OBJECTIVE: To evaluate whether subjects with subclinical recurrent neck pain have an altered interpretation of neck proprioceptive signals.
DESIGN: A comparative group design.
SUBJECTS: Twelve subjects with recurrent neck pain and 12 control subjects.
METHODS: The shoulder, head, trunk and whole-body position were measured under the following conditions: active and passive elevation and depression of the right shoulder and vibration of the trapezius muscle.
RESULTS: During passive shoulder movements both groups moved their head in the opposite direction (p < 0.05). During passive elevation subjects with recurrent neck pain showed a significant over-appraisal of shoulder position. Both groups responded similarly to active movements. Subjects with recurrent neck pain and control subjects showed similar trunk and head-to-trunk movements during shoulder movements. Subjects with recurrent neck pain, however, made larger movements compared with healthy subjects (p < 0.05). During trapezius muscle vibration similar whole-body movements were found in both groups.
CONCLUSION: These results show a modified interpretation of neck proprioceptive signals in subjects with recurrent neck pain and may reflect an offset in the egocentric reference frame or a decreased capacity to switch between reference frames. Better insight into these mechanisms might lead to better evaluation and treatment of subjects with recurrent neck pain and to a reduction in recurrent episodes.
Key words: neck pain, proprioception, reference frame, postural control, shoulder, head.
J Rehabil Med 2008; 40: 426–432
Correspondence address: Simon Brumagne, Department of Rehabilitation Sciences, Faculty of Kinesiology and Rehabilitation Sciences, K. U. Leuven, Tervuursevest 101, BE-3001 Leuven, Belgium. E-mail: Simon.Brumagne@faber.kuleuven.be
Submitted February 15, 2007; accepted January 22, 2008
INTRODUCTION
Neck pain is a common medical condition causing substantial personal and financial costs (1). The pain is often persistent or recurrent in nature. The underlying mechanisms of neck pain are still a matter for debate. One of the identified problems in persons with neck pain is a change in proprioception. Persons with neck pain have been observed to have less accurate and precise position sense of the head and the upper limbs (2–4). The majority of investigators have concentrated on proprioceptive acuity when investigating position and movement sense. They have assumed that the peripheral sensory signals were impaired because of pain and injury. We believe, however, that central nervous system (CNS) changes rather than peripheral signal problems are the key to understanding the clinical observations. The interpretation and transformation of afferent signals into useful information could play an important role in effective neck position and movement sense (5).
The central nervous system uses the signals provided by the proprioceptors to build up an internal reference frame of the musculoskeletal system (body schema) and to recalibrate it (6, 7). In order to form a useful kinaesthetic sensation the CNS has to interpret the proprioceptive signals within a reference frame (5). Two kinds of reference frames are frequently described: a reference frame associated with the body (egocentric) and a reference frame associated with the external world (allocentric) (5). The same proprioceptive signals can elicit different kinesthetic sensations and the mechanisms of interpretation determining the characteristics of these sensations are closely connected with the choice of a reference system (8, 9). Knox & Hodges (10) and Knox et al. (11, 12) have shown that the perceived position of the head and neck influences the perception of elbow positioning. Knowledge of head position thus appears to be a vital component in the organization of intrinsic sensory information for upper limb position and possibly for other joints.
An important property of the postural control system is its ability to gate sensory input in accordance with the internal representation of the current posture, so as to avoid undesirable responses triggered by external or internal perturbations (13). If the quality of input from a particular body location deteriorates due to injury, disease, or normal ageing, the CNS might increase the weighting of input from other locations. Re-weighting of sensory signals (gain control process) has been reported in patients with spasmodic torticollis and in patients with low back pain (14, 15). The proprioceptive deficits found in persons with neck pain could be a consequence of re-weighting of sensory signals. Altered proprioceptive signals from neck muscles may result in the CNS down-weighting proprioceptive signals from neck muscles and instead up-weighting other sensory inputs, such as signals from trunk muscles or vestibular signals.
The purpose of the present study was to evaluate how neck proprioceptive signals are processed and interpreted within an egocentric reference frame and to determine whether this is altered in subjects with subclinical unilateral recurrent neck pain (RNP).
