INTERVAL TRAINING-INDUCED ALLEVIATION OF RIGIDITY AND HYPERTONIA IN PATIENTS with PARKINSON’S DISEASE IS ACCOMPANIED BY INCREASED BASAL SERUM BRAIN-DERIVED NEUROTROPHIC FACTOR
A repeated-measures, case series pilot study
Jarosław Marusiak, PhD1, Ewa Żeligowska, MSc1, Joanna Mencel, MSc1, Katarzyna Kisiel-Sajewicz, PhD1, Joanna Majerczak, MD, PhD2, Jerzy A. Zoladz, DSc, PhD2, Artur Jaskólski, PhD1 and Anna Jaskólska, PhD1
From the 1Department of Kinesiology, Faculty of Physiotherapy, University School of Physical Education, Wroclaw and 2Department of Muscle Physiology, Chair of Physiology and Biochemistry, University School of Physical Education, Krakow, Poland
OBJECTIVE: To examine the effects of cycloergometric interval training on parkinsonian rigidity, relaxed biceps brachii muscle tone in affected upper extremities, and serum level of brain-derived neurotrophic factor.
DESIGN: Case series, repeated-measures design, pilot study.
SUBJECTS/PATIENTS: Eleven patients with mild-to-moderate Parkinson’s disease (Hoehn & Yahr scale 2.3 ± 0.72), recruited from a neurological clinic, underwent cycle training and were tested along with non-trained, healthy control subjects (n = 11) in a motor control laboratory.
METHODS: Patients underwent 8 weeks of interval training (3 × 1-h sessions weekly, consisting of a 10-min warm-up, 40 min of interval exercise, and 10-min cool-down) on a stationary cycloergometer. Parkinsonian rigidity (Unified Parkinson’s Disease-Rating-Scale) in the upper extremity, resting biceps brachii muscle tone (myometric stiffness and frequency), and brain-derived neurotrophic factor level were measured 1–3 days before interval training cycle started and 6–10 days after the last training session.
RESULTS: Training resulted in a decrease in rigidity (p = 0.048) and biceps brachii myometric muscle stiffness (p = 0.030) and frequency (p = 0.006), and an increase in the level of brain-derived neurotrophic factor (p = 0.035) relative to pre-training values. The increase in brain-derived neurotrophic factor level correlated with improvements in parkinsonian rigidity (p = 0.025), biceps brachii myometric stiffness (p = 0.001) and frequency (p = 0.002).
CONCLUSION: Training-induced alleviation of parkinsonian rigidity and muscle tone decrease may be associated with neuroplastic changes caused by a training-induced increase in the level of brain-derived neurotrophic factor.
Key words: Parkinson’s disease; rigidity; exercise; neuroplasticity; brain-derived neurotrophic factor.
J Rehabil Med 2015; 47: 00–00
Correspondence address: Jarosław Marusiak, Department of Kinesiology, Faculty of Physiotherapy, University School of Physical Education, Al. I. J. Paderewskiego 35, Building P4, 51-612 Wroclaw, Poland. E-mail: jarekmarusiak@gmail.com
Accepted Oct 22, 2014; Epub ahead of print Dec 9, 2014
INTRODUCTION
Previous studies indicate that physical activity improves motor functioning and clinical symptoms of Parkinson’s disease (PD) (1–6), possibly through positive neuroplastic changes in the central nervous system (4). An 8-week training regimen on a tandem cycloergometer at a high rate alleviated the main symptoms of PD and improved dexterity of the upper extremities in patients with PD (1), leading to speculation that training may induce neuroplastic changes in the basal ganglia by increasing the secretion of brain-derived neurotrophic factor (BDNF) and/or glial cell line-derived neurotrophic factor (4). It was reported recently that physical training increases the serum level of BDNF (5, 6) and simultaneously attenuates inflammation and improves the health status of patients with PD (5). Another study showed that high-intensity treadmill training improves corticomotor excitability in patients with PD (2), possibly normalizing pathologically increased spinal cord reflexive activity (7, 8). Of the various types of training undergone by PD patients (tandem cycloergometer (1) and treadmill (2), the common factor that can be linked to the improvement in symptoms is the high intensity of the exercise, consistent with studies on animal models of PD (4). However, high-intensity treadmill training at home has challenges, and tandem cycling requires a trainer/partner, making these forms of excercise less-than-ideal for most patients.
Thus, assuming that patients with PD can be instructed and motivated to pedal on a single cycloergometer at high speeds, we administered a high-intensity training protocol (1) structured as interval training (IT), which is more effective and enjoyable (9) than continuous training. To our knowledge no data has been reported so far showing the effect of intensive cycloergometer IT on resting muscle state in relation to changes in serum BDNF in patients with PD.
