OBJECTIVES: Variables from a study of patients with rheumatoid arthritis were linked to the International Classification of Functioning, Disability and Health (ICF) Core Set for rheumatoid arthritis. The purpose of this analysis was to evaluate the ICF Core Sets for rheumatoid arthritis for assessing the functional outcomes of the rheumatoid hand.
DESIGN: Prospective cohort.
SUBJECTS: A total of 142 subjects with rheumatoid arthritis.
METHODS: Patients who elected to have or not have arthroplasty were linked with the ICF Core Sets. Study variables were assigned into one of the Core Set blocks that compose the ICF model. The blocks were then entered into multiple regression models to determine the contribution of each block in explaining the variation in hand outcome at enrollment, as well as the change in hand outcome after one year.
RESULTS: Seventy percent of the reported hand outcome at enrollment was explained by the ICF Core Set blocks. For change in hand outcome at one year, the ICF Core Set blocks measured at enrollment explained 18% of the variance.
CONCLUSION: The components of the ICF Core Set for rheumatoid arthritis explained much of the variation in hand functioning for patients with rheumatoid arthritis, but were not predictive of the change in hand functioning after one year.
Key words: rheumatoid arthritis; ICF; silicone metacarpophalangeal joint arthroplasty; Michigan Hand Outcomes Questionnaire; hand surgery.
J Rehabil Med 2011; 00: 00–00
Correspondence address: Kevin C. Chung, Section of Plastic Surgery, The University of Michigan Health System, 1500 E. Medical Center Drive, 2130 Taubman Center, SPC 5340, Ann Arbor, MI 48109-5340, USA. E-mail: kecchung@med.umich.edu
Submitted May 13, 2010; accepted October 29, 2010
INTRODUCTION
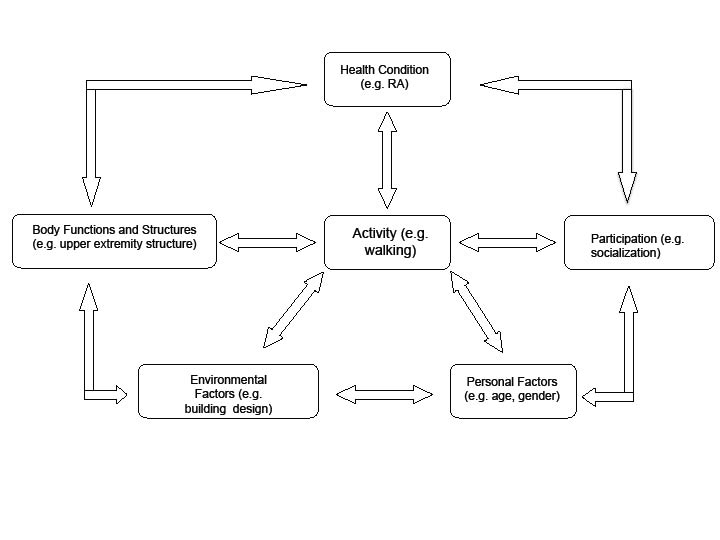
The World Health Organization (WHO) instituted a robust program to classify functioning and health with the introduction of the International Classification of Functioning, Disability and Health (ICF) in 2001 (1). In the past, the field of rehabilitation classified a patient’s functioning and disability based mainly on the medical factors associated with a diagnosis. The ICF framework of classifying function and disability is multi-dimensional and based on a “biopsychosocial model” that takes into account contextual factors that affect functioning, such as personal and environmental factors. Rather than focusing on a single part of the body, the ICF looks at the whole person and how that person functions in society. The ICF (Fig. 1) incorporates 5 major components and includes: Body structure, Body function, Personal factors, Environmental factors, and Activity/Participation. Each component is composed of categories and subcategories. For example, the body structure component includes upper extremity structure and lower extremity structure categories.
Fig. 1. The International Classification of Functioning, Disability and Health model. RA: rheumatoid arthritis.
