Sinikka H. Peurala, PhD, PT1, Auli H. Karttunen, MSc, PT2, Tuulikki Sjögren, PhD, PT2, Jaana Paltamaa3, PhD, PT and Ari Heinonen, PhD, PT2
From the 1Validia Rehabilitation, Helsinki, 2Department of Health Sciences, University of Jyväskylä and 3School of Health and Social Studies, JAMK University of Applied Sciences, Jyväskylä, Finland
OBJECTIVE: To examine the effect of randomized controlled trials of walking training on walking and self-care in patients with stroke.
DATA SOURCES: MEDLINE, CINAHL, Embase, PEDro, OTSeeker, Central, and manual search to the end of August 2012.
STUDY SELECTION: English, Finnish, Swedish, or German language walking training randomized controlled trials for patients over 18 years of age with stroke.
DATA SYNTHESIS: The meta-analyses included 38 randomized controlled trials from 44 reports. There was high evidence that in the subacute stage of stroke, specific walking training resulted in improved walking speed and distance compared with traditional walking training of the same intensity. In the chronic stage, walking training resulted in increased walking speed and walking distance compared with no/placebo treatment, and increased walking speed compared with overall physiotherapy. On average, 24 training sessions for 7 weeks were needed.
CONCLUSION: Walking training improves walking capacity and, to some extent, self-care in different stages of stroke, but the training frequency should be fairly high.
Key words: stroke; rehabilitation; recovery of function; walking; outcome measures; International Classification of Functioning; Disability and Health.
J Rehabil Med 2014; 46: 387–399
Correspondence address: Sinikka H. Peurala, Validia Rehabilitation Lahti, Launeenkatu 10, FI-15100 Lahti, Finland. E-mail: sinikka.peurala@validia.fi or sinikka.peurala@gmail.com
Accepted Jan 21, 2014; Epub ahead of print Apr 10, 2014
Introduction
Stroke is a leading cause of disability in many western countries. The ability to perform basic activities of daily living is initially reduced in 3 out of 4 patients with stroke (1). The most severely affected activities are the abilities to transfer, dress and walk. However, it has been shown that early intensive physiotherapy is related to better and faster recovery in the aforementioned activities in patients with stroke (2–5). Recovery of walking to regain independence in daily life is one of the main goals of stroke patients. It has been shown that to improve specific motor skills requires training in relevant tasks in patients with stroke (6). Traditional walking training includes walking with essential walking aids/orthosis combined with verbal and manual guidance. Additional rehabilitation tools during walking training consist of visual cues (7), concurrent cognitive tasks, i.e. dual tasks (8), musical feedback (9) and/or functional electrical stimulation (10). In addition, treadmill training with partial body weight support was introduced as a strategy for gait rehabilitation more than 10 years ago (11–13).
More recently, various electromechanical-assisted gait training devices have been developed. The Gait Trainer and the Driven Gait Orthosis were the first of these (14, 15). On the Gait Trainer, the patient is supported with a harness and the feet are placed on motor-driven footplates. On the Driven Gait Orthosis, the patient moves on a treadmill operated according to a pre-programmed physiological gait pattern and an exoskeleton-type robot. A Cochrane Review of electromechanical-assisted training for walking after stroke reported that the use of the Gait Trainer and Driven Gait Orthosis could reduce dependency in walking by 25% (16). One of the latest developments is the G-EO Walker, based on the end-effector principle. The patient is placed in a harness and stands on 2 foot-plates, the trajectories of which are freely programmable. This allows practice of simulated floor walking and climbing up and down stairs (17).
To date, evidence on the effects of walking training is either scarce or is mainly method-specific (13, 16). Moreover, it appears that the outcomes in terms of speed and functional walking ability obtained by different treatment modes have not been translated to yield comparable data (18). It is well known that after stroke the physical functioning recovery progress is rapid and partly spontaneous, but later the results achieved are slower and often becomes sustaining. Therefore it is reasonable to investigate the effect of walking training at different stages of stroke.
The purpose of this study was to investigate the evidence for the effect of walking training on walking and self-care in patients at different stages of stroke, i.e. in the acute, subacute and chronic stages of stroke. We linked the outcome measures of the International Classification of Functioning, Disability and Health (ICF) Mobility and Self-care domains according to the international guidelines and other literature (19–23). In the Mobility domain we used only the category of Walking. Evidence on the effect of walking training was gathered by meta-analyses, in which stroke patients were divided into 3 rehabilitation subgroups. In addition, evidence was gathered about the number and quality of randomized controlled trials (RCTs).
Material and methods
The search strategy was originally performed for physiotherapy interventions on stroke (Fig. 1) (24). In later updates, the key words in the search were “disease-specific”, and “exercise-specific”, i.e. walking, ambulation or gait. The full search strategies are available on request from the corresponding author, but the Ovid MEDLINE search strategy is given as an example in Appendix SI1.

A systematic literature search was conducted in the Ovid MEDLINE database, the Cumulative Index to Nursing & Allied Health Literature, Embase, the Physiotherapy Evidence Database, the Cochrane Central Register of Controlled Trials, and by manual search. Two information specialists performed the searches in these electronic databases in conjunction with the researchers.