Subjects who experience symptoms of neck pain but are not yet receiving any treatment are classified as having minor musculoskeletal or “subclinical” neck pain (16). The features characterizing this group are of particular interest because they might lead to a better classification of persons with neck pain and to a better early intervention to stop the progression of neck pain. Active and passive shoulder movements and trapezius muscle vibration were used to investigate the proprioceptive contribution to shoulder, head and trunk position and whole-body orientation. Both active and passive shoulder movements were used because it has been shown that active contraction of muscles increases proprioceptive acuity compared with passive movements (17). This is partly due to the sensitization of the fusimotor system through active contraction. Another important aspect of active movement is that the efference copy (corollary discharge) of the motor command gives feedback and therefore contributes to the sensation of force, heaviness and timing and updating the body schema (18). Muscle vibration is an effective method of selectively activating the muscle spindles (19). Muscle/tendon vibration and microneurography studies have demonstrated a major role for muscle spindles in proprioception (19, 20). Muscle spindles activated by vibration cause a sensation of muscle lengthening, thereby creating an illusion of movement. During active, passive or illusory shoulder movements the body equilibrium is disturbed. The CNS tries to counterbalance this disturbance by correcting the body position and orientation. This correction of body position and orientation reflects to which extent the afferent muscle signals are used in postural control. It also could reflect which part of the body is seen as a stationary point and which body part as moving (egocentric reference frame) (5).
METHODS
Subjects
Twelve young subjects with subclinical unilateral RNP and 12 asymptomatic control subjects gave their informed consent to participate in the study. Subjects in the neck pain group (2 men, 10 women, mean age 23 years (standard deviation (SD) 3.3)) had recurrent neck and shoulder pain (region of the upper trapezius) at the right side for a period of at least 6 months (mean duration 39 months (SD 19.9)). A body chart was used to determine location of neck pain. Subjects were excluded if they had bilateral neck pain or any neurological signs, had other musculoskeletal or neurological disorders, had undergone cervical spine surgery or were undergoing treatment at the time of testing. They were not taking medication at the time of testing. Asymptomatic control subjects (4 men, 8 women; mean age 22 years (SD 1.4)) were included if they were free of neck pain and had no neurological or musculoskeletal disorders. All subjects were right-handed.
Subjects with neck pain completed the Neck Disability Index (mean 6.25 (SD 2.34)). The maximum score for this index is 50 (21). The subjects selected for this study were only mildly disabled (score between 5 and 14). They experienced symptoms of neck pain, but were not yet receiving any treatment and so were classified as having minor musculoskeletal or “subclinical” neck pain. They also indicated their perceived pain before, during and after the test on a 10-cm visual analogue scale (VAS) anchored with “no pain” (score = 0) and “the worst possible pain imaginable” (score = 10). None of the subjects experienced pain before, during or after the test protocol. The mean VAS score during the day was 1.6 cm (SD 1.0). All procedures were conducted according to the Declaration of Helsinki and the study was approved by the local ethics committee. Due to logistical limitations the test leader could not be blinded.
Materials
An electrogoniometer (Biometrics, Amsterdam, The Netherlands) was placed on the sternum and the right clavicle to measure the elevation and depression of the shoulder. For the head movement a piezoresistive accelerometer (IC Sensors, Bedlington, Northd, UK) was placed on the left temporal bone. To record possible differences in the position of the foot centre of pressure (COP) during the different test conditions, subjects stood on a 6-channel force plate (Bertec, OH, USA), feet 10 cm apart in a comfortable self-selected toe position. The force plate data, electrogoniometer and accelerometer data were sampled at 500 Hz using a Micro1401 data acquisition system and Spike2 software (Cambridge Electronic Design, Cambridge, UK). A body chart was used to evaluate the subjective shoulder position (Fig. 1). The distance between the lines was 5º. The lowest line was 65º and the highest was 150º. The lines represent the angle between sternum and clavicle. The entire procedure was recorded using a digital video camera (DCR-TVR80E, Sony Corporation, Tokyo, Japan), placed 3 m in front of the subject and perpendicular to the manubrium sterni of the subject. In order to be able to measure trunk movement and head movement relative to trunk movement, reflective markers were placed on the extremitas acromialis of the clavicle, on the head (one on the top of the nose, one between the eyes and one on the temporal bone) and on the sternum (one on the manubrium sterni and one 10 cm below).
