Ada Tang, PhD1, Susan Marzolini, MSc2,4,5, Paul Oh, MSc4,5, William E. McIlroy, PhD2,4,5 and Dina Brooks, PhD2,3,4,5,6
From the 1Department of Physical Therapy, University of British Columbia, Vancouver, 2Institute of Medical Science, 3Department of Physical Therapy, University of Toronto, 4Toronto Rehabilitation Institute, Toronto, 5Heart and Stroke Foundation of Ontario Centre for Stroke Recovery, Sunnybrook Health Sciences Centre, Ontario and 6Department of Kinesiology, University of Waterloo, Waterloo, Canada
Objective: Community exercise programs are effective in improving aerobic capacity after stroke, but individual variability in training responses has not previously been studied. This study examined explanatory factors for post-program responses in aerobic capacity (VO2peak) with respect to training parameters and participant characteristics.
DESIGN: Secondary analysis of data from a prospective cohort study.
Subjects: Thirty-two participants, mean age 63 years (standard deviation (SD) 13), 29 months (SD 26) post-stroke.
Method: Following a 6-month community exercise program, multivariate regression analysis was performed to assess for correlates with change in VO2peak with respect to training parameters (exercise duration and intensity) and participant characteristics (lower limb and balance impairment, aerobic and walking capacity).
Results: The mean improvement in VO2peak was 11% (SD 19), but individually, changes ranged from 32% decline to 56% improvement. Regression analysis revealed that only 2 variables, baseline aerobic capacity and balance impairment, accounted for the training-related change in VO2peak (R2 = 0.20, p = 0.045).
Conclusions: There is considerable inter-individual variability in stroke exercise program responses in aerobic capacity. Lower baseline aerobic capacity and better balance were associated with greater training-related improvements in VO2peak. Identifying individuals who are more likely to respond to exercise programs may inform future work in establishing effective interventions suited for different subsets of the stroke population.
Key words: stroke; exercise training; oxygen consumption; outcome; rehabilitation
J Rehabil Med 2013; 45: 00–00
Correspondence address: Dr D Brooks, Department of Physical Therapy, University of Toronto, 160-500 University Avenue, Toronto Ontario, M5G 1V7 Canada, E-mail: dina.brooks@utoronto.ca
Submitted March 13, 2012; accepted July 11, 2012
Introduction
A Cochrane review and meta-analysis of aerobic exercise training trials for individuals with stroke have demonstrated that such training is effective in improving cardiorespiratory fitness (1, 2). Similarly, community-based stroke exercise programs (vs. institution or laboratory-based interventions) have demonstrated similar results of increased aerobic capacity (3, 4). We recently demonstrated the feasibility of adapting an existing outpatient, community-based cardiac rehabilitation program for individuals with mild to moderate disability following stroke and its effectiveness in increasing aerobic capacity (VO2peak) (5). As typical of most clinical trials, we reported overall results of post-program changes for the entire cohort but by doing so, individual differences are not captured. On closer examination of the data, we noted a large variability in individual participants’ responses to the exercise intervention. The factors that potentially influence post-training fitness gains have only been examined in a few studies, and no previous study has identified subgroups of response to aerobic exercise among individuals with stroke.
How much exercise is performed during the program, characterized by indices of exercise time or training intensity, can affect the extent to which gains in aerobic fitness are observed. Very little is known about outcome predictors following aerobic exercise intervention after stroke. Macko and colleagues reported that increasing treadmill speed predicted post-intervention gains in VO2peak after stroke, whereas progressing the duration of the training sessions predicted improvement in 6-Minute Walk Test (6MWT) distance (6). In other populations, higher intensity aerobic exercise was found to be more effective in increasing VO2peak among young, healthy individuals (7) and, in older adults, higher levels of volume of physical activity was associated with better cardiorespiratory fitness (8). Greater exercise frequency appears to play a role in greater aerobic gains post-myocardial infarction (9).