Only RCTs for patients over 18 years of age with stroke published in English, Finnish, Swedish or German were included in the study. In addition, only RCTs in which walking training was specified either as minutes separately from other therapy or as achieved walking distance during the intervention were included. Exclusion criteria were non-randomized and non-controlled pre-experimental studies and protocols. Studies were collected up to August week 4, 2012. Bibliographies of walking review articles, narrative reviews and abstracts published in conference proceedings were also evaluated for relevant publications. In addition, citation tracking of all walking RCT article references was conducted. Inclusion of articles was based on agreement between 2 independent reviewers (SP & TS/JP/T-KT/HS/AK).
Methodological quality or risk of bias was assessed based on individual trial characteristics using the criteria of van Tulder et al. (25). The items concern internal validity related to selection bias, performance bias, attrition bias and detection bias. All 11 items were rated as “yes”, “no” or “don’t know”. If item A was rated “don’t know”, a request was sent to the authors to clarify whether the method of randomization was adequate. If 2 requests were not answered, the study was accepted, but the method remained rated as “don’t know”. RCTs were considered as high-level, acceptable or poor depending on the yes-rated items and number of subjects (see Appendix I) (26). Methodological quality and content analysis were evaluated by 2 blinded and independent assessors (SP & T-KT/HS/AK). Disagreements were resolved by consensus between the reviewers; where this was not possible the issue was decided by a third reviewer (TS, JP or AH). Inter-rater reliability for the methodological quality assessment was computed by Kappa statistics, in which Adequate is 0.40–0.74 and Excellent ≥ 0.75 (27).
The meta-analyses were performed in 3 rehabilitation subgroups as follows: (i) walking training vs no or placebo treatment, (ii) walking training vs overall physiotherapy, and (iii) specific walking training methods vs walking on the floor with walking aids and with the help of verbal and/or manual guidance of a therapist if needed, but not with any additional methods (hereafter termed traditional walking training). Specific walking training methods include tools that are not used in traditional walking training, e.g. auditory feedback during gait, and treadmill and electrical-assisted walking training. The meta-analyses in each rehabilitation subgroup were divided into 3 stroke groups: acute (less than 1 month after stroke), subacute (1–6 months after stroke) and chronic (more than 6 months after stroke).
In the meta-analyses, Cochrane Collaboration’s Review Manager 5.2 program was used to calculate pooled effect estimates for combinations of single study effects. The inverse-variance random-effects method was used. Random-effect meta-analysis assumes that effects that are estimated are not identical in different studies but follow a distribution. The centre of this distribution describes the mean of the effects and its width the degree of heterogeneity. For continuous outcomes 2 summary statistics were available: the mean difference and the standardized mean difference, i.e. the effect size. The mean difference was used when studies had outcomes in the same scale (in all the walking parameters), whereas when outcomes in different scales were reported, the standardized mean difference was used (in Self-care).
Change values for outcomes and their standard deviations (SDs) were used, if they were available. If not, then the final values were used. If neither were reported, a request for this information was sent to the authors. If 2 requests were not answered the RCT was not entered into the meta-analyses. In studies in which multiple comparisons had been done, for example 2 or more intervention groups vs a control group, the number of controls in the group was divided amongst the comparisons. The overall effect was tested with the z-test, where the null hypothesis is that there is no difference between an intervention group and a control group. Poor-quality studies were not included in the meta-analyses.
Depending on the quality and number of RCTs, the meta-analysis evidence was rated in 4 categories: high, moderate, low or no evidence (see Appendix II) (28).
Results
Altogether 44 papers reporting 38 RCTs (6 papers reported the same RCT twice) published during the period 1992–2012 fulfilled the inclusion criteria (Fig. 1 (8, 9, 73–114)). After contacting the authors, the randomization method used remained unclear in 3 studies (Table I). The methodological quality of the included RCTs was generally good: mean 6.0 (SD 1.6) yes scores, range 3–9 (Table I). In our quality assessment, the Kappa for inter-rater reliability was 0.80 (almost perfect agreement, p < 0.001).
Seven RCTs in 9 articles reported walking training compared with no or placebo treatment (8, 73–80). Eleven RCTs compared walking training with overall physiotherapy (81–91). Twenty-one RCTs in 24 articles were analysed in a subgroup of specific vs traditional walking training (9, 92–114).
