Yoon Seok Yang1#, Soo Ick Cho1#, Min Gyu Choi2, Young Hee Choi3, In Suk Kwak4, Chun Wook Park1 and Hye One Kim1
Departments of 1Dermatology, 3Pathology, and 4Anesthesiology and Pain Medicine, College of Medicine, Hallym University, and 2Department of Computer Science, Kwangwoon University, Seoul, Korea
#These authors contributed equally and should be considered co-first authors.
Post-burn pruritus is a common distressing consequence of burn wounds. Empirical treatment often fails to have a satisfactory outcome on post-burn pruritus, as the mechanism of post-burn pruritus has not been fully elucidated. The aim of this study was to evaluate the manifestation of transient receptor potential (TRP) channels in post-burn pruritus. Fifty-one burn patients with (n = 33) or without (n = 18) pruritus were investigated, including skin biopsies. Not unexpectedly, the scarred body area was larger in the former group. In immunohistochemistry, TPRV3 was significantly elevated in the epidermis of burn scars with pruritus. Furthermore, real time- PCR showed that mRNA of TRPA1 and TRPV4 was increased in itching burn scars. Staining for substance P and CGRP did not differ between the 2 grouped, but the former neuropeptide was increased in burn scars. These results may help determine a specific therapeutic approach for post-burn pruritus. Key words: post-burn pruritus; TRPA1; TRPV3; TRPV4.
Accepted Apr 1, 2014; Epub ahead of print Apr 3, 2014
Acta Derm Venereol
Prof. Hye One Kim, Department of Dermatology, College of Medicine, Hallym University, Kangnam Sacred Heart Hospital, 948-1, Daerim 1-dong, Yeungdeungpo-gu, Seoul, Korea. E-mail: hyeonekim@gmail.com
Pruritus following burns is a common and stressful complication after burn injury (1). A prospective longitudinal cohort study identified that the percentage of patients affected by mild to severe pruritus was 87% at 3 months, 70% at 12 months and 67% at 24 months after the burn (2). Still, the pathophysiology of post-burn pruritus has not been fully investigated.
Transient receptor potential (TRP) channels are a group of ion channels located mostly on the plasma membrane of numerous cells. TRP channels are distributed in non-neuronal skin cells such as keratinocytes or merkel cells as well as neuronal cells (3). There are 2 major subtypes of TRP: Group 1 includes TRPC (canonical), TRPV (vanilloid), TRPM (melastatin), TRPN (NOMPC-like) and TRPA (ankyrin); group 2 includes TRPP (polycystin) and TRPML (mucolipin) (4). Many of these channels mediate various sensations such as pain, hotness, warmth or coldness, different kinds of tastes, pressure, and vision (4). It is now emerging that these channels are integrators of diverse noxious stimuli, including those that induce sensations of itch (5, 6). It was shown that TRPV1-expressing neurons are needed for pruritus induction by histamine as well as pain (7). Recently, other TRP channels have also been investigated as the main inducer of itch (5, 6). TRPV1, 3, 4 and TRPA1 were chosen for this study since they are expressed in skin and have been suggested to be related to itching in previous studies (8–11).
The aim of this study was to (i) determine the clinical and morphologic characteristics of burn scars with pruritus using non-invasive bioengineering techniques, (ii) investigate histopathological findings in scars accompanying post-burn pruritus, (iii) investigate the distribution of TRPV1, TRPV3, TRPV4 and TRPA1 in burn scar by immunohistochemistry (IHC), (iv) determine the expression levels of mRNA of TRPV1, TRPV3, TRPV4 and TRPA1 in burn scars by quantitative real-time PCR, and (v) compare these results with normal skin tissues from the same donors and with those of non-pruritic burn patients.
MATERIAL AND METHODS (see Appendix S11)
RESULTS
Characteristics of patients. The 51 burn patients consisted of 37 males and 14 females with ages ranging between 8 and 60 years (mean ± SD, 35.0 ± 14.8). The mean duration of burn injury was 113 ± 132 months. The most common burn sites were the hands/feet (n = 20, 39.2%), followed by arms/legs (n = 19, 37.3%), head/neck (n = 7, 13.7%) and the trunk (n = 5, 9.8%). The patients had total body surface area (TBSA) from 1–97% (mean ± SD, 23.4 ± 25.2). Thirty-four patients (66.7%) had undergone surgical procedures (split thickness skin graft, n = 21; full thickness skin graft, n = 7; and burn scar contracture, n = 6). The most common cause of the burn injuries was scalding (37.3%), followed by flame (31.4%) and electric burn (7.8%).