Furthermore, the neurological and functional sequelae that result from stroke may also influence the degree to which training-related improvements in fitness levels can occur. Individuals with significant functional limitations post-stroke may encounter greater challenges in achieving exercise-training targets, relative to those who are higher functioning. Lower limb motor impairment and challenges in balance control may also contribute to compromised ambulatory capacity and functional mobility, thereby contributing to sedentary behaviours outside of the exercise program. Indeed, impaired balance is a predictor for functional mobility among individuals with stroke (10) and slower walking speeds are associated with poorer function and community participation (11). These individuals likely continue in sedentary lifestyles, despite participating in regular exercise programs, and may not attain the same training-related benefits as those who engage in non-exercise physical activity (12).
The primary objective of this study was to examine correlates in subgroups of exercise responses following a community-based exercise program for individuals with stroke. It was hypothesized that exercise training parameters and participant characteristics related to baseline motor impairment, functional mobility and fitness level would be associated with post-intervention change (≥ or <10% change) in aerobic capacity (VO2peak). Findings from the present study will identify factors related to degree of training-related benefit observed among individuals with stroke, and may shape future trials to establish interventions geared for optimal benefit to different participant subgroups.
Methods
This study is a secondary analysis of a larger trial, the details of which are described elsewhere (5). In brief, the main trial examined the feasibility and effects of a 6-month community cardiac rehabilitation program of aerobic and resistance training, adapted for individuals with mild to moderate disability from stroke (Chedoke-McMaster Stroke Assessment leg impairment score ≥ 3 (indicating marked spasticity and weakness; a maximum score 7 indicates normal timing and coordination of complex movement patterns) (13)). Study participants were recruited from outpatient stroke rehabilitation programs, clinics and community programs. All subjects were at least 3 months post-stroke and able to walk at least 10 m independently with our without aids. Prior to the start of the program, all participants performed a symptom-limited graded maximal exercise test (on cycle ergometer or treadmill) to evaluate baseline fitness and to screen for cardiovascular abnormalities that would preclude participation in the program. A metabolic cart measured breath-by-breath gas exchange, and a ramp protocol was used with incremental increases in work rate. Heart rate and blood pressure were measured throughout the test, and an electrocardiogram was monitored for abnormalities. The American College of Sports Medicine (14) criteria for test termination were followed.
Details of the exercise program are described elsewhere (5). In brief, the 6-month program included once weekly, 90-min, supervised on-site exercise sessions, and at-home sessions 4 days per week that were done on their own time. A combination of aerobic exercise (30–60 min, 5 days per week) and resistance training (2–3 sessions per week) was used. Training modality was individually prescribed according to the participants’ abilities: overground walking, treadmill, cycle ergometer for the aerobic training, and dumbbells, resistance bands, or body weight for resistance training. The same exercises were performed during on-site and at-home sessions, where one aerobic training and one resistance training session per week were performed on-site and the remaining (4 aerobic training and 1–2 resistance training) sessions were performed independently at home. Intensity was monitored using 10-s pulse rate or heart rate monitors, and ratings of perceived exertion. Progression of the exercise training was prescribed and closely monitored by the program staff, who were trained in physical therapy and exercise science.
The participants also attended a program orientation session that included instruction on how to complete weekly exercise diaries that were used to monitor progress. All training sessions were tracked via weekly exercise diaries. Blank copies of the exercise diaries were distributed each week during the on-site visits, and completed diaries were returned the following week.
The current study is a secondary analysis of the data from the 32 participants who completed the program and had pre-post measures of VO2peak. Study procedures were followed in accordance to institutional guidelines and were approved by local university and hospital research ethics committees. Informed written consent was obtained from all participants for the larger trial.
Response subgroups
In the main trial, there was a significant overall post-training improvement in VO2peak (5), but upon closer inspection of individual participant data, a wide range in responses in VO2peak was noted. Percent change in VO2peak was calculated as [Post-training VO2peak – Baseline VO2peak]/Baseline VO2peak. The sample was divided into the following two subgroups:
Change in VO2peak was selected as the outcome of interest as it is the gold standard measure for evaluating aerobic capacity. In the absence of established thresholds for change in VO2peak in the stroke population, we turned the literature with healthy adults to justify our choice in using 10% change as the threshold for defining the subgroups, where an improvement of one metabolic equivalent (approximately 3.5 ml × kg–1 × min–1) is considered clinically important in healthy populations (15–17) and would represent approximately 10% of normative values for VO2peak among 60–70 year old adults (14, 18). Since this was the average age of participants enrolled in the current study, the cut point of 10% change in VO2peak was applied.