|
Table I. Quality analysis of 44 randomized controlled trials (RCT) reports concerning walking training in stroke |
||||||||||||||
|
Study |
Randomization method acceptablea |
Concealment of treatment allocationa |
Group similaritya |
Blinding of patient |
Blinding of care provider |
Blinding of outcome assessor |
Determination of co-interventions |
Compliance |
Drop-outs and reasonsa |
Timing of outcomes similar |
Intention-to-treat |
Number of yes and quality levelb |
||
|
Walking training vs No/placebo treatment |
||||||||||||||
|
Ada et al., 2003 (8) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
Yes |
Don’t know |
7 High |
||
|
Chu et al., 2004 (73) |
Don’t know |
No |
Yes |
Don’t know |
No |
Yes |
Don’t know |
Yes |
Yes |
Don’t know |
Yes |
5 Poor |
||
|
Kang et al., 2012 (74) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Don’t know |
Yes |
Yes |
Don’t know |
6 High |
||
|
Luft et al., 2008 (75) |
Yes |
Don’t know |
Yes |
No |
No |
Yes |
Don’t know |
Don’t know |
No |
Yes |
No |
4 Acceptable |
||
|
Macko et al., 2005 (76) |
Yes |
No |
Yes |
No |
No |
No |
Don’t know |
Yes |
Yes |
Don’t know |
Don’t know |
4 Acceptable |
||
|
Moore et al., 2010 (77) |
Yes |
No |
Yes |
No |
No |
Don’t know |
Yes |
Don’t know |
No |
Yes |
Don’t know |
4 Acceptable |
||
|
Salbach et al., 2005 (78) |
Yes |
Yes |
Yes |
No |
Don’t know |
Yes |
Don’t know |
Yes |
Yes |
Yes |
No |
7 High |
||
|
Salbach et al., 2004 (79) |
Yes |
Yes |
Yes |
No |
Don’t know |
Yes |
Don’t know |
Yes |
Yes |
Yes |
No |
7 High |
||
|
Yang et al., 2007 (80) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
Yes |
Yes |
8 High |
||
|
Walking training vs Overall physiotherapy |
||||||||||||||
|
Dias et al., 2007 (81) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Don’t know |
Don’t know |
Yes |
Don’t know |
5 Acceptable |
||
|
Duncan et al., 2011 (82) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Yes |
Yes |
No |
Yes |
Yes |
8 Acceptable |
||
|
Fisher et al., 2011 (83) |
Yes |
Don’t know |
Yes |
No |
No |
Don’t know |
Don’t know |
Yes |
Yes |
Yes |
Yes |
6 Acceptable |
||
|
Globas et al., 2012 (84) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
9 High |
||
|
Høyer et al., 2012 (85) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
Yes |
Yes |
8 High |
||
|
Jonsdottir et al., 2010 (86) |
Yes |
Don’t know |
Yes |
Don’t know |
No |
Yes |
Don’t know |
Don’t know |
Yes |
Yes |
Yes |
6 Acceptable |
||
|
Kuys et al., 2011 (87) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
Yes |
Yes |
8 High |
||
|
Mayr et al., 2007 (88) |
Yes |
Don’t know |
Yes |
No |
No |
Yes |
Don’t know |
Don’t know |
Yes |
Yes |
Yes |
6 Acceptable |
||

|
Table I. Contd. |
||||||||||||||
|
Study |
Randomization method acceptablea |
Concealment of treatment allocationa |
Group similaritya |
Blinding of patient |
Blinding of care provider |
Blinding of outcome assessor |
Determination of co-interventions |
Compliance |
Drop-outs and reasonsa |
Timing of outcomes similar |
Intention-to-treat |
Number of yes and quality level |
||
|
Park et al., 2011 (89) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
No |
Yes |
Yes |
No |
6 Acceptable |
||
|
Yang et al., 2005 (90) |
Yes |
Yes |
Yes |
Don’t know |
Don’t know |
No |
Yes |
Don’t know |
Yes |
Yes |
Yes |
7 High |
||
|
Yen et al., 2008 (91) |
Yes |
Yes |
Yes |
No |
No |
No |
Don’t know |
Yes |
Yes |
Yes |
Yes |
7 High |
||
|
Specific vs Traditional walking training |
||||||||||||||
|
Ada et al., 2010 (92) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
No |
Yes |
7 High |
||
|
da Cunha-Filho et al., 2001 (93) |
Yes |
Don’t know |
Yes |
No |
Don’t know |
Don’t know |
Don’t know |
Yes |
No |
No |
No |
3 Poor |
||
|
da Cunha et al., 2002 (94) |
Yes |
Don’t know |
Yes |
Don’t know |
Don’t know |
Don’t know |
Yes |
Yes |
Yes |
Yes |
No |
6 Acceptable |
||
|
Dean et al., 2010 (95) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
Yes |
Yes |
8 High |
||
|
Eich et al., 2004 (96) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
Yes |
Yes |
8 High |
||
|
Hidler et al., 2009 (97) |
Yes |
Don’t know |
Yes |
Don’t know |
Don’t know |
Don’t know |
Yes |
Don’t know |
Don’t know |
Yes |
No |
4 Acceptable |
||
|
Husemann et al., 2007 (98) |
Yes |
Yes |
Yes |
Don’t know |