Comparison of clinical features between burn patients with and without pruritus (Table I). Thirty-three (64.7%) of the 51 patients suffered from pruritus on their burn scars (Group A) and 18 did not (Group B). The mean intensity of pruritus was 4.6 ± 3.3 on the VAS. Group A patients were slightly older (mean ± SD, 38.0 ± 12.8 vs. 29.4 ± 16.8 years; p = 0.068) and had broader TBSA (mean ± SD, 25.6 ± 25.3 vs. 9.7 ± 12.5%; p = 0.004) than Group B patients. The most common scar site in Group A was the hands/feet and arms/legs (n = 12, 36.4%), and in Group B was the hands/feet (n = 8, 44.4%). The distribution of scar sites was similar in both groups (p = 0.656). The mean duration of the burn injury was shorter in Group A than in Group B, although not significant. Group A had a higher proportion of males (84.8% vs. 50%; p = 0.008). The 2 groups had similar proportions of previous surgical procedures. As subjective scar severity, the total PSAS scores were significantly higher in Group A than in Group B (35.7 ± 9.7 vs. 21.6 ± 6.1; p < 0.001). As objective scar assessment, total OSAS scores were also significantly higher in Group A than in Group B (31.4 ± 7.0 vs. 22.6 ± 6.48; p = 0.001). With non-invasive bioengineering measurements, there was no significant difference in MI, EI, TEWL and sebum excretion between Group A and B. Only the thickness of burn scar according to ultrasonography was greater in Group A than in Group B (Table I).
Table I. The clinical and histopathological differences between burn patients with (Group A) and without (Group B) pruritus
|
Group A |
Group B |
p-value |
|
|
Clinical characteristics |
|||
|
Age, years |
38.0 ± 12.8 |
29.39 ± 16.84 |
0.068 |
|
Burned area (% of TBSA) |
25.6 ± 25.3 |
9.7 ± 12.5 |
0.004* |
|
Gender (male), % |
84.8 |
50.0 |
0.008* |
|
Previous procedures, % |
69.7 |
61.1 |
0.534 |
|
Duration of burn injury, days |
90.2 ± 129.9 |
153.79 ± 128.1 |
0.100 |
|
Total PSAS |
35.7 ± 9.7 |
21.6 ± 6.1 |
< 0.001* |
|
Total OSAS |
31.4 ± 7.0 |
22.6 ± 6.4 |
0.001* |
|
Non-invasive measurements |
|||
|
Melanin index |
274.0 ± 129.9 |
254.6 ± 138.6 |
0.681 |
|
Erythema index |
408.3 ± 107.7 |
399.9 ± 124.0 |
0.834 |
|
TEWL |
28.5 ± 15.4 |
34.6 ± 15.6 |
0.268 |
|
Sebum secretion |
12.6 ± 29.8 |
6.6 ± 14.6 |
0.524 |
|
Thickness of the scar |
0.3 ± 0.2 |
0.2 ± 0.1 |
0.036* |
|
Histological features |
|||
|
Mononuclear cell infiltration score |
1.5 ± 0.9 |
1.3 ± 0.8 |
0.444 |
|
Mast cell counts |
9.7 ± 5.3 |
8.5 ± 3.8 |
0.413 |
|
Elastic fibres (density), % |
21.2 |
61.1 |
0.004* |
|
Collagen fibres (density), % |
78.8 |
88.9 |
0.366 |
|
Neuropeptides (IHC), semiquantitative scoring (0–4) |
|||
|
Substance P in epidermis |
2.8 ± 0.6 |
2.5 ± 0.9 |
0.299 |
|
Substance P in dermis |
0.8 ± 1.3 |
0.8 ± 1.1 |
0.973 |
|
CGRP in epidermis |
1.9 ± 1.1 |
1.6 ± 1.0 |
0.408 |
|
CGRP in dermis |
0.4 ± 0.7 |
0.5 ± 0.9 |
0.572 |
*Significantly different at p < 0.05
TBSA: total body surface area; PSAS: patient scar assessment scale; OSAS: observer scar assessment scale; TEWL: transepidermal water loss; IHC: immunohistochemitry.