To investigate IT-related changes in the upper extremity muscles, clinical parkinsonian rigidity and resting muscle tone were evaluated using myometry. As reported previously, myometry is a sensitive method for detecting differences in resting muscle tone between patients with PD and healthy individuals (10), and medication-induced changes in the resting muscle tone of patients with PD (11). It was predicted that IT would alleviate rigidity and decrease resting muscle tone (myometric stiffness and frequency), and would be accompanied by an increase in serum BDNF level.
The aim of this pilot study was to estimate parkinsonian rigidity, myometric stiffness and frequency, and serum BDNF level in patients with PD before and after 8 weeks of cyclorgometric IT. The results have practical implications for understanding the mechanisms of physical training-induced amelioration of PD symptoms, and will form the basis for a prospective randomized control trial study.
METHODS
Subjects
Eleven patients with mild-to-moderate PD (12) (5 women and 6 men; mean age 71 years (standard deviation (SD) 10), mean body mass 70 kg (SD 7), mean height 163 cm (SD 9), mean illness duration 8 years (SD 4)) (Table I; see also description of the studied patients elsewhere (5)) were recruited from a neurological clinic; 11 healthy control subjects with no neurological or neuromuscular disorders (9 women and 2 men; mean age 77 years (SD 8), mean body mass 66 kg (SD 9), mean height 158 cm (SD 5)) also participated in the study. Exclusion criteria for PD patients were cardiovascular and respiratory symptoms and motor deficits that would affect high-speed pedalling on the cycloergometer. Subjects were informed about the aim of the study and gave written consent prior to the study. The study was approved by local ethics committee and carried out in accordance with the principles of the Declaration of Helsinki.
|
Table I. Clinical and anthropometric characteristics of patients with Parkinson’s disease |
||||||||
|
PD patient |
Age, years/sex |
Body weight, kg |
Height, cm |
Disease duration, years |
Affected upper extremity |
Hoehn & Yahr stage score |
Rigidity (UPDRS, item 22) |
|
|
Pre-IT score |
Post-IT score |
|||||||
|
PD#1 |
79/M |
71 |
157 |
10 |
R |
2.5 |
7 |
3 |
|
PD#2 |
69/M |
84 |
182 |
7 |
R |
1.0 |
3 |
2 |
|
PD#3 |
65/F |
64 |
162 |
7 |
L |
2.5 |
6 |
4 |
|
PD#4 |
68/M |
78 |
167 |
13 |
R |
2.5 |
4 |
6 |
|
PD#5 |
65/F |
61 |
155 |
2 |
R |
1.5 |
3 |
2 |
|
PD#6 |
60/M |
79 |
169 |
14 |
R |
3.0 |
9 |
8 |
|
PD#7 |
84/M |
73 |
165 |
4 |
L |
1.5 |
2 |
2 |
|
PD#8 |
78/F |
67 |
159 |
8 |
L |
3.0 |
5 |
5 |
|
PD#9 |
62/M |
63 |
161 |
16 |
R |
3.0 |
10 |
7 |
|
PD#10 |
58/F |
67 |
152 |
6 |
L |
3.0 |
5 |
4 |
|
PD#11 |
88/F |
67 |
149 |
4 |
R |
2.0 |
2 |
0 |
|
PD: Parkinson’s disease; UPDRS; Unified Parkinson’s Disease Rating Scale; IT: interval training; R: right side; L: left side; Pre-IT: before 8 weeks of interval training; Post-IT: after 8 weeks of interval training. |
||||||||
Experimental procedure
PD patients performed 8 weeks of IT on a stationary cycloergometer. Before and after the training cycle (pre- and post-IT, respectively) neurological symptoms of PD were assessed, and serum BDNF levels and myometry measurements were performed in a motor control laboratory. For all the above examinations, the pre-IT testing session was conducted 1–3 days before the IT cycle started, but the post-IT session was performed from 6–10 days after the last training session to avoid the lasting effect of training on the measured outcomes. PD patients were trained during their on-phase (when parkinsonian symptoms are alleviated after medication, which were mainly levodopa and piribedil or ropinirole) and tested during their off-phase (after an overnight 12-h withdrawal of anti-parkinsonian medication). In healthy, non-trained controls blood samples only were obtained, in order to evaluate changes in level of serum BDNF over the 8-week period.