Don’t know |
Yes |
Yes |
Don’t know |
No |
Yes |
Don’t know |
6 Acceptable |
||
|
Kosak & Reding, 2000 (99) |
Don’t know |
Don’t know |
Yes |
No |
No |
Don’t know |
Yes |
Don’t know |
Yes |
Yes |
Don’t know |
4 Poor |
||
|
Langhammer & Stanghelle, 2010 (100) |
Yes |
Yes |
Yes |
No |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
9 High |
||
|
Morone et al., 2011 (101) & 2012 (102) |
Yes |
Don’t know |
No |
No |
No |
Yes |
Don’t know |
Yes |
Yes |
Don’t know |
Yes |
5 Acceptable |
||
|
Morris et al., 1992 (103) |
Yes |
Yes |
Yes |
Don’t know |
Don’t know |
Yes |
Yes |
Don’t know |
No |
Yes |
Don’t know |
6 Acceptable |
||
|
Ng et al., 2008 (104) |
Yes |
Don’t know |
No |
No |
No |
No |
Don’t know |
Yes |
Yes |
Yes |
Yes |
5 Acceptable |
||
|
Nilsson et al., 2001 (105) |
Yes |
Don’t know |
Yes |
No |
No |
Yes |
Yes |
Don’t know |
Yes |
Yes |
No |
6 Acceptable |
||
|
Olawale et al., 2011 (106) |
Yes |
No |
Yes |
No |
No |
Don’t know |
Don’t know |
Don’t know |
Yes |
Yes |
Don’t know |
4 Acceptable |
||
|
Peurala et al., 2005 (107) |
Yes |
Yes |
Yes |
No |
No |
Don’t know |
Don’t know |
Yes |
Yes |
Yes |
No |
6 High |
||
|
Peurala et al., 2009 (108) |
Yes |
Yes |
Yes |
No |
No |
No |
Don’t know |
Yes |
Yes |
Yes |
No |
6 High |
||
|
Pohl et al., 2002 (109) |
Yes |
Don’t know |
Yes |
No |
No |
Yes |
Don’t know |
Don’t know |
Yes |
Yes |
No |
5 Acceptable |
||
|
Pohl et al., 2007 (110) |
Yes |
Yes |
Yes |
No |
No |
No |
Don’t know |
Yes |
Yes |
Yes |
Yes |
7 High |
||
|
Schaeur & Mauritz, 2003 (9) |
Yes |
No |
Yes |
No |
No |
Don’t know |
Don’t know |
Don’t know |
Don’t know |
Yes |
Don’t know |
3 Poor |
||
|
Schwartz et al., 2009 (111) |
Don’t know |
Don’t know |
Yes |
No |
No |
No |
Don’t know |
Don’t know |
Yes |
Yes |
Yes |
4 Poor |
||
|
Tong et al., 2006 (112) |
Yes |
Don’t know |
No |
No |
No |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
6 Acceptable |
||
|
Thaut et al., 1997 (113) |
Yes |
Yes |
Yes |
Don’t know |
Don’t know |
Yes |
Don’t know |
Don’t know |
Don’t know |
Yes |
Don’t know |
5 Acceptable |
||
|
Thaut et al., 2007 (114) |
Yes |
Yes |
Yes |
Yes |
No |
Yes |
Don’t know |
Don’t know |
Don’t know |
Yes |
Don’t know |
6 Acceptable |
||
|
The methodological quality of the RCTs was rated using criteria and decision rules modified from van Tulder et al. (2003) (25). aThese columns indicate the rating items needed for high-level RCTs with the total number of “yes” answers ≥ 6 and the number of patients ≥ 30 (25). bThe RCT was rated acceptable if the number of “yes” answers was ≥ 4 containing A, and poor if the number of “yes” answers was 0–3 or ≥ 4, but A was “don’t know” or if the number of patients in the group was ≤ 5. Poor-quality studies were not accepted for meta-analysis. |
||||||||||||||
Only outcome measures linked to the ICF Walking category, such as walking speed, walking distance, walking independence, and the Self-care domain were used. Table SI1 shows the precise outcomes. Walking speed (m/s) was most often measured with the 10-m walk test (115). Walking distance was measured with the 6-min walk test (116), and walking independence was evaluated with the Functional Ambulatory Category (115). Outcome measures linked to Self-care were the Functional Independence Measure (117) and Barthel Index (118). In the Functional Ambulatory Category, Functional Independence Measure, and Barthel Index, a higher score denotes better functioning.
Walking training vs no/placebo treatment
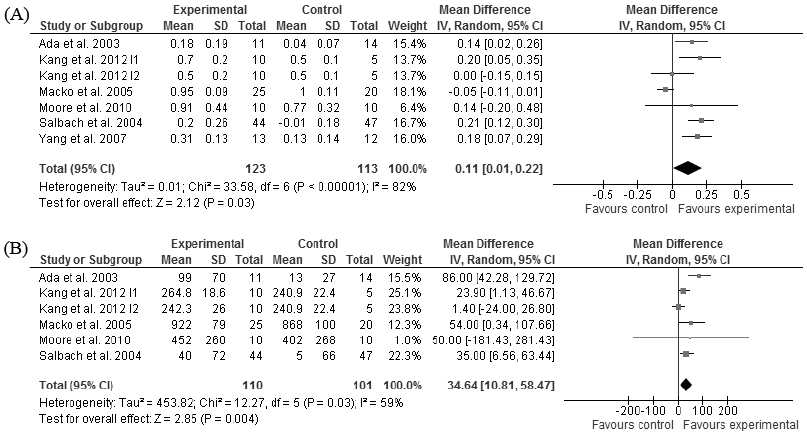
All the patients analysed in this rehabilitation subgroup were in the chronic stage of stroke. High evidence showed that walking training increased walking speed compared with no/placebo treatment (p = 0.03, Fig. 2A) (8, 74, 76–77, 79–80). Data from one study (acceptable quality) were not available for the meta-analyses (75) and the quality of another study was poor (73) (Tables I and SI1). High evidence also showed that walking training increased walking distance compared with no/placebo treatment (p = 0.004, Fig. 2B) (8, 74, 76–77, 79). These benefits in terms of walking speed and distance were achieved with walking training from 12 to 18 sessions during 4–6 weeks. Walking training time varied across studies from 30 to 60 min per session.