Histopathological analysis. In all patients, the epidermis was significantly thicker in the burn scars than in normal skin (7.5 ± 3.0 vs. 4.0 ± 1.0; p < 0.001). The mast cell counts were significantly greater in the burn scars than in normal skin (8.7 ± 4.1 vs. 7.1 ± 2.9; p = 0.002). The mast cell distribution was more diffuse in burn scars than in normal skin (92.1% vs. 78.4%; p = 0.014). Collagen bundles were thinner in the burn scars than normal skin (51.0% vs. 2.0%, p < 0.001) and more dense than in normal skin (80.4% vs. 3.9%, p < 0.001). The elastic fibres within the dermis were sparser in burn scars than in normal skin (41.1% vs. 9.8%; p = 0.049). Mononuclear infiltration was almost the same in burn scars and normal skin (data not shown). With regard to SP and CGRP, only the expression of SP in the epidermis was elevated in burn scars (1.97 ± 0.62 vs. 2.63 ± 0.73; p = 0.002) (data not shown).
There were no significant differences in normal skin between Groups A and B. In the burn scars, elastic fibres within the dermis were sparser in Group A than in Group B (21.2% vs. 61.1%; p = 0.004). However, other histopathological parameters did not show any significant differences between Groups A and B (Table I).
Immunoreactivity of TRP channels in burn scar and normal skin (Figs 1, 2). Immunoreactivity of TRPs was demonstrated overall on basal and suprabasal keratinocytes of burn scar and normal skin; in particular, TRPV3 stained the basal layer of the epidermis. In the dermis, more intense expression of TRPV4 and TRPA1 was seen, mainly on mononuclear cells and nerve fibres. In addition, anti-TRPA1 markedly stained the mast cells.
The baseline of TRP channels in normal skin was varied from subject to subject, so the ratio (%) of the changes of TRP channel expression in burn scars vs normal skin in the same subject were analyzed.
Only TRPV3 expression in the epidermis was significantly increased in Group A compared to Group B (95.3 ± 137.0% vs. 9.9 ± 93.0%, p = 0.048). TRPV4 and TRPA1 expression also increased in Group A, but the result was not statistically significant (20.2 ± 69.4% vs. 0.2 ± 44.0%, p = 0.415 and 12.1 ± 49.2% vs. –0.4 ± 64.7%, p = 0.582, respectively).
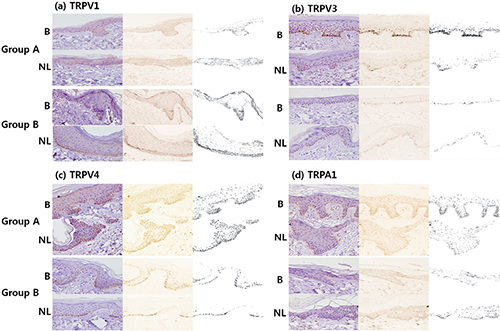
Fig. 1. Immunohistochemical study of normal skin and burn scars with various transient receptor potential (TRP) channels (a, TRPV1; b, TRPV3; c, TRPV4; d, TRPA1). All could be demonstrated on basal and suprabasal keratinocytes of burn scars and normal skin. (b) Particularly in the itching burn scars, there was prominant staining of TRPV3 in basal keratinocytes. (c, d) TRPV4 and TRPA1 expression was also increased in Group A, but that was not statistically significant. Group A: patient with pruritus, Group B: patient without pruritus B: burn scar, NL: normal skin.
Only TRPV3 immunoreactivity had a positive correlation with the VAS level of pruritus (r = –0.430; (p = 0.018).