The ICF classification system was designed to be comprehensive and includes over 1400 categories. The extensive nature of the ICF system makes it cumbersome to use for clinical or research purposes. The ICF checklist and ICF Core Sets were developed to make use of the ICF more practical. The ICF checklist is a shorter version with 125 categories, whereas the Core Sets are smaller versions of the ICF that utilize only those categories that are applicable to a specific disease or condition. The Core Sets, similar to the overall ICF, were designed through consensus and testing (2–4). An ICF Comprehensive Core Set was developed for rheumatoid arthritis (RA) and has 96 categories (5). The RA Core Set defines “what to measure” for RA outcomes, but does not define “how to measure” (6). Comparing RA Core Set categories with study measurements will determine how well a study has covered “what to measure”. Researchers working on rheumatic diseases can use the ICF framework for RA to get consensus to measure all the components of outcomes that are pertinent to these patients. Linking study measurements to the ICF Core Set will allow researchers to determine how well a study covers ICF categories, and this in turn will determine how well the study measures functioning and health of the patients (4). The ICF has been used extensively (7) and applied to several other conditions, including brain injuries (8), spine injuries (9, 10), back pain (11, 12), breast cancer (13), stroke (14, 15), and Crohn’s disease (16).
In 2004, the authors began a multi-center, international study funded by the National Institutes of Health (NIH), to assess hand outcomes after the silicone metacarpophalangeal arthroplasty (SMPA) procedure to correct deformities in rheumatoid hands. The study was designed with knowledge of the multi-factorial nature of this chronic disease. Previous studies of SMPA outcomes relied on physical measurements such as grip and pinch strength and range of motion. This study was unique because of the collaboration between rheumatologists and hand surgeons, as well as the use of patient-reported outcomes through questionnaires. Comprehensive hand outcomes were measured in the study by the Michigan Hand Outcomes Questionnaire (MHQ). The MHQ is a validated questionnaire (17, 18) that is employed internationally for the assessment of hand diseases. We have found the MHQ to have particular suitability for rheumatoid hand problems (19). In this paper, we linked study measurements to the ICF RA Core Set to determine the study’s ability to assess hand function using the MHQ as our reference standard. The results will demonstrate the utility of the ICF RA Core Set for assessment of outcomes for patients with RA.
Methods
This multi-center study is a prospective cohort of patients with RA with severe deformities of the metacarpophalangeal joint (MCPJ) resulting in ulnar subluxation and extension lag. Participants chose either to undergo SMPA (cases) or not to undergo SMPA (controls). Participants were not randomized due to strong patient preferences regarding surgery. In pilot testing prior to the start of the study, we found that most patients would not consent to randomization because they have an inherent preference whether or not to have surgery. Patients diagnosed with RA were referred by their rheumatologists to 1 of the 3 study sites: The University of Michigan (Ann Arbor, MI), Curtis National Hand Center (Baltimore, MD) and Pulvertaft Hand Centre (Derby, England). All aspects of the study were approved by the Institutional Review Boards at University of Michigan, Curtis National Hand Center and the Pulvertaft Hand Centre. Study subjects were required to be between 18 and 80 years of age, able to read English and have a defined degree of deformity at the MCPJs (the sum of the mean MCPJ ulnar drift and mean MCPJ extensor lag equal to or greater than 50). Exclusion criteria included: health problems that would prohibit surgery, extensor tendon ruptures in the study hand, swan-neck or boutonniere deformities that would require surgery, previous MCPJ replacement and the initiation of disease-modifying anti-rheumatic drugs (DMARDs) within 3 months of enrollment. Once eligibility was determined, patients were informed about the surgical procedure and post-operative therapy. Patients then decided whether or not to have surgery. Those declining surgery were able to cross over to have surgery after one year in the study.
Study assessments were completed at enrollment, 6 months, 1 year, 2 years, and 3 years. Only the data for one year outcomes was complete at the time of analysis. The primary outcome instrument for measuring hand performance in the study is the MHQ (17, 18). The MHQ is a validated hand-specific outcome questionnaire and has been applied in researching outcomes for RA (20–22). The MHQ contains 6 scales: (i) overall hand function, (ii) activities of daily living (ADL), (iii) pain, (iv) work performance, (v) aesthetics, and (vi) patient satisfaction. An MHQ summary score is calculated by averaging the 6 scales, and ranges from 0 to 100, with higher scores indicating better performance, and measures comprehensive hand outcomes.