Walking training vs overall physiotherapy
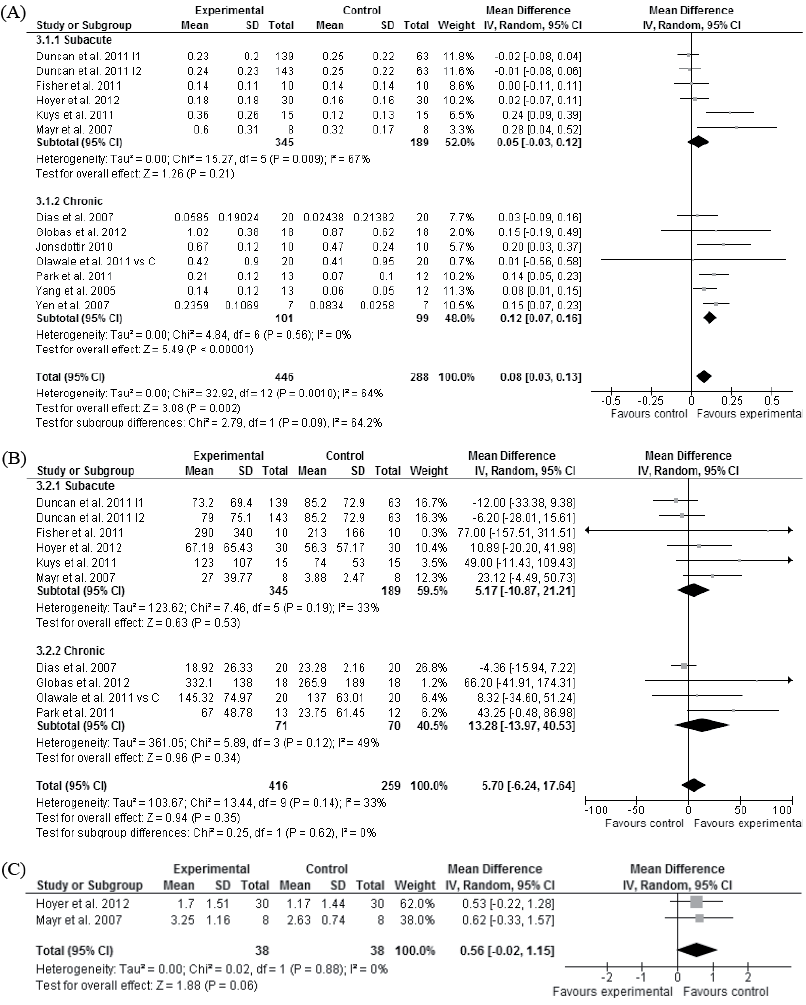
Moderate evidence showed that walking training did not improve walking speed more than overall physiotherapy in the subacute stage of stroke (p = 0.21, Fig. 3A) (82–83, 85, 87, 88). Whereas high evidence showed that walking training improved walking speed compared with overall physiotherapy in the chronic stage of stroke (p < 0.001, Fig. 3A) (Tables I and SI1) (81, 84, 86, 89–91, 106). The improvement in walking speed in the chronic stage of stroke was achieved with walking training from 12 to 36 sessions over 3–12 weeks. Walking training time in these studies varied from 20 to 60 min per session.
Moderate evidence showed that walking training did not improve walking distance in the subacute (82–83, 85, 87, 88) or chronic (81, 84, 89, 106) stages of stroke (p = 0.53 and p = 0.34, Fig. 3B), but there was a trend of improved walking independence compared with overall physiotherapy in the subacute stage of stroke (p = 0.06, Fig. 3C) (85, 86).
Specific vs traditional walking training
Moderate evidence in the acute (94, 104–105, 108, 113–114) and high evidence in the subacute (96, 98, 103, 109–110) stage of stroke showed that specific walking training improved walking speed more than traditional walking training (p < 0.001 and p = 0.03, Fig. 4A). High evidence showed that specific and traditional walking training are comparable in gaining walking speed in the chronic stage of stroke (p = 0.65, Fig. 4A) (97, 100, 106–107). The improved walking speed in the subacute stage of stroke was achieved with walking training from 20 to 30 sessions over 4–6 weeks. Walking training time in these studies varied from 20 to 60 min per session. Five studies were not included in the meta-analysis, because their quality was rated as poor (9, 99, 111), data were not available for the meta-analyses (99) or the data (93) were gathered from and analysed for the same group of subjects as that in the analysis (94) (Tables I and SI1).