Factors explored
The following factors were considered as potential correlates with post-intervention change in VO2peak:
A) Exercise training parameters – time and intensity of exercise performed
Training parameters, in terms of amount (time) and intensity of exercise performed, were hypothesized to be related to degree of change in VO2peak. Exercise was prescribed 5 days per week, for durations ranging from 10 to 50 min per session. Exercise time performed each week was extracted from the exercise diaries. Conservatively, a time of “0” was assigned if an exercise diary was not submitted that week. Exercise intensity was quantified using the mean heart rate achieved during each exercise session, and calculated as a percentage of heart rate reserve (HRR), where HRR = [peak heart rate – resting heart rate] × [%] + resting heart rate.
B) Participant characteristics
Participant characteristics that reflected baseline impairment, functional mobility and fitness level were also considered as potential factors related to degree of change in VO2peak.
The following variables were hypothesized to be associated with change in VO2peak, as individuals with less neuromotor impairment and functional limitations would likely demonstrate greater improvements in fitness. The Chedoke-McMaster Stroke Assessment (13) Leg and Foot impairment inventory scores were combined as an index of lower limb impairment, balance ability was assessed using the Berg Balance Scale (19) and walking capacity was evaluated using the 6MWT (20).
Additionally, baseline VO2peak was also considered as a potential contributor to post-training change in aerobic capacity, where we anticipated that lower baseline fitness would be associated with greater improvement. Previous studies have reported similar relationships with walking measures, where individuals with lower baseline scores demonstrated greater gains relative to those started at higher functional levels (21, 22).
Analysis
Descriptive statistics were used to describe the groups. Multivariate regression analysis was performed to determine factors associated with post-training change in VO2peak. Variables were removed in an iterative process until the model was significant. To ensure assumptions of the multivariate regression were met, tolerance values and variance inflation factors were examined for multi-collinearity. Statistical Package for the Social Sciences (Version 17.0, Chicago IL) was used with a significance level of p < 0.05.
Results
For the 32 participants, mean percent change in VO2peak was 11.2% (standard deviation (SD) 18.8), but values ranged across participants from -32% to 56% improvement (Fig. 1). There were 17 participants in the Non-Response group who demonstrated a mean change in VO2peak of -2.8% (SD 9.9), and 15 in the response group who improved with a mean of 27% (SD 12.9). Participant characteristics for the non-response and response groups can be found in Table I. There were no differences in baseline characteristics between the two groups (all p > 0.20).
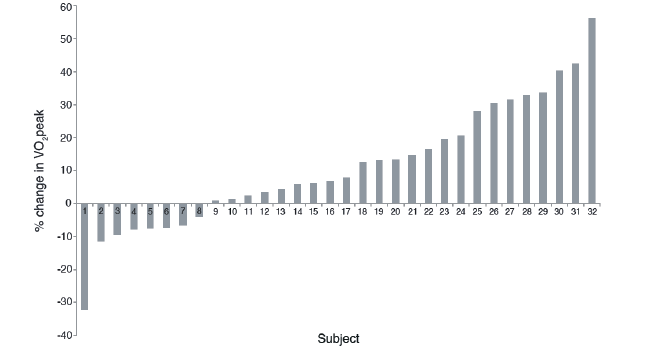
Fig. 1. Individual participant data for percent change in aerobic capacity (VO2peak).