Other study assessments included functional measurements (grip strength, pinch strength), arc of motion and degree of ulnar drift for all 4 fingers, a measurement of ADL (Jebson-Taylor test) (23, 24), overall health assessment (Arthritis Impact Measurement Scales 2 (AIMS2)) (25), and a medication assessment. Grip strength was measured in kilograms using a JAMAR dynamometer (Asimow Engineering, Los Angeles, CA, USA). Pinch strength was also measured in kilograms using a standard Preston pinch gauge (J. A. Preston, Clifton, NJ, USA). Grip and pinch strength were measured 3 times for both hands. Arc of motion and ulnar drift were measured in degrees by a trained hand therapist. The Jebson-Taylor test simulates ADL and includes 7 timed tasks: (i) writing a short sentence, (ii) turning over 7,62 × 12,7 cm cards, (iii) picking up small objects and placing them in a container, (iv) stacking checkers, (v) simulated eating, (vi) moving large, empty cans, and (vii) moving large, weighted cans. The writing component was not included in our assessments due to the difficulty with interpreting the results. The AIMS2 questionnaire has 12 scales that can be combined into 6 domains that include: physical (mobility, hand and arm function, ADL, etc.), affect (level of tension, mood) symptom (pain), social (social interaction), role (work), and comorbidity. AIMS2 scores range from 1 to 10, with lower scores indicating better health status.
One of the ICF components, body structure, was not included in the analysis. The body structure component pertains to a particular body part of interest. In this study there is only one structure of interest, the hand, and thus body structure does not vary among subjects. Using the ICF RA Core Set as a guide, we grouped study variables into 1 of the 4 remaining components. Our study was not designed using the RA Core Set, and therefore many categories that make up the components were not utilized by our study. In the analysis and in the remainder of this paper, the main ICF components will be referred to as “blocks”.
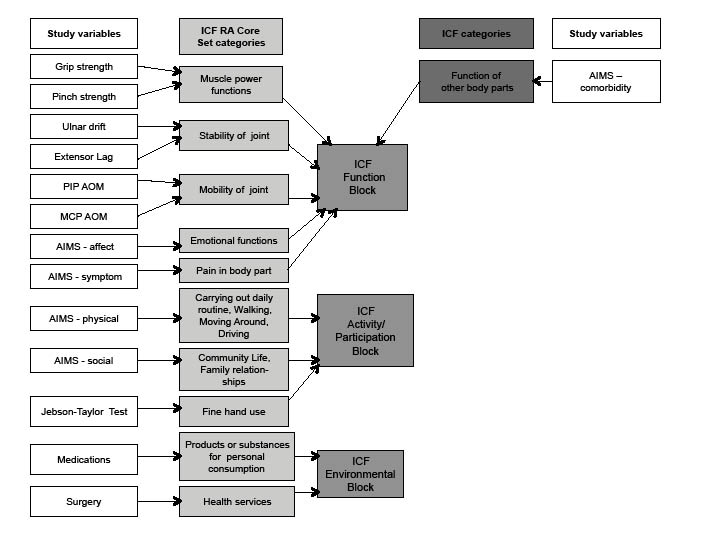
Linkage was accomplished using the methods described by Cieza et al. (26, 27). Each variable used in the analysis was linked to a specific category in the RA Core Set. If an appropriate category was not available in the Core Set, variables were linked to the full ICF. Fig. 2 shows how the individual variables were linked to ICF RA Core Set categories and then to broad ICF blocks. For example, grip and pinch strength were linked to the category muscle power function in the RA Core Set, which is part of the ICF Function block. The study variable, comorbidity was not included by the RA Core Set, therefore we linked that variable to the main ICF categories. The outcome of the linkage is summarized in Table I.
Fig. 2. Linkage of study variable to International Classification of Functioning, Disability and Health (ICF).
|
Table I. Study variables included in International Classification of Functioning, Disability and Health blocks
|
|
Personal factors
|
Environmental factors
|
Body function
|
Activity/Participation
|
|
Age
|
Medications
|
Grip strength
|
AIMS – physical
|
|
Sex
|
Surgical groupa
|
Pinch strength
|
Jebson-Taylor test
|
|
Education
|
|
Ulnar drift
|
AIMS – social
|
|
Race
|
|
Extensor lag
|
|
|
Country
|
|
PIP AOM
|
|
|
|
|
MCP AOM
|
|
|
|
|
AIMS – affect
|
|
|
|
|
AIMS – comorbidity
|
|
|
|
|
AIMS – symptom
|
|
|
aUsed in the Michigan Hand Outcomes Questionnaire change score model only.
MCP: metaearpophalangeal joint; AIMS: Arthritis Impact Measurement Scales; PIP: proximal interphalangeal joint; AOM: arc of motion.
|
The demographic variables of age, gender, level of education, race and country were included in the personal factor block. The majority of study outcome variables were linked to the body function block and included grip and key pinch strength, degree of ulnar drift and extensor lag, average proximal interphalangeal joint (PIP) arc of motion, average MCPJ arc of motion, AIMS2 affect, AIMS2 comorbidity (number of comorbid conditions), and AIMS2 symptom.