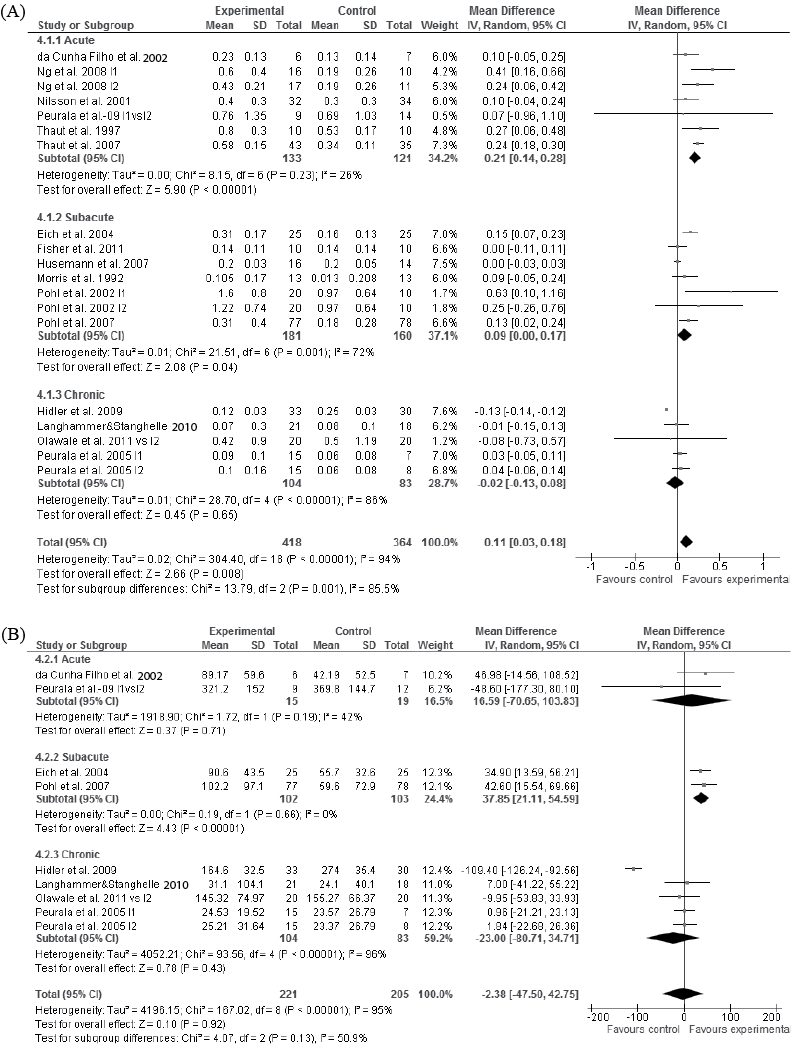
Low evidence showed that specific walking training did not improve walking distance more compared with traditional walking training in the acute (94, 108) stage of stroke (p = 0.71, Fig. 4B). High evidence showed that specific walking training increased walking distance more (p < 0.001), in the subacute (96, 110), but not in the chronic (97, 100, 106–107) stage of stroke, compared with traditional walking training (p = 0.43, Fig. 4B) (77, 88). The improved walking distance in the subacute stage of stroke was achieved with walking training from 20 sessions over 4 weeks or 30 sessions over 6 weeks. In both studies walking training time was 20 min per session. Two poor-quality studies were not included in the meta-analysis (Tables I and SI1) (93, 99). All the included studies used the 6-min walk test as an outcome measure.
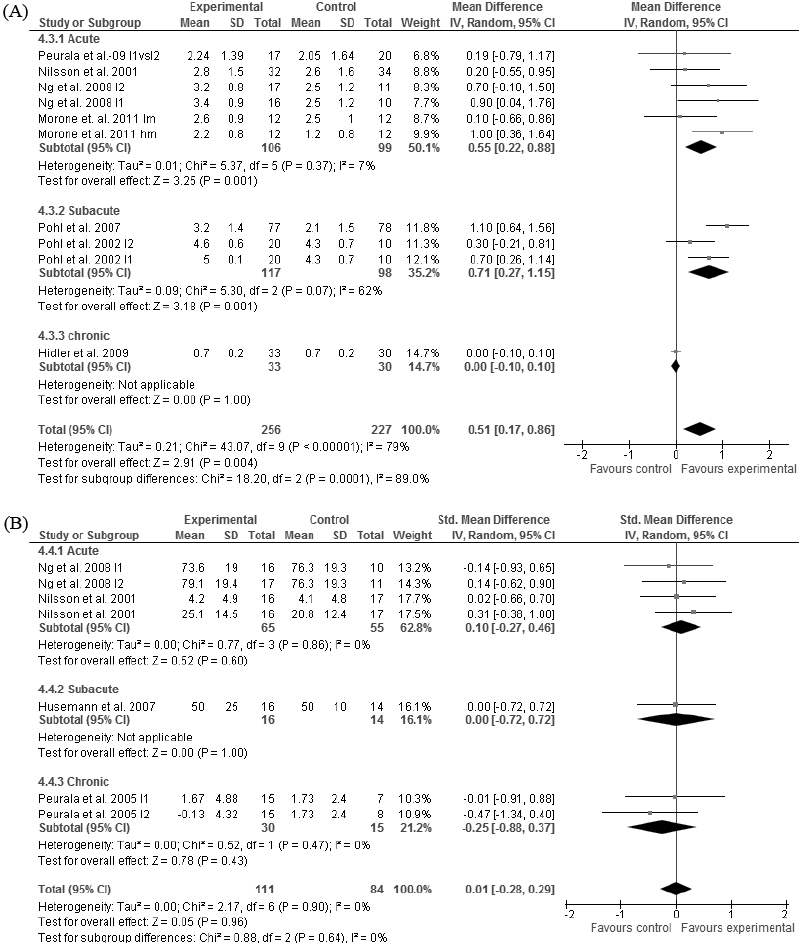
Moderate evidence showed that specific walking training improved walking independence more than did traditional walking training in the acute (p = 0.001, (101, 104, 105, 108) and subacute (109, 110) stages of stroke (p = 0.001, Fig. 5A). A single study with low evidence indicated that specific walking training did not improve walking independence compared with traditional walking in the chronic stage of stroke (p = 1.0, Fig. 5A) (97). In 6 studies data were not available for the meta-analyses (96, 98), the quality was rated as poor (99, 111) or the data (93) were gathered from and analysed for the same group of subjects as in the analysis (94) (Tables I and SI1).