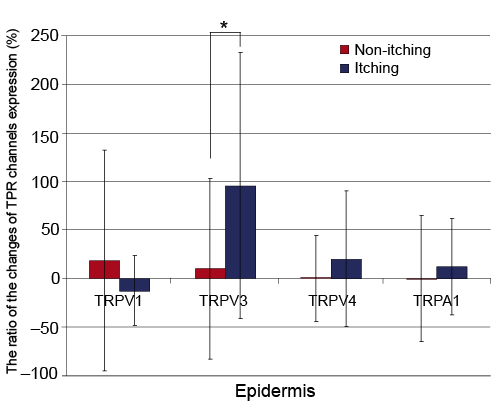
Fig. 2. Comparison of protein expression of Transient receptor potential (TRP) channels of non-itching and itching burn scars. The relative differences in the immunohistochemical staining intensity of various TRP channels are presented as the ratio of the changes of TPR channels expression between itching (n = 33) or non-itching (n = 18) burn scars and the normal skin of the same patients using software. Only TRPV3 expression in the epidermis was more significantly increased in itching burn scars than in non-itching burn scars. Other TRP channels did not show a significant difference in the epidermis and dermis.
mRNA quantification of TRP channels in burn scar and normal skin. Burn scars had elevated levels of TRPV1 and TRPV4 mRNA compared to normal skin (TRPV1; 1.755 ± 1.431 vs. 0.851 ± 0.414, p = 0.001, TRPV4; 1.189 ± 0.612 vs. 0.764 ± 0.347, p = 0.009). TRPV3 and TRPA1 did not show any significant difference. In burn scars, Group A had increased TRPA1 mRNA compared to Group B, while other channels did not (0.781 ± 0.285 vs. 0.456 ± 0.229, p = 0.006).
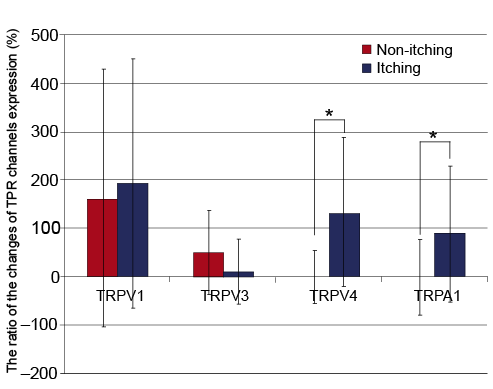
In the same way as described above, the ratio of the changes of TRP channels expression from burn scars to normal skin in the same subjects were calculated. TRPV4 (131.34 ± 151.7% vs. 1.3 ± 54.9%, p = 0.002) and TRPA1 (90.2 ± 139.5% vs. 0.2 ± 77.4%, p = 0.011) were more significantly elevated in Group A than Group B (Fig. 3).
In the same manner, the changes in mRNA of TRP channels expression from burn scars to normal skin were analysed, and the correlation between VAS and TRPV1 was r = –0.018 (p = 0.933); TRPV3 r = 0.053 (p = 0.802); TRPV4 r = 0.511 (p = 0.009); and TRPA1 r = 0.409 (p = 0.042). TRPA1 and TRPV4 showed a positive correlation in which expression of mRNA increases according to the level of pruritus.
DISCUSSION
In this study, we have demonstrated the clinical, histopathological characteristics and the expression of TRPA1, TRPV3 and TRPV4 in the skin of burn patients with or without post-burn pruritus. The patients with post-burn pruritus had broader burned areas and more severe scar grade determined by subjective and objective methods than those without pruritus. In IHC, TPRV3 was more strongly stained in the epidermis of burn scars of post-burn pruritus patients than in normal skin of the same patients. TRPV4 and TRPA1 also exhibited increased expression of burn scars of post-burn pruritus, but it was not statistically significant. No TRP channels showed changes in the dermis of the pruritic burn scar by IHC. Similarly, the mRNA of TRPA1 and TRPV4 was significantly increased in burn scar tissue of patients with post-burn pruritus compared to those of non-itching burned patients.