To determine how well the study measurements, after linking to the ICF RA Core Set, predicted hand outcomes, we used a multiple regression model with the MHQ summary score at baseline as the response variable. Actual study measurements were used in the regression model. The multiple regression model was constructed using hierarchical regression, a technique that manages the order of variable sets (or blocks) entered into the model. This allowed for assessment of the ICF Core Sets’ contribution to the model by examining the influence of each Core Set block in a sequential way, such that the relative importance of a block of predictors may be judged on the basis of how much it adds to the prediction, over and above that can be accounted by other important predictors. We were also able to assess the significance of individual study variable in predicting the outcome. The order of entry for blocks was determined so that the potentially confounding contextual variables are entered first in order to control for their effects. Based on this reasoning, the personal factor block was entered first, followed by the environmental factor block, body function block, and the activity/participation block. Because we were also interested in how well the ICF Core Sets measured at baseline predict change in hand outcomes, we also modeled the change in the MHQ summary score at one year as the response variable. Because some patients received the surgical treatment, the change score model also included a surgery indicator variable as a predictor in the environmental block to adjust for differences in MHQ scores due to the surgical procedure. The regression models used complete data only, thus excluding those with any missing predictor or response variables. To check the sensitivity of the model results, we repeated the analysis using multiple multivariate imputation methods to impute values for the missing predictor variables. A total of 5 imputation data-sets were created. The predictive models were then fit across the 5 imputed data-sets, resulting in a single model with parameter estimates and standard errors combined across the imputed data-sets, considering both the between- and within-imputation variance (28). Statistical analyses were conducted using SPSS version 16 (SPSS Inc., Chicago, IL, USA) and Stata version 11 (StataCorp LP, College Station, TX, USA).
Results
A total of 158 subjects enrolled in the SMPA trial, of whom 67 were surgical cases and 91 were non-surgical controls. Eight of these were considered as loss at baseline and 5 withdrew from the study. There was incomplete information for the remainder of the subjects. A total of 142 patients had MHQ data available at enrollment, and of them, 130 were included in the complete data analysis. Of the 130 subjects, 49 were surgical and 81 were non-surgical. The mean age of the subjects was 61 years; with 91% and 71% being white and female, respectively (Table II). The majority of subjects were from the USA (65%). The analysis of changes in hand outcomes required hand outcomes data from both baseline and one year, resulting in 129 patients with the MHQ data available at both times, and complete data in 111 patients. Of these 111 cases, the total number of surgical subjects was 38.
|
Table II. Overall demographics for the baseline (n = 130) and 1 year change (n = 111) in Michigan Hand Outcomes Questionnaire response
|
|
Characteristic
|
Baseline
|
1 year change
|
|
Gender, n (%)
|
|
|
|
Male
|
37 (29)
|
32 (29)
|
|
Female
|
93 (71)
|
79 (71)
|
|
Age, years, mean (median) [min–max]
|
61.0 (60.9)
[35.2–80.5]
|
60.8 (60.9) [35.2–80.5]
|
|
Race, n (%)
|
|
|
|
Non-white
|
12 (9)
|
10 (9)
|
|
White
|
118 (91)
|
101 (91)
|
|
Education, n (%)
|
|
|
|
High school or less
|
57 (44)
|
50 (45)
|
|
More than high school
|
73 (56)
|
61 (55)
|
|
Income, n (%)
|
|
|
|
< $20,000
|
|
28 (25)
|
|
$20,000–$50,000
|
|
47 (42)
|
|
> $50,000
|
|
36 (32)
|
|
Country, n (%)
|
|
|
|
USA
|
85 (65)
|
67 (60)
|
|
UK
|
45 (35)
|
44 (40)
|
|
Group, n (%)
|
|
|
|
Non-surgical group
|
81 (62)
|
73 (66)
|
|
Surgical group
|
49 (38)
|
38 (34)
|
The results of the hierarchical regression model using the baseline MHQ summary score are shown in Table III. The R2 shows the percent of variation in comprehensive hand outcomes that is explained by the blocks of variables sequentially added. For example, the personal block explains only 1.6% of the variation in hand outcomes. The addition of the environmental variables in the model increases the R2 to 2.3%, which shows that the increase in hand outcomes variation explained by the environment variables was 0.7%, controlling for personal variables. Controlling for personal and environmental variables, the body function variables explained 57.2% more of the variance in hand outcomes (p < 0.001 for change in R2), and controlling for personal, environmental and body function variables, the body activity variables explained 10.4% more of the variance in hand outcomes (p < 0.001). The results indicate that the ICF component groups explain 70% of comprehensive hand outcomes reported by patients enrolled in the study, with the majority explained by the ICF body function component.