Moderate evidence indicated that specific and traditional walking training are comparable in terms of gains in self-care in the acute stage of stroke (p = 0.60, Fig. 5B) (104, 105). Single studies indicated with low evidence that specific walking training did not improve self-care compared with traditional walking in either the subacute (p = 1.00) (98) or chronic stages of stroke (p = 0.43, Fig. 5B) (107). In one study data for the meta-analyses were not available (13), and in another study the outcome was not measured at the end of the intervention (101).
Discussion
Our final analysis comprised 44 publications including 38 RCTs on various aspects of walking training post-stroke. Only RCTs in which walking training was specified either as minutes apart from other therapy or as achieved walking distance during the intervention were included in the analyses. The level of evidence was determined on the basis of both the methodological quality and the results of the meta-analyses. The meta-analyses were run with walking and self-care outcome measures where available. High evidence is not supposedly to change its magnitude and/or its direction accordingly to new studies.
We found high evidence that, in the chronic stage of stroke, walking training improved walking compared with no or placebo treatment or overall physiotherapy, but traditional and specific walking training methods were comparable (Table II). This result highlights the fact that, even in the chronic stage of stroke, significant changes in walking ability can be achieved. Furthermore, in line with previous studies (121), this evidence supports the notion that exercise therapy should be task-specific, suggesting that re-learning or improving walking after stroke can be done best with walking training.
|
Table II. Level of evidence of walking training in the acute, subacute and chronic stage of stroke |
|||
|
Meta-analyses/ stage of stroke |
Walking training vs no/placebo treatment |
Walking training vs overall physiotherapy |
Specific vs traditional walking training |
|
Acute |
No evidence |
No evidence |
Moderate for walking speed – Specific walking training better Low for walking distance – Comparable Moderate for walking independence – Specific walking training better Moderate for self-care – Comparable |
|
Subacute |
No evidence |
Moderate for walking speed and distance – Comparable Moderate for walking independence – Walking training trend to be better |
High for walking speed and distance – Specific walking training better Moderate for walking independence – Specific walking training better Low for self-care – Comparable |
|
Chronic |
High for walking speed and distance – Walking training better |
High for walking speed – Comparable Moderate for walking distance – Comparable |
High for walking speed and distance – Comparable Low for walking distance – Comparable Low for self-care – Comparable |
In addition, high evidence was found that in the subacute stage of stroke, additional walking training tools such as musical feedback or body-weight-supported treadmill training improved walking better compared with same intensity traditional walking training (Table II). In the acute stage of stroke this evidence was moderate (Table II). Thus, we suggested that additional walking training tools should be used wherever possible in the acute and subacute stages of stroke.
Our analysis showed that walking speed is 0.21 m/s better in the acute and 0.09 m/s better in the subacute stage of stroke in specific walking training compared with traditional walking training, whereas in the chronic stage of stroke no such difference was observed between the training modalities. Fulk & Echternach (120) studied the minimal detectable change of gait speed in subjects undergoing rehabilitation after stroke. They found that the minimal detectable change at the 90% confidence level was 0.07 m/s with subjects who required physical assistance to walk, 0.18 m/s with subjects who required an assistive device to walk, and 0.36 m/s with subjects who were able to walk without physical assistance. The minimal detectable overall change was 0.30 m/s with patients in the acute and subacute stages of stroke (120). In our study, it can be speculated that within each stroke group minimal detectable walking speed was achieved.
Overall, our analyses showed that walking training from 12 to 36 sessions (mean 24 sessions) for 4–12 weeks (mean 7 weeks) was needed for effective improvements in walking speed and distance in patients with stroke. In general, walking training time in analysed studies varied from 20 to 60 min per session, but most commonly it was 30 or 45 min (mean 37 min). However, the level of evidence seemed to increase with longer training time (i.e. from 40 to 60 min per session). This suggests that the effective walking training frequency should be fairly high, on average 3–5 times per week, in sessions lasting from 20 to 60 min, in order to improve walking performance.