In order to control whether there was muscle activity during passive movements and vibration of the trapezius muscle, and to see whether there are differences in muscle activity between subjects with RNP and control subjects, electromyographic (EMG) recordings were obtained from the right upper trapezius muscle and the right latissimus dorsi muscle (Myosystem, Noraxon, USA). The surface-electrodes (Medicotest blue sensor, INC, Rolling Meadows, IL, USA) were placed 2 cm apart, over the muscle belly, and perpendicular to the direction of the muscle fibres. The first electrode of the upper trapezius muscle was placed 2 cm lateral from the midpoint of the line between C7 and the acromion, the second one lateral from the first. The latissimus dorsi muscle’s electrodes were placed just below the teres major muscle. A ground electrode was placed on the spina iliaca anterior superior. The EMG data were amplified (× 1000), band-pass filtered (10–500 Hz) and sampled at 2000 Hz using a Micro1401 data acquisition system and Spike2 software (Cambridge Electronic Design, Cambridge, UK).
For the vibration condition a muscle vibrator (Maxon Motors, Sachseln, Switzerland) was used. The frequency of vibration was set at 70 Hz and the amplitude was approximately 0.5 mm.
Experimental procedure
Subjects stood barefoot in a comfortable position on the force plate. Each subject was tested under 6 experimental conditions sequentially presented as follows: (i) quiet stance, (ii) active shoulder elevation, (iii) active shoulder depression, (iv) passive shoulder elevation, (v) passive shoulder depression, (vi) upper trapezius muscle vibration. The right side was tested.
For the active elevation condition, the subject was asked to lift the right shoulder as high as possible and to hold it there. For the active shoulder depression condition, the subject was asked to depress the right shoulder as low as possible and to hold it. A demonstration was given by the test leader prior to the start of each condition. For the passive conditions, an examiner standing beside the subject moved the shoulder in maximal elevation or depression. Force was applied on the arm above the elbow (Fig. 2). During these trials force plate data were not used in the final data analysis. For the vibration condition, the muscle vibrator was placed on the right upper trapezius muscle and fixed by means of elastic Velcro straps.
Fig. 2. (a) Active and (b) passive shoulder elevation.
One trial was performed for each experimental condition. Each trial lasted 60 sec. The movements or vibration began 15 sec after the start of the trial and lasted 20 sec. A body chart was placed in front of the subject at eye-level, in order to avoid any head movement. At the end of the 20 sec subjects had to point with their left index finger their shoulder position on a body chart (Fig. 1). For this reason visual information was available during all trials.
Data analysis
The data were collected, processed and calculated using Spike2 and Microsoft Excel software. Displacements of the COP in anterior-posterior direction were estimated from the raw force plate data. Further data reduction was performed by calculating the mean COP displacement (in cm) and the mean head and shoulder displacements (in degrees) during 15 sec prior to and 15 sec during the movement or vibration. Trunk movements and head movements relative to the trunk were measured from the markers on the video recordings. The vertical was considered as zero degrees. If the position of the trunk or of the head relative to the trunk of a subject was not vertical in normal standing, the responses to the perturbations were calculated relative to the “normal position” of that subject.
The EMG signals were rectified. Further data reduction was performed by calculating the average root mean square (RMS) values of the EMG signals during 15 sec prior and 15 sec during the movement or vibration. The RMS values were normalized as a percentage of maximum voluntary contraction (MVC). To induce a MVC of the trapezius muscle the subject was asked to elevate the shoulder maximally and to hold the shoulder in that position while an examiner tried to pull the arm down as hard as possible. To induce a MVC of the latissimus dorsi muscle the subject, in prone position arms alongside, was asked to lift the arm backwards and to hold it there while an examiner tried to push the arm down.
For continuous data a repeated measures analysis of variance (ANOVA 5*2) was used, with the 5 conditions being the repeated-measures factor and the 2 groups the between-groups factor. When significant interaction effects were found post-hoc Tuckey tests were defined to identify specific differences. A Mann-Whitney U test was used for comparisons of discrete data between groups. Probability (p) values less than 0.05 were reported as statistically significant.
RESULTS
Head movement
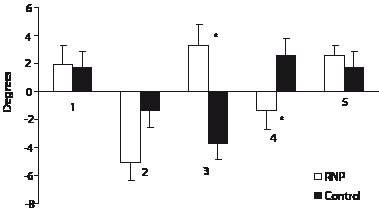
A significant interaction was found between groups and conditions for head movement (F[4,22] = 5.95, p < 0.001). During passive shoulder movements significant differences in head movement were found between subjects with RNP and control subjects. During passive elevation subjects with RNP bent their head to the ipsilateral side, whereas control subjects moved their head to the contralateral side (p = 0.004). During passive depression subjects with RNP bent their head to the contralateral side, whereas control subjects moved it to the ipsilateral side (p = 0.04) (Fig. 3). During active elevation, active depression and trapezius muscle vibration no differences were found between both groups (p > 0.05). Fig. 4 displays the movements of the head during the different conditions for subjects with RNP and control subjects.
