Peter Holló, Jozsef Szakonyi, Dorottya Kiss, Hajnalka Jokai, Attila Horváth and Sarolta Kárpáti
Department of Dermatovenereology and Dermatooncology, Semmelweis University, Mária str. 41, HU-1085 Budapest, Hungary. E-mail: holpet@bor.sote.hu
Accepted August 17, 2011.
Lichen planus (LP) is a relatively common inflammatory dermatosis (with a prevalence of approximately 0.5%), characterized by violaceous, polygonal, flat pruritic papules. It can involve both skin and mucous membranes and usually occurs in middle-aged persons. Mild cases usually heal spontaneously, but patients with widespread lesions are sometimes hard to treat. Different treatment modalities are available. Topical anti-inflammatory treatment with steroid ointments or calcineurin inhibitors have limited efficacy. Phototherapy, systemic treatment with retinoids, and depot steroids have been administered with limited success, and systemic ciclosporin is usually effective.
Tumour necrosis factor alpha (TNF-α) has a key role in the pathogenesis of LP (1). Thalidomide, a potent anti-inflammatory drug, acting inter alia through TNF-α-mediated cellular responses, has been found effective in the treatment of LP (2). Only a few data on the efficacy of TNF-α inhibitors in LP are available in the literature (3, 4).
CASE REPORT
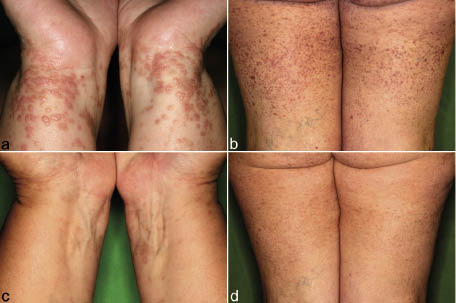
A 39-year-old woman developed 2–3-mm large itchy purplish lichenoid papules and plaques with Wickham’s sign on the whole body surface, in especially large numbers on the buttocks, lower legs and forearms. White reticular striation and plaques were found bilaterally on the buccal mucosa. The diagnosis of lichen planus was confirmed by histology. The patient had no previous history of any other diseases. A general examination did not detect any underlying systemic disorders or background infections. The patient did not take any drugs. Hepatitis B and C serologies were negative. Topical steroid creams, systemic glucocorticoids (0.5 mg/kg, 4-week-long therapy) and 20 sessions of psoralen plus ultraviolet A (PUVA) treatment were administered without any therapeutic effect. Acitretin therapy maintained for 3 months at a dose of 0.5 mg/kg/day did not result in clinical improvement. A side-effect of hair loss was detected. Meanwhile, her skin symptoms progressed significantly. Ten months after disease onset the TNF-α inhibitor adalimumab was requested for individual off-label use, and initiation of therapy was permitted by the National Institute of Pharmacology.
Adalimumab was administered subcutaneously for 3 months at the recommended dose (80 mg induction dose followed by 40 mg every second week from day 7) usually accepted for the treatment of psoriasis (Fig. 1).
Itching decreased after 2 weeks, accompanied by a slow regression of the skin symptoms. The cutaneous lesions healed in 2 months with residual pigmentation. Since the patient remained symptom-free, treatment was terminated. During a 6-month follow-up period no recurrence of skin or mucosal symptoms was observed. No side-effects of the adalimumab therapy were detected.
Fig. 1. (a, b) Disseminated papules of lichen planus, (c, d) regressed after adalimumab treatment.
DISCUSSION
Production or release of TNF-α within the LP lesions may play a substantial regulatory role in the immunopathogenesis of the disease (1).
The use of TNF-α inhibitor biologics for the management of LP has been previously described in the literature. In one case report etanercept was found to be beneficial for oral LP, and in another adalimumab for cutaneous and mucosal LP (3, 4). Etanercept was also reported to be effective in the treatment of nail LP (5).
At the same time LP is discussed nowadays as one common side-effect of TNF-α. Considering the same phenomenon in psoriasis that TNF-α inhibitors sometimes provoke psoriatic skin signs, an alternative pathway of immune-mediated inflammatory processes should be hypothesized in TNF-α inhibitor induced forms of both skin diseases (6).
This study further confirms the rarely reported observation that TNF-α inhibitors (here adalimumab) are effective in the treatment of LP. The therapeutic value of this novel off-label use should be assessed in controlled clinical trials.
The authors declare no conflicts of interest.
REFERENCES