The length of time after the burn injury, TBSA, the number of surgical procedures and psychological characteristics comprised the risk factors of post-burn pruritus in previous studies (2, 14). In our study, a broader TBSA and male gender were characteristic features in patients with post-burn pruritus. The TBSA results was in agreement with previous studies. It would appear that male patients tend to have broader scar areas because they are more often burned in serious industrial accidents, with the result that they have a higher prevalence of post-burn pruritus. With non-invasive measurements, a thickened burn scar was a significant feature of post-burn pruritus, suggesting that hypertrophic and immature burn scars are closely related to pruritus as revealed in previous studies (2, 15). However, there were no differences in those histopathological features between pruritic and non-pruritic burn scars.
Recently, TRP channels are expected to play a new role with the pruritus pathogenesis because of an assumption that the itching sensation would be significantly affected by temperature (16). As thermo-sensitive channels in most cases, TRP channels are believed to have a close link with the itch pathway (17–19). Additionally, the itching sensation is thought to be related not only to the neuronal network in the skin but also to non-neuronal cells such as mast cells and keratinocytes (20). More studies are needed to discuss how TRP channels explain both neuron and non-neuronal cell networks in the itch pathway at the same time, and yet, what is being considered possible here is that TRP channels could be a part of the itch pathway. Finally, it is revealed that some antagonists or strong agonists have potential as antipruritic medication (20–23). Since TRPA1 is activated by bradykinin and other known pruritogenic inflammatory agents, it may be related to the pruritus (20).
Real time-PCR and IHC results confirmed that mRNA and protein levels of TRPA1 are increased in patients with post-burn pruritus. Previous studies have confirmed that TRPA1 is expressed in non-neuronal cells such as keratinocytes, melanocytes, and fibroblasts as well as cutaneous sensory neurons in the skin (24). Although the results were not statistically significant, it is interesting that tissues of patients with post-burn pruritus had a higher expression of TRPA1 in the epidermis than those without pruritus. In particular, TRPA1 is strongly stained in mast cells in the dermis. In addition, real time-PCR results confirmed that mRNA levels of TRPA1 are increased in patients with post-burn pruritus. Generally, the post-burn pruritus is not relieved by antihistamines, which would make the symptoms intractable to treatment. A previous study reported that TRPA1 is connected to histamine-independent, Mas-related G protein-coupled receptor-mediated itch (25). Taken together, it can be hypothesised that TRPA1 expression on nerve endings, keratinocytes, mast cells and other non-neuronal cells contribute to the post-burn pruritus.
In this study, TRPV3 was significantly increased in the epidermis of patients with post-burn pruritus. Recent studies reported that TRPV3 was related to pruritus (26–28). An investigation with transgenic mice, gain-of-function of TRPV3 caused pruritus (27). TRPV3 is also believed to be responsible for development of the epidermal layer, which is important to the skin barrier function and the pathogenesis of xerotic eczema (28, 29). Since TRPV3 is mostly observed in the epidermis and not much in the dermis with IHC, there seemed to be no significant differences in the data with real-time PCR that included not only epidermis but also thicker dermis (21).
TRPV4 mRNA was significantly increased and TRPV4 protein was also elevated in dermis of the patients with post-burn pruritus. TRPV4 is present in sensory neurons and epidermal keratinocytes (8, 21). TRPV4 becomes active at temperatures higher than 25°C but also by arachidonic acid and its derivatives, the endocannabinoid anandamide via the generation of lipid peroxidation products such as eicosanoid, the recognised inflammatory pruritogens (21, 30). In addition, TRPV4 interacts with TRPV1 for the skin permeability barrier homeostasis, and if it is dysregulated, prutitus occurs eventually (21, 31). This may suggest that TRPV4 is also related to pruritus.
To the best of our knowledge, no study relating to post-burn pruritus has examined the correlation between the level of pruritus and TRP channels expression. While our study had a limitation on the number of patients, our findings showed that more severe pruritus was related to TRPV3, TRPA1 and TRPV4. It will be meaningful to conduct research on developing a treatment with an antagonist against TRPA1 and TRPV4 for patients who suffer from severe pruritus.
ACKNOWLEDGEMENTS
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2012R1A1B3002196, 2011-0013003, 2011-0012878), Hallym University Research Fund (HURF-2013-33), the Research Grant of Kwangwoon University in 2014 and Amore Pacific Co. Ltd., 2011.

1http://www.medicaljournals.se/acta/content/?doi=10.2340/00015555-1858
REFERENCES