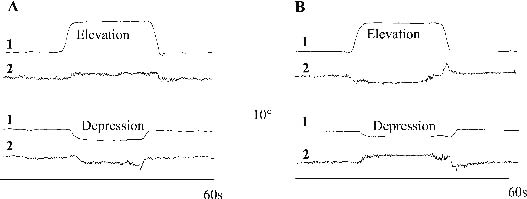
Fig. 3. Shoulder (1) and head (2) movement of (A) a representative subject with recurrent neck pain and (B) a control subject during elevation and depression of the shoulder.
Fig. 4. Head movement during: 1, active shoulder elevation; 2, active shoulder depression; 3, passive shoulder elevation; 4, passive shoulder depression; 5, trapezius muscle vibration (*p < 0.05). Negative values are movements to the left. RNP: recurrent neck pain.
Trunk movement
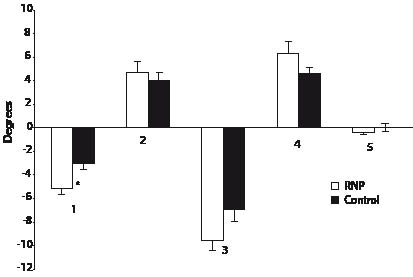
A significant interaction was found between groups and conditions for trunk movement (F[4,22] = 3.38, p < 0.02). During active and passive elevation both groups showed a movement of the trunk to the left side. Active and passive depression was associated with a movement of the trunk to the right side. During active elevation subjects with RNP made larger trunk movements than control subjects (p = 0.006). No trunk movements were found during trapezius muscle vibration. Fig. 5 shows the results of the trunk movements during the different conditions.
Fig. 5. Trunk movement during: 1, active shoulder elevation; 2, active shoulder depression; 3, passive shoulder elevation (p = 0.06); 4, passive shoulder depression; 5, trapezius muscle vibration (*p < 0.05). Negative values are movements to the left. RNP: recurrent neck pain.
Head position relative to trunk position
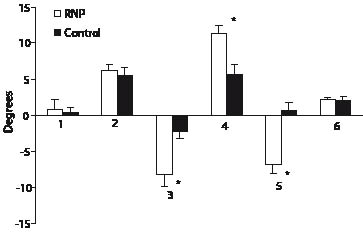
A significant interaction was found between groups and conditions for head position relative to trunk position (F[4,22] = 1.31, p < 0.001). When the position of the head was measured relative to the trunk position, subjects with RNP moved their head significantly away from the side of trunk bending during active shoulder elevation (p < 0.001), during active shoulder depression (p < 0.001), during passive shoulder elevation (p < 0.001) and during passive shoulder depression (p < 0.001). For example, depression of the right shoulder led to lateral bending of the trunk to the right side and lateral bending of the head to the left side. Control subjects only made significant movements during active and passive shoulder elevation (p = 0.004 and p = 0.002, respectively). Subjects with RNP made significantly larger movements compared with control subjects during active shoulder depression (p = 0.008), during passive shoulder elevation (p = 0.003) and during passive shoulder depression (p < 0.001). No differences in head on trunk position were found in quiet standing, nor during vibration of the trapezius muscle (Fig. 6).
Fig. 6. Movement of the head relative to the trunk during: 1, normal position; 2, active shoulder elevation; 3, active shoulder depression; 4, passive shoulder elevation; 5, passive shoulder depression; 6, trapezius muscle vibration (*p < 0.05). Negative values are movements to the left. RNP: recurrent neck pain.
Body orientation (COP)
No interaction effects were found between groups and conditions for body orientation. During active conditions no changes in COP were found. During vibration of trapezius muscle no differences in COP movements were found between the RNP group and healthy subjects. During vibration the COP moved significantly forwards in both groups, 1.6 cm (SD 1.7) (p = 0.03) in the RNP group and 1.4 cm (SD 1.5) (p = 0.04) in the control group.