Training protocol
IT consisted of 3 × 1-h training sessions per week for a total of 24 sessions, each consisting of a 10-min warm-up at slow voluntary speed, 40 min high-intensity interval exercise, and a 10-min cool-down at slow voluntary speed on a stationary cycloergometer (Ergomedic 874E; Monark, Vansbro, Sweden) that allowed measurements of cadence (revolutions per minute, rpm) and power (W). The session’s interval part consisted of 8 sets of 5-min intervals consisting of 3 min cycling at ≥ 60 rpm, preferably at 80–90 rpm (fast phase of IT); and 2 min at ≤ 60 rpm (slow phase of IT). Patients were rhythmically cued using a metronome and additional verbal encouragement to pedal at the appropriate speed for the fast or slow phase was provided by an instructor. All patients complied with instructions and accomplished the whole 8-week IT. The maximum heart rate (HRmax; in beats per minute, bpm) was measured with a Polar system (Kempele, Finland), and cadence and power were monitored and recorded during each training session (warm-up, interval exercise, and cool-down phases) on a computer. Patients’ perception of exercise intensity during fast vs slow phases was assessed using the Borg Rating of Perceived Exertion (RPE). Mean values from 24 training sessions for fast and slow phases were: cadence 67 rpm (SD 15) vs 42 rpm (SD 5); power 42 W (SD 37) vs 24 W (SD 21); %HRmax, 68% (SD 10) vs 62% (SD 8); and Borg RPE 17 (very hard) vs 13 (somewhat hard). Additional details of the training protocol and patients’ exercise data are provided elsewhere (5).
Clinical assessment of Parkinson’s disease
Patients with PD were clinically tested by an experienced neurologist blinded to the other patients’ results using the Hoehn & Yahr scale and Unified Parkinson’s Disease Rating Scale (UPDRS) (12). Parkinsonian rigidity scores of affected/more affected upper extremities (item 22 of UPDRS) and the sum of the motor components of the UPDRS (items 18–31) were assessed for each patient.
Myometric measurements
A model MyotonPRO myometer (Myoton AS, Tallinn, Estonia) was used to assess the mechanical properties of relaxed biceps brachii muscle (BB) of affected/more affected upper-extremities. Subjects were tested in the supine position with upper extremities along the trunk and with supination of the forearm (10, 11). An experimenter placed the testing end of the myometer on the skin surface overlying the relaxed BB and acquired 10 consecutive recordings. Stiffness (S-MYO; N/m) and frequency (F-MYO; Hz) were analysed, since they may reflect subtle alterations in the mechanical properties of muscle, associated with changes in the activity of motor units (11).
Blood sampling and brain-derived neurotrophic factor measurements
The detailed procedure has been described previously (5). In patients with PD and control subjects, pre- and post-IT blood samples were taken in the fasting state at rest between 08.00 h and 10.00 h. Blood was collected in anticoagulant-free tubes containing a clotting activator and maintained on ice (4°C) for 1 h, then centrifuged to isolate the serum, which was removed and stored at −80°C. An assay/microplate reader was used to determine BDNF concentration.
Statistical analysis
Myometric parameters were expressed as mean values calculated from 10 consecutive recordings. Trial-by-trial reproducibility of the measurements was assessed by intra-class correlation coefficient (ICC). The Shapiro-Wilk test was used to estimate parameter distribution. The Wilcoxon signed-rank test or Student’s t-test, as appropriate, was used for comparing pre- and post-IT measurements. The Pearson or Spearman’s correlation coefficients (r and ρ, respectively), as appropriate, was used to quantify the relationships between absolute values of IT-related changes in serum BDNF level, S- and F-MYO, and parkinsonian rigidity. p ≤ 0.05 was considered statistically significant for all analyses.
RESULTS
Reproducibility of myometric measurements
Pre- and post-IT ICC values for stiffness and frequency (pre: S-MYO = 0.998, F-MYO = 0.997 and post: S-MYO = 0.997, F-MYO = 0.992) were highly reproducible.
Effect of interval training on improvement in subjective and objective outcomes
Following training, parkinsonian rigidity decreased by 24% (p < 0.05) (Fig. 1A), the sum of the motor components of the UPDRS decreased by 25% (p < 0.05) (Fig. 1B), and S-MYO and F-MYO decreased by 18% and 13%, respectively (Fig. 1C, D) (p < 0.05). The serum BDNF level increased by 34% (p = 0.035) in PD patients, whereas no change was observed in the healthy control group (2% decrease; p = 0.809). Further analysis of these changes revealed positive correlations between the IT-induced increase in serum BDNF and decreases in parkinsonian rigidity (ρ = 0.603; p = 0.025), S-MYO (r = 0.827; p = 0.001), and F-MYO (r = 0.782; p = 0.002).
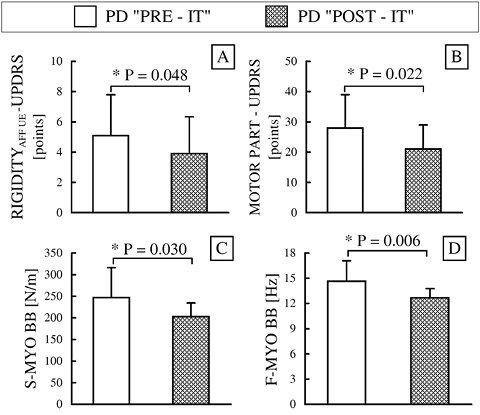
Fig. 1. (A) Clinical assessment of parkinsonian rigidity. (B) Parkinson’s disease (PD) motor symptoms based on the Unified Parkinson’s Disease Rating Scale (UPDRS); (C) myometric stiffness for biceps brachii muscle (BB); (D) myometric frequency for BB. Pre- and post-interval training (IT) values (before and after 8 weeks of IT, respectively). Data represent mean (column bars) ± standard deviation (error bars). *p < 0.05 (between sessions). S-MYO: myometric measurement of stiffness; F-MYO: myometric measurements of frequency.
