions of the flexor tendons of all fingers. She had complete loss of sensation at the palmar side of her hand.
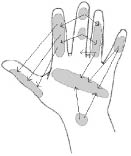
We examined this subject approximately 17 months after injury. The patient was still receiving outpatient rehabilitation and still had severe sensory loss at her left hand palm. Her hand was red and moist. She experienced typical neuropathic pain, and took amitriptyline to reduce the pain. The 0–10 VAS analogue pain score was 7. When testing with cotton swab, pinprick and tuning fork at different locations on her injured left hand, she repeatedly reported being touched at 2 spots simultaneously on the same hand (Fig. 3). The sensations at the referral site were described as tingling. With the pinprick, the sensations were less strong than with the cotton swab, but at the same location. When asked, the patient mentioned that, shortly after the injury, when touching or grasping something with her healthy hand it also felt as if she were simultaneously grasping something with her injured hand.
Fig. 3. The referred sensations in case 3, 17 months after complete injury of the ulnar and median nerve. When being touched at one location on the injured hand with a cotton swab, tuning fork or pinprick, the patient reported the feeling of being touched at 2 locations on this hand. These referred sensations are indicated by arrows, showing the locations of the stimulation (the start of the arrow) and the locations where the patient also perceived a sensation (the point of the arrow). The arrows are a sample of a large number of referred sensations reported in this case.
Twenty-seven months after injury we tested her again; she no longer had neuropathic pain and had stopped using amitriptyline. She used her hand in all her daily activities and had stopped receiving outpatient rehabilitation. On the VAS scale she scored 1 out of 10. The referred sensations at cotton swab stimulation showed the same pattern as at the first measurements, both at the dorsal and the palmar side of her hand. The patient described similar referred sensations, but in smaller regions, and the patient could evoke the referred sensations herself. During pinprick testing at the top of her second finger, she described the sensation of being touched with a thicker pin than was actually used. On some referral sites the subject sensed a soft touching of her finger. When testing with air blowing on the top of her third finger she sensed a soft touching of her fourth finger.
DISCUSSION
The aim of this study was to explore whether patients with traumatic peripheral injury perceived referred sensations in places other than the hand. We found 3 patients in which these referred sensation existed. The referred sensation locations followed a pattern that can be predicted based on the homuncular somatosensory body map, and were similar to most studies of referred sensations reported after arm amputation (14). These neural somatotopic patterns clearly differ from the real anatomical organization of the body. In none of our cases were referred sensations reported in the non-somatotopical way, as reported by Borsook et al. (12), Grusser et al. (11) and Moore et al. (13).
Various short- and long-term mechanisms have been proposed to explain cortical reorganization after nerve injury (15, 16). A key factor is the loss of sensory input (deafferentation) in cases of amputation and nerve lesions. At the central level this could cause denervation supersensitivity or disinhibition (e.g. in the thalamus and somatosensory cortex), resulting in activation of these areas from adjacent or heavily connected cortical areas via formerly silent synapses (17). At a later stage structural changes, such as sprouting and synaptogenesis, can change the connectivity within cortical neural circuits. These changes following loss of input may be an attempt by the organism to stay in contact with the body parts in spite of nerve lesions. Phantom pain and referred sensations could be a consequence of such a reorganization process. In our study, referred sensations were perceived at the ipsilateral cheek, ear, and shoulder and in the contralateral hand to the nerve injury, i.e. body areas that are adjacent or heavily interconnected in the brain.
The patients were not aware of the referred sensations before testing, and the pattern of the referred sensations changed over time, supporting the idea from other studies that mislocalization subsides when using the affected limb due to relearning to interpret somatosensory information accurately (14). Interestingly, the referred sensation could be found within days after the injury. For example, in our second case, we saw an increase in referral sites during the period of surgery and immobilization after the surgery, which decreased very rapidly following removal of the cast. This indicates that the referred sensations are not dependent on the longer-term cortical adaptation to injury.
In most studies, such as those of Ramachandran & Hirstein (5) and McCabe et al. (7), patients could not evoke the referred sensations themselves and the referred sensations were not perceived when the stimulation was made visible. In our study and in the study of Maihofner et al. (18), patients reported referred sensations with their eyes open and closed, but only after they were made aware of the referred sensations. In our study, none of the patients were aware of the referred sensations before we tested them. In all patients, the referred sensations were found when using cotton swabs. Referred sensations during pinprick, vibration and air blowing were also reported, but less frequently than after touch. This fits well with reports of referred sensation in other conditions (5, 8). The referred sensations in our subjects were found not only ipsilaterally, but also contralaterally, both for touch and vibration. Contralateral referred sensations might be explained by the fact that the hand is represented not only in the contralateral hemisphere but also in the ipsilateral hemisphere (2).
In other patient populations, such as patients with phantom pain after arm amputations or with CRPS, has been suggested that there is a correlation between pain, especially phantom pain, and referred sensations (10, 14, 18, 19). Maihofner et al. (18) reported 1 case out of 3 patients with CRPS type II with sensory mislocalization, and in our study there was one patient with referred sensations who also had CRPS type II. However, in a study on patients with brachial plexus avulsion injury and surgical repairs, Htut et al. (9) found no statistical significant difference between pain scores in patients with referred sensations and those in patients without referred sensations. Grusser et al. (11) did not find a significant correlation between non-painful referred sensations induced by painful or non-painful stimuli and cortical reorganization, and suggested that painful and non-painful phantom phenomena are mediated by different neural substrates. Based on the small number of subjects in our studies we are not able to relate the development of pain syndromes directly to the presence of referred sensations.
In addition to the small number of subjects and the irregular times of follow-up, a limitation of this study is that the assessment of referred sensations is a subjective procedure and may depend on how the examiner explains the assessment. Although the assessment was conducted using a standard protocol and the examiner tried to respond in as neutral a manner as possible to the information provided by the patients, there is always a risk that patients are influenced by the examiner.
Evidence for the cortical plasticity that is the basis of the phenomenon of referred sensations is extensive (20), indicating that the brain is a dynamic, continuously changing neural organization in which every motor output, sensory input or environmental context induces neural plastic changes, such as sensitization, reorganization and remapping. Future studies may indicate which individual differences could explain why some patients have referred sensations and other patients do not. Further research is required to determine the factors that influence recovery of hand function after nerve injury.
REFERENCES