In a previous review, Dickstein (18) suggested that selection of the most appropriate method for walking training may be guided by a pragmatic approach. Although we agree with Dickstein’s suggestion that walking training itself is the most important issue in walking rehabilitation, our analyses of specific walking training (as we defined it) produced significantly better results compared with traditional walking training in the acute and subacute stages of stroke. Dickstein only had speed as an outcome and she used different subgroups in her analyses that made the comparison with our analysis somewhat different. In her review, she included other than RCTs in the analyses and did not take into account the level of the evidence of the meta-analysis in the conclusions.
This study had some limitations. First, we restricted our search to English, Finnish, Swedish, or German language publication, which may have resulted in publication bias. Secondly, in the present meta-analyses we focused only on the ICF Mobility domain, specifically walking speed, walking distance, walking independence, and ICF Self-care domain. Walking improvement is one of the main rehabilitation goals of patients with stroke, and the most common training in physiotherapy concerns walking (99, 122). In addition, in these patients the aspects of functioning that are most severely affected are transferring, dressing and walking (1). Thirdly, the analysed studies also included outcome measures of the other ICF components of body functions (e.g. spatio-temporal gait characteristics) and activity and participation (e.g. Berg Balance Scale), but we did not run a meta-analysis on these because they were very infrequent and were distributed across different subgroups. Fourthly, we did not use standard mean differences, which may have given more power to the statistics, but are difficult for a clinician to interpret; thus, we ran analyses as mean differences in order to provide clinicians with more practical results. Fifthly, the meta-analysis and results did not consist of follow-ups and therefore did not provide evidence about long-term effects. Sixthly, we excluded 12 RCTs that studied only different aspects of specific walking training (40, 44–50, 72, 104, 112, 107). Those single studies of different aspects give a more detailed picture of specific methods, such as speed-dependent treadmill training vs limited progressive treadmill training (45, 72).
These results indicate that the key issues in walking training are frequency and duration, i.e. number of sessions, training weeks and training minutes. In a systematic review and meta-analysis, Stoller et al. (123) studied training parameters in more detail, such as intensity of maximum heart rate reserve or resistance progression. They showed that patients with stroke may benefit from cardiovascular exercise during sub-acute stages to improve peak oxygen uptake and walking distance. Cardiovascular exercise was mainly performed by leg cycle ergometry, treadmill or stepper.
In conclusion, walking training produced training specific results in patients with stroke. Walking training, especially specific walking training, proved to be effective in improving walking speed, distance, and ability in different stages of stroke, thus improving mobility and, to some extent, self-care.
Clinical messages
• Walking training frequency should be fairly high; on average 3–5 times per week, in sessions lasting from 20 to 60 min, in order to be effective in improving walking performance.
• Walking training produces training specific results, i.e. improves walking speed, distance, and ability, that is, mobility and to some extent self-care.
ACKNOWLEDGEMENTS
The authors would like to thank the information specialists Anitta Pälvimäki at the Jyväskylä University Library and Irma Helldan at the Social Insurance Institute of Finland, physiotherapists and master students Tiina-Kaisa Tuupainen, Hanna Sahinoja at the Department of Health Sciences, University of Jyväskylä, for her help during the data collection, and Atte Hänninen, MSc, at the Department of Mathematical Information Technology, University of Jyväskylä, for statistical assistance.
This work was supported by the Social Insurance Institution of Finland (grants no. 31863) and Academy of Finland (grant no. 114291).

1http://www.medicaljournals.se/jrm/content/?doi=10.2340/16501977-1805
References
Appendix I. Criteria for evaluating methodological quality or risk for bias of randomized controlled trials (RCT) for walking training in stroke (28) (from ref 124, published with permission) | |||
Methodological quality of RCT (26) | Yes scores of van Tulder evaluation (maximum 11) | Demands of special criteria in van Tulder evaluation (24) | Other criteria |
High | ≥ 6 | Method of randomization adequate (A) Concealed allocation (B) Groups similar at baseline regarding the most important prognostic indicators (C) Drop-out rate described and acceptable (I) | ≥ 30 subjects in a study |
Acceptable | ≥ 4 | Method of randomization adequate (A) | |
Poor | ≥ 4 or 0–3 or | Method of randomization NOT adequate (A) N/A N/A | N/A N/A Number of subjects ≤ 5 in intervention or control group |
The methodological quality of the RCTs was rated using criteria and decision rules modified from van Tulder et al. (25). These include 11 items presented in Table I. All items were rated as “yes”, “no” or “don’t know”. N/A: not applicable. | |||
Appendix II. Criteria for determining level of evidence in meta-analyses of randomized controlled trials (RCT) for walking training in stroke (from ref 124, published with permission) | |
Evidence | Criteria |
High | At least 2 high-quality RCTs with parallel results |
Moderate | High-quality RCT or several high-quality RCTs with some contradictions in results or several acceptable-quality RCTs with parallel results |
Low | High-quality RCTs with notable contradictions of results or at least 1 acceptable RCT |
No evidence | Poor-quality RCT or RCTs or no RCTs |