|
Table I. Participant characteristics of each subgroup based on post-program change in aerobic capacity (VO2peak) (Response ≥ 10% change vs non-response <10% change) |
||
|
Response n = 15 |
Non-response n = 17 |
|
|
Age, years, mean (SD) |
63.7 (12.1) |
61.7 (13.3) |
|
Sex, men/women, n |
11/4 |
11/6 |
|
Time post-stroke, months, mean (SD) |
34.7 (32.1) |
23.8 (19.3) |
|
Gait aid used, none/cane/walker, n |
6/6/3 |
7/5/5 |
|
CMSA lower limb impairment, mean (SD) |
9.9 (2.5) |
9.1 (3.0) |
|
Berg Balance Scale, mean (SD) |
47.1 (11.5) |
45.6 (11.8) |
|
6MWT, m, mean (SD) |
306.2 (158.9) |
275.2 (126.5) |
|
Aerobic capacity, mean (SD) |
||
|
Baseline VO2peak, ml × kg–1 × min–1 |
13.8 (4.4) |
15.8 (5.2) |
|
Post-training VO2peak, ml × kg–1 × min–1 |
17.3 (5.2) |
15.3 (5.0) |
|
Change in VO2peak, ml × kg–1×min-1 |
3.5 (1.7) |
–0.6 (1.8) |
|
Change in VO2peak, % |
27.0 (12.9) |
–2.8 (9.9) |
|
Exercise parameters, mean (SD) |
||
|
Exercise time, min/week |
83.6 (65.2) |
76.3 (49.2) |
|
Exercise intensity, % HRR |
51.3 (21.2) |
50.4 (26.1) |
|
Classes attended, % |
85.0 (12.2) |
83.0 (12.7) |
|
Completed diaries submitted, % |
78.2 (21.4) |
74.4 (20.5) |
|
CMSA: Chedoke-McMaster Stroke Assessment; 6MWT: 6-minute walk test; HRR: heart rate reserve; SD: standard deviation. |
||
From the results of the maximal exercise tests, peak heart rate achieved was 91% (SD 23) of age-predicted maximum heart rate (calculated as 220 – age, and adjusted for participants (n = 18) taking anti-hypertensive medications as 70% × age-predicted maximal heart rate (23)), and peak respiratory exchange ratio (calculated as VCO2/VO2) was 1.2 (SD 0.16). Over the course of the program, there were no changes in medication use or dosages. Seventy-six percent of all exercise diaries were submitted (maximum 24 diaries).
Factors associated with post-program change in VO2peak
Potential correlates were first entered into the multivariate regression model (Model 1). Variables were then removed from subsequent iterations until the model was significant (Model 5). In this final model, baseline VO2peak and Berg Balance Scale score explained 20.5% of the variance of percent change in VO2peak (F(2,27) = 3.5, p = 0.045). Results from the regression analysis are presented in Table II.
|
Table II. Multivariate regression to determine correlates with percent change in aerobic capacity (VO2peak) |
|||||
|
Correlates |
R2 |
R2 change |
Unstandardized Β (SE) |
Standardized B |
p-value |
|
Model 1: F(6,23)=1.43 |
0.272 |
0.272 |
0.25 |
||
|
6MWT |
0.005 (0.04) |
0.04 |
0.89 |
||
|
Baseline VO2peak |
–2.18 (0.9) |
–0.56 |
0.02 |
||
|
Berg Balance Scale |
0.88 (0.5) |
0.48 |
0.07 |
||
|
CMSA lower limb impairment |
–1.86 (1.6) |
–0.28 |
0.28 |
||
|
Exercise intensity |
0.05 (0.1) |
0.07 |
0.71 |
||
|
Exercise time |
0.05 (0.1) |
0.16 |
0.45 |
||
|
Model 2: F(5,24)=1.79 |
0.271 |
–0.001 |
0.15 |
||
|
Baseline VO2peak |
–2.16 (0.8) |
–0.56 |
0.02 |
||
|
Berg Balance Scale |
0.91 (0.4) |
0.49 |
0.04 |
||
|
CMSA lower limb impairment |
–1.73 (1.4) |
–0.26 |
0.22 |
||
|
Exercise intensity |
0.05 (0.1) |
0.07 |
0.70 |
||
|
Exercise time |
0.05 (0.1) |
0.16 |
0.43 |
||
|
Model 3: F(4,25)=2.27 |
0.266 |
–0.005 |
0.09 |
||
|
Baseline VO2peak |
–2.14 (0.8) |
–0.55 |
0.01 |
||
|
Berg Balance Scale |
0.93 (0.4) |
0.50 |
0.03 |
||
|
CMSA lower limb impairment |
–1.77 (1.3) |
–0.27 |
0.20 |
||
|
Exercise time |
0.05 (0.1) |
0.16 |
0.43 |
||
|
Model 4: F(3,26)=2.85 |
0.248 |
–0.018 |
0.06 |
||
|
Baseline VO2peak |
–1.90 (0.7) |
–0.49 |
0.02 |
||
|
Berg Balance Scale |
0.95 (0.4) |
0.51 |
0.03 |
||
|
CMSA lower limb impairment |
–1.60 (1.3) |
–0.24 |
0.24 |
||
|
Model 5: F(2,27)=3.48 |
0.205 |
–0.043 |
0.045 |
||
|
Baseline VO2peak |
–1.82 (0.7) |
–0.47 |
0.02 |
||
|
Berg Balance Scale |
0.7 (0.4) |
0.38 |
0.06 |
||
|
6MWT: – 6-minute walk test; CMSA: Chedoke-McMaster Stroke Assessment; HRR: Heart rate reserve; SE: standard error. |
|||||
Discussion
This secondary analysis study is the first to identify and examine subgroups of post-stroke exercise responses in aerobic capacity. We previously reported that a 6-month adapted community-based exercise program was effective in improving overall average aerobic capacity for individuals with stroke (5) but upon closer inspection of the individual participant responses, considerable between-subject variability was noted. We demonstrated that baseline aerobic capacity and balance ability accounted for 20% of the variance of change in VO2peak.