DISCUSSION
Interval training-induced improvements in parkinsonian rigidity and muscle tone
The increased muscle tone and rigidity in PD are caused mainly by the impairment of central extrapyramidal dopaminergic brain regions that affect the activity of motor neurones in the spine, leading to an increased tonic stretch reflex (7, 8). Intake of L-dopa restores extrapyramidal system activity and alleviates parkinsonian rigidity (7, 8), and consequently improves muscle tone in PD patients (11). Tandem cycloergometer training produces beneficial effects on basal ganglia functioning that are similar to those of anti-parkinsonian medication (1); indeed, high-intensity treadmill training improved central tonic inhibition in PD patients, which is also related to extrapyramidal system functioning (2). The results of these studies indicate that exercise can potentially induce neuroplastic changes (4), since patients consciously adjusted their activity to the set pace and intensity. The role of rhythmicity and cognitive engagement in goal-based exercise was also evidenced in a study where PD symptoms were alleviated and motor control was improved after 13 weeks of dance training (3). Therefore, based on the previous studies, it can be postulated that IT with rhythm cuing, verbal encouragement, and cognitive engagement can affect neuroplastic potential in a manner similar to other types of goal-based exercise. It can also be assumed that the beneficial changes recorded in the present study, i.e. the IT-related alleviation of parkinsonian rigidity and decrease in BB muscle tone, result from an improvement in basal ganglia functioning (such as that produced by L-dopa intake) and consequent effects on spinal cord activity-related muscle tone regulation (7, 8).
Interval training-induced increase in basal serum brain-derived neurotrophic factor
It has been reported that physical exercise increases serum BDNF levels in healthy humans (13) and PD patients (5, 6), reflecting the level of the BDNF in the brain (14). BDNF is a biochemical signal responsible for neuronal protection, survival, and remodelling, axonal and dendritic growth, and synaptogenesis (4). In the human brain, BDNF is expressed in the hippocampus, claustrum, amygdala, bed nucleus of the stria terminalis, septum, and the nucleus of solitary tract (15). Some of these areas constitute, or are connected with, the basal ganglia. In the PD patients examined here, increased BDNF expression resulting from cycloergometer interval training might have stimulated neuroplastic mechanisms that improve functioning of pathologically altered neuronal networks in basal ganglia, and especially in the dopaminergic nigrostriatal tract, thereby ameliorating the symptoms of PD (4).
Correlation analysis showed that increased serum BDNF level was positively correlated with an improvement in parkinsonian rigidity and decrease in resting muscle tone. These correlations provide support for the occurrence of IT-induced neuroplastic changes in the basal ganglia of these PD patients. Although the results do not provide direct evidence of structural changes in the basal ganglia, they are supported by studies in animal models of PD (16, 17), which showed that an increased neurotrophin level in the basal ganglia has a cytoprotective effect on dopaminergic structures, ultimately improving basal ganglia functioning, PD symptoms, and motor control.
Two limitations to this study are the small sample size and the inclusion of healthy subjects as the control group. A delayed entry study design was initially considered but not pursued, since patients’ physical activities could undergo season-specific changes. All PD patients in this study underwent 8 weeks of IT during the spring, eliminating this potential variability. The fact that pre- and post-IT serum levels of BDNF did not differ in non-trained PD patients in another study (6) or in the non-trained healthy controls tested here, and that an increase in BDNF levels in PD patients resulted from training and was correlated with improved subjective and objective outcomes, indicates that positive changes in these patients were not time-dependent fluctuations.
The findings from this pilot study provide support for the beneficial effects of exercise in PD rehabilitation, and will form the basis for a future randomized control trial study.
We conclude that the training-induced alleviation of parkinsonian rigidity and decrease in muscle tone may be associated with neuroplastic changes in the central nervous system caused by a training-induced increase in basal serum brain-derived neurotrophic factor.
Acknowledgements
The authors would like to thank the PD patients who participated in this study.
This study was supported by the Ministry of Science and Higher Education of Poland (grant no. 0247/p01/2010/70) awarded to J. Marusiak. J. Majerczak and J. A. Zoladz were supported by funds for the statutory research in 2013 for the University School of Physical Education in Krakow, Poland.
References