Perception of shoulder position
Subjects with RNP appraised their shoulder position significantly higher during passive elevation compared with the subjects of the control group (p < 0.05). Table I shows the mean results of the measured shoulder position and of the subject’s appraisal of shoulder position.
Electromyography
No differences were found between subjects with RNP and control subjects for trapezius and latissimus dorsi EMG RMS values in the different conditions (p > 0.05). There was a significant increase in trapezius muscle activity of 25.4% (SD 13.5%) during active elevation (p < 0.001). During passive elevation there was a significant increase in 14.9% (SD 14.7) (p < 0.001). This means that there was no complete relaxation of trapezius muscle during passive elevation. During active depression there was an increase in latissimus dorsi RMS value of 9.6% (SD 6.1%) (p < 0.001).
DISCUSSION
This study examined how neck proprioceptive signals are processed and interpreted within an egocentric reference frame and whether this is altered in subjects with subclinical unilateral RNP. This was achieved by manipulating shoulder position and muscle spindle information and examining alterations in shoulder, head and trunk position and whole-body orientation. The most important finding was that subjects with RNP moved their head in a different way compared with control subjects in response to passive shoulder movements. Subjects with RNP moved their head away from the depressed shoulder and towards the elevated shoulder. The opposite movements were made by control subjects. Subjects with RNP also showed larger head and trunk movements than control subjects.
To our surprise, both groups showed head movements opposite to the direction of the shoulder movements during active shoulder movements. A possible explanation for these contralateral head movements is that subjects tried actively to correct for a postural disturbance. Preparatory postural adjustments are demonstrated with voluntary arm movements (22, 23). Anticipatory control of head posture has also been shown during head and trunk perturbations (24). The preparatory head movements seen in our study may be an adequate reaction during short, active movements. Subjects with RNP, however, also showed the contralateral head movements during passive shoulder depression. Prolonged contralateral bending of the head during passive depression of the shoulder could cause undue loading on the cervical spine structures and could be a mechanism for pain and recurrences.
Several reasons for the differences in head movements between subjects with RNP and control subjects can be hypothesized. It has been proposed that pain may affect fusimotor drive and thus the sensitivity of muscle spindles (25, 26). As there was no pain at the time of testing, this mechanism cannot explain entirely the findings in this study. Another possible cause of disturbed peripheral signals could be a higher magnitude of activity of the upper trapezius muscle, but no differences were found between subjects with RNP and control subjects. Recurrent episodes of neck pain, however, may have altered muscle spindle sensitivity in the trapezius muscle and so have made the signals less reliable (25, 26). This mechanism may also have affected other shoulder muscles and could explain why a reduced proprioceptive acuity of the shoulder has been found in persons with neck pain (3). This is also in agreement with the findings of Knox et al. (27). They found that changes in head position induced greater elbow joint position errors in patients with whiplash associated disorders, which can be explained by a reduced knowledge of head position.
The effects of receptor stimulation in the neck are also dependent on the reference system in which the signals are interpreted (5, 6). The same proprioceptive signal may cause different kinesthetic sensations depending on the choice of the reference system. The differences in head movements between subjects with RNP and control subjects during passive shoulder movements may have been caused by a different choice of reference frame possibly due to re-weighting of sensory signals. If the quality of input from a particular body location deteriorates due to injury, disease, or normal ageing, the CNS may adapt by up-weighting the input from other locations (15, 28). It is likely that the control subjects used the shoulder and scapula as a reference. This explains why they moved their head in the same direction as their shoulder. To dynamically stabilize the scapula a coordinated recruitment of the three portions of the trapezius muscle is necessary (29). As our subjects with RNP showed neck and shoulder pain (upper trapezius region), the dynamic stability of their scapula may be impaired. Therefore, subjects with RNP may have chosen to use the trunk as a reference rather than the unstable shoulder/scapula. In the current study control subjects also seemed to switch from one reference frame to another when changing from active shoulder movements to passive movements, as the direction of their head movement changed from active to passive conditions. Perhaps the tactile cues from the hands of the researcher around the arm enhances the feeling of a fixed arm and shoulder, thereby causing a switch from a head and trunk reference frame to a shoulder reference frame. This is in line with the findings of Gurfinkel & Levik (5), where a light touch made subjects switch from a trunk fixed reference frame to an arm fixed reference frame. In addition a distortion of the egocentric reference frame may have led to the larger movements observed in subjects with RNP. Further research about the choice of reference frame, however, is necessary to support this hypothesis. Further to this hypothesis, changes in reference frame and associated changes in posture and movement amplitude could put stress on cervical joints, muscles and neural tissues and may be the cause of the recurrent character of the neck pain.