This study is the first to characterize subgroups of participants with stroke across a range of training-related responses in VO2peak, from improvement to decline. Multivariate regression analysis revealed that baseline aerobic capacity was negatively correlated with post-training change in VO2peak, suggesting that individuals with the most compromised fitness levels may serve to gain the greatest benefit from an exercise intervention. Previous studies have reported a similar relationship with functional mobility interventions after stroke, where greater improvements were observed among participants with low or moderate baseline function in gait speed (21) and 6MWT distance (22) relative to those who started at higher functional levels. Langhammer and Lindmark dichotomized a sample of 75 participants with stroke and found that, following intensive exercise training, participants with lower baseline functional ability demonstrated greater improvement in motor function, balance, performance of activities of daily living and mobility relative to those with higher baseline function (24). No previous study has characterized responses in aerobic capacity following exercise interventions.
That individuals with more compromised fitness levels serve to make greater gains in VO2peak has important implications on the health of those with stroke. The inverse relationship between fitness and cardiovascular risk has been well established, whereby individuals in the lowest category of maximal aerobic capacity had higher rates of all-cause mortality and cardiovascular events (25). More importantly, the relationship may not necessarily be a linear one, as the largest reductions in cardiovascular risk have been demonstrated among individuals in the lowest percentiles of fitness levels. In a meta-analysis of studies examining the influence of physical fitness and heart disease risk factors, the greatest reduction in relative risk for coronary heart disease or cardiovascular disease was found between the lowest and second-lowest categories (26). While these meta-analyses were focused on healthy populations, it is not known whether similar risk reductions are present among individuals with stroke. Certainly, our findings are aligned with those of Langhammer & Lindmark (24), who demonstrated that individuals with stroke with lower baseline functional ability make greater functional gains. Together, these results suggest that different exercise models may be required for different cohorts of the stroke population for optimal benefit. VO2peak values we observed were comparable to previous reports confirming that the fitness after stroke is compromised relative to age-matched non-stroke controls (27, 28). Results from this study underscore the importance of implementing strategies to increase aerobic capacity in the stroke population, particularly amongst those who are at greatest risk for recurrent events.
While VO2peak is a physiological outcome, we hypothesized that neuromotor or functional factors may also contribute to participants’ ability to achieve prescribed exercise parameters to maximize improvement in aerobic capacity. Berg Balance Scale score, a functional measure of balance, was positively correlated with post-intervention change in VO2peak. The community exercise program did employ a variety of training modalities, but the majority of participants used walking (overground or treadmill) as their means for exercise training (5). In order to optimize aerobic benefit from walking, balance control is an important contributing factor in achieving adequate gait speed and duration of training.
There is growing awareness of the importance of non-exercise physical activity and its contribution to maintaining and increasing fitness levels (12). Physical activity levels are known to be low after stroke (29) and previous studies have reported that Berg Balance Scale scores were associated with home and community ambulatory activity among individuals with stroke (30, 31). It is possible that individuals with better balance were more physically active outside of formal exercise sessions (31), which may have also contributed to greater post-program improvements observed in VO2peak.