During the vibration condition both groups showed an increased body sway in anterior direction. This means that control subjects as well as subjects with subclinical RNP use the proprioceptive neck muscle spindle signals in the regulation of whole-body orientation. We expected that subjects with RNP might pay less attention to signals coming from the affected side because of sensory down-weighting and therefore might have shown no increase in body sway. Sensory down-weighting was found in patients with low back pain (15). Brumagne et al. (15) showed that patients with low back pain show less body sway when spinal muscles were vibrated than healthy subjects. This is probably because they learned to ignore the less reliable afferent signals from spinal muscles and rely more on other ankle muscle for postural control.
The normal reactions during trapezius muscle vibration stimulation, seen in subjects with RNP, suggest that they have no peripheral receptor problems for the upper trapezius muscle. We could not, however, rule out the existence of receptor problems for other shoulder/neck muscles. In contrast to the normal reaction during vibration, the processing of proprioceptive signals in the CNS during active and passive conditions in subjects with RNP differed from the control subjects. During both active and passive shoulder movements subjects with RNP showed similar contralateral head movements, while control subjects showed different head movements during active vs passive shoulder movements. This indicates that subjects with RNP tend to use the same reference frame during both active and passive movements, while control subjects showed a flexibility to switch between reference frames depending on the postural condition.
In future research it might be useful to use other measures of internal representation of body orientation, such as the subjective vertical, as a measure of egocentric reference frame. It would also be useful to test the left side to evaluate whether changes in reference frame also become obvious when the non-affected side is tested.
Finally, some methodological aspects of this experiment need to be discussed. The method of evaluating the subjective shoulder position is not optimal, as the subjects have to move their left arm to show their shoulder position on the body chart. This movement elicits afferent signals, which may have influenced the choice of reference frame for interpretation of the signals coming from the right shoulder. Another shortcoming of the study is that because of logistical limitations the person conducting the test was not blinded.
A last important remark is that the groups were small and that the subjects with RNP were only mildly disabled. The differences between both groups might become more obvious when testing patients with more severe neck pain.
In conclusion, subjects with RNP showed a modified interpretation of proprioceptive signals reflected in changes in head position and in larger head and trunk movements during active and passive shoulder movements. This may be due to an offset in the egocentric reference frame or to a reduced capacity to switch reference frames according to the postural condition, rather than a peripheral receptor problem only, and could be an explanation for the recurrent character of neck pain in patients. The larger movements of the head relative to the trunk might additionally cause undue load on the joints, the muscles and the neural structures. Better insight into these mechanisms might lead to better evaluation and treatment of subjects with RNP and to a reduction in recurrent episodes.
ACKNOWLEDGEMENTS
This work was supported by grant 1.5.104.03 (Fund for Scientific Research – Flanders).
The authors thank Sue Morris for her linguistic suggestions.
REFERENCES
1. Coté P, Cassidy J, Carroll L. The Saskatchewan health and back pain survey. The prevalence of neck pain and related disability in Saskatchewan adults. Spine 1998; 23: 1689–1698.
2. Heikkila H,Wenngren B. Cervicocephalic kinesthetic sensibility, active range of cervical motion, and oculomotor function in patients with whiplash injury. Arch Phys Med Rehabil 1998; 79: 1089–1094.
3. Sandlund J, Djupsjöbacka M, Ryhed B, Hamberg J, Björklund M. Predictive and discriminative value of shoulder proprioception tests for patients with whiplash-associated disorders. J Rehabil Med 2006; 38: 44–49.
4. Knox JJ, Beilstein DJ, Charles SD, Aarseth GA, Rayer S, Treleaven J, Hodges PW. Changes in head and neck position have a greater effect on elbow joint position sense in people with whiplash-associated disorders. Clin J Pain 2006; 22: 512–518.
5. Gurfinkel V, Levik I. Reference systems and interpretation of proprioceptive signals. Hum Physiol 1998; 24: 46–55.
6. Lackner J, DiZio P. Vestibular, proprioceptive, and haptic contribution to spatial orientation. Annu Rev Psychol 2005; 56: 115–147.