Interestingly, 6MWT, which reflects community walking activity in individuals with stroke (29, 31), was not associated with change in VO2peak. Its close association with Berg Balance scale score (r = 0.58, p < 0.0001) partially explains its low contribution to the overall model. The lack of association may also be explained by the differences in testing modality between these two tests: a functional walking test versus a graded exercise test where most were performed on cycle ergometer. Post-stroke neuromotor impairments, such as balance impairment and lower limb weakness and incoordination, may influence performance on a functional walk test more so than aerobic fitness, whereas performing graded exercise tests on cycle ergometer offers the advantages of body weight support and the feet being affixed to the pedals (32). Indeed, earlier studies that have examined the relationship between 6MWT distance and VO2peak among individuals with stroke have reported modest (32, 33) or non-significant correlations (34). Arguably, while these studies were cross-sectional analyses that examined relationships at a single time point, the results from the current are still aligned with these early findings with no association between 6MWT distance and changes in VO2peak over time.
We acknowledge subgroup comparisons were not performed, as we were underpowered to carry out such analyses. The sample size determined for the original trial (5) aimed to detect changes across time, not for subgroup analysis. A larger sample sufficiently powered for stratified subgroup analyses may be the focus for future work. Rather, results from this study highlight the heterogeneity within the stroke population given the wide range of program responses observed in the main trial. Of note, there were participants in the non-response subgroup who demonstrated a decline in aerobic capacity despite the intervention. In previous longitudinal observational studies of individuals with stroke, factors such as inactivity, cognitive function, fatigue, depression (35) and access to post-discharge rehabilitation (36) are related to functional deterioration in the first year following stroke. Ongoing work may seek to explain why there are differences in the direction of change, or explore other factors that may account for benefit or decline in a program’s effects. It is possible that these individuals were at risk for even greater decline in fitness levels had it not been for the exercise intervention. These results underscore the importance of establishing interventions that may be preventative to counteract such deterioration.
There are several limitations to this secondary analysis. Firstly, as previously mentioned, the sample size used was calculated for the original trial (5) to detect changes across time, not for subgroup analysis. A larger sample sufficiently powered for stratified subgroup analyses may be the focus for future work. Secondly, there are limitations related to the analysis of the exercise diary data. We employed a conservative estimate of exercise performed by assuming that “0” min of exercise was completed if an exercise diary was not submitted that week. This may have underestimated the actual amount completed. Post-hoc multivariate regression analysis was performed to evaluate the potential influence of program adherence on VO2peak change. The addition of class attendance and percentage of exercise diaries submitted did not provide additional explanation of the variance of the post-program change in VO2peak (R2 = 0.203, F(2,28) = 3.57, p = 0.042), but baseline VO2peak and class attendance remained in the final model. Finally, we acknowledge the potential contribution of other factors not examined in this study that may account for post-program change in aerobic capacity and provide additional explanation of the variance in the regression model. Since our study sample represented a relatively “young” cohort of individuals with stroke, future work may include older participants from a broader age range to examine the influence of age on VO2peak change. A larger sample would also allow for exploration of other contributing factors, such as gender-specific differences in exercise response, changes in muscle strength and endurance, presence of co-morbidities, or non-physical factors such as psychological or emotional status, social support, or level of cognitive function.
In summary, this study is the first to describe participants who responded with improvement in aerobic capacity following exercise using a community-based exercise intervention for stroke survivors. Baseline fitness level and functional balance accounted for 20% of the variance of post-training change in VO2peak. Looking beyond group means provides us with an opportunity to explore factors that may influence the degree to which gains are achieved between response subgroups. The results from this investigation are important first steps in identifying individuals who are more likely to respond to this intervention and may be used to inform future work in designing programs with optimal benefit for different subsets of the stroke population.
Acknowledgement
This study was supported by the Heart and Stroke Foundation of Ontario (Grant #SRA5977). DB is a Canada Research Chair, AT was supported by the Heart and Stroke Foundation of Canada, Canadian Institutes of Health Research, Canadian Stroke Network, Government of Ontario/Heart and Stroke Foundation of Ontario, Toronto Rehabilitation Institute, Physiotherapy Foundation of Canada and the University of Toronto, SM was supported by the University of Toronto. We acknowledge the support of the Heart and Stroke Foundation of Ontario, and the Toronto Rehabilitation Institute who receives funding under the Provincial Rehabilitation Research Program from the Ministry of Health and Long Term Care in Ontario.
References