7. Sainburg R, Ghez C, Kalakanis D. Intersegmental dynamics are controlled by sequential anticipatory, error correction, and postural mechanisms. J Neurophysiol 1999; 81: 1045–1056.
8. Holden M, Ventura J, Lackner J. Stabilization of posture by precision contact of the index finger. J Vestib Res 1994; 4: 285–301.
9. Lackner J, DiZio P. Aspects of body self-calibration. Trends Cogn Sci 2000; 4: 279–288.
10. Knox J, Hodges P. Changes in head and neck position affect elbow joint position sense. Exp Brain Res 2005; 165: 107–113.
11. Knox J, Coppieters M, Hodges P. Do you know where your arm is if you think your head has moved? Exp Brain Res 2006; 173: 94–101.
12. Knox J, Cordo P, Skoss R, Durrant S, Hodges P. Illusory changes in head position induced by neck muscle vibration can alter the perception of elbow position. Behav Neurosc 2006; 120: 1211–1217.
13. Ivanenko YP, Solopova IA, Levik YS. The direction of postural instability affects postural reactions to ankle muscle vibration in humans. Neurosci Lett 2000; 292: 103–106.
14. Anastasopoulos D, Nasios G, Mergner T, Maurer C. Idiopathic spasmodic torticollis is not associated with abnormal kinesthetic perception from neck proprioceptive and vestibular afferences. J Neurol 2003; 250: 546–555.
15. Brumagne S, Cordo P, Verschueren S. Proprioceptive weighting changes in persons with low back pain and elderly persons during upright standing. Neurosci Lett 2004; 366: 63–66.
16. Lee H, Nicholson L, Adams R, Bae S-S. Proprioception and rotation range sensitization associated with subclinical neck pain. Spine 2005; 30: E60–E67.
17. Gandevia S, McCloskey D. Joint sense, muscle sense and their combination as position sense, measured at the distal interphalangeal joint of the middle finger. J Physiol 1976; 260: 387–407.
18. Gandevia S. Kinesthesia: roles for afferent signals and motor commands. In: Rowell L, Shepherd J, editors. Handbook of physiology, section 12n exercise: regulation and integration of multiple systems. New York: Oxford University Press; 1996, p.128–172.
19. Burke D, Hagbarth K-E, Hofstedt L, Wallin B. The responses of human muscle spindle endings to vibration of non-contracting muscles. J Physiol (Lond) 1996; 261: 673–693.
20. Cordo P, Gurfinkel V, Bevan L, Kerr G. Proprioceptive consequences of tendon vibration during movement. J Neurophysiol 1995; 74: 1675–1688.
21. Vernon H, Mior S. The neck disability index: a study of reliability and validity. J Manipulative Physiol Ther 1991; 14: 409–415.
22. Bouisset S, Richardson J, Zattara M. Do anticipatory postural adjustments occurring in different segments of the postural chain follow the same organisational rule for different task movement velocities, independently of the inertial load value? Exp Brain Res 2000; 132: 79–86.
23. Cordo P, Nashner L. Properties of postural adjustments associated with rapid arm movements. J Neurophysiol 1982; 47: 287–302.
24. Danna-Dos-Santos A, Degani AM, Latash ML. Anticipatory control of head posture. Clin Neurophysiol 2007; 118: 1802–1814.
25. Capra N, Hisley C, Masri R. The influence of pain on masseter spindle afferent discharge. Archives of Oral Biology 2007; 52: 387–390.
26. Thunberg J, Hellström F, Sjölander P, Bergenheim M, Wenngren B, Johansson H. Influences on the fusimotor-muscle spindle system from chemosensitive nerve endings in cervical facet joints in the cat: possible implications for whiplash induced disorders. Pain 2001; 91: 15–22.
27. Knox J, Beilstein D, Charles S, Aarseth G, Rayar S, Treleaven J, Hodges P. Changes in head and neck position have a greater effect on elbow joint position sense in people with whiplash-associated disorders. Clin J Pain 2006; 22: 512–518.
28. Popa T, Bonifazi M, Della Volpe R, Rossi A, Mazzocchio R. Adaptive changes in postural strategy selection in chronic low back pain. Exp Brain Res 2007; 177: 411–418.
29. Mottram S, Woledge R, Morrissey D. Motion analysis study of a scapular orientation exercise and subjects ability to learn the exercise. Man Ther 2007; Epub ahead of print.