Table III also shows the effect of individual variables that constitute the blocks for all patients. Significant predictors of hand outcomes at baseline included: country – USA (B = –8.1, p = 0.006), AIMS – physical (B = –4.4, p < 0.001), and AIMS – symptom (B = –1.8, p = 0.004). The results show that, US-based patients, poor physical function, and greater pain (AIMS – symptom) are each associated with worse hand outcomes at baseline. Although the body function block was significant, we did not find any of the individual traditional physical measures in the body function block to be significantly predictive of hand outcomes.
|
Table III. Results of hierarchical regression model for baseline Michigan Hand Outcomes Questionnaire summary score using International Classification of Functioning, Disability and Health Core Set blocks (n = 130)
|
|
Blocks
|
Variable
|
Coefficient
|
SE
|
p-value
|
R2 (R2 change, p-valuea)
|
|
|
Constant
|
75.921
|
12.533
|
< 0.001
|
|
|
1
|
Race: whiteb
|
–7.154
|
3.870
|
0.067
|
0.016 (0.016, 0.850)
|
|
|
Education: more than high schoolb
|
–0.231
|
2.427
|
0.924
|
|
|
|
Gender: maleb
|
–0.856
|
3.025
|
0.778
|
|
|
|
Age
|
–0.069
|
0.129
|
0.593
|
|
|
|
Country: USAb
|
–8.111
|
2.873
|
0.006
|
|
|
2
|
Medication: DMARDSb
|
0.255
|
2.430
|
0.917
|
0.023 (0.007, 0.630)
|
|
|
Medication: anti-inflammatoriesb
|
–6.703
|
4.662
|
0.153
|
|
|
3
|
Grip strength
|
0.204
|
0.259
|
0.433
|
0.595 (0.572, < 0.001)
|
|
|
Key pinch strength
|
0.337
|
0.849
|
0.693
|
|
|
|
Mean degree of ulnar drift
|
0.066
|
0.084
|
0.437
|
|
|
|
Mean degree of extensor lag
|
0.031
|
0.093
|
0.738
|
|
|
|
Mean arc of motion – MCP joint
|
0.156
|
0.108
|
0.152
|
|
|
|
Mean arc of motion – PIP joint
|
0.083
|
0.058
|
0.152
|
|
|
|
AIMS – Affect
|
–1.317
|
0.816
|
0.110
|
|
|
|
AIMS – Symptom
|
–1.770
|
0.598
|
0.004
|
|
|
|
AIMS – Comorbidity
|
–0.754
|
1.101
|
0.495
|
|
|
4
|
AIMS – Social
|
0.029
|
0.762
|
0.969
|
0.700 (0.104, < 0.001)
|
|
|
AIMS – Physical
|
–4.370
|
0.808
|
< 0.001
|
|
|
|
Jebson-Taylor score
|
–0.024
|
0.084
|
0.779
|
|
|
aTesting for significant change in R2.
bThe reference categories are as follows: for Race – other non-white; for Education – high school or less; for Gender – female; for Country – UK; for Medication – biologics.
SE: standard error; DMARDS: disease-modifying anti-rheumatic drugs; AIMS: Arthritis Impact Measurement Scales; MCP: metacarpophalangeal; PIP: proximal interphalangeal.
|
The hierarchical model results for the one-year change in MHQ summary score are shown in Table IV. The ICF blocks explain 45% of the variation in the one-year change in MHQ summary scores. Among the individual variables, only the surgical group indicator was a significant predictor of change in hand outcomes at one year (B = 23.52, p < 0.001), indicating that surgical patients on average experience a 23.5-point higher MHQ score at 12 months compared with their non-surgical counterparts. The fact that none of the other predictors were significant suggests that the complexity of the rheumatoid hand condition makes prediction of one year outcomes highly challenging, despite this detailed assessment of a comprehensive list of factors within the ICF framework.
|
Table IV. Results of hierarchical regression model for 1-year change in Michigan Hand Outcomes Questionnaire summary score using International Classification of Functioning, Disability and Health Core Set blocks measured at baseline (n = 111)
|
|
Blocks
|
Variable
|
Coefficient
|
SE
|
p-value
|
R2 (R2 change, p-valuea)
|
|
|
Constant
|
–0.497
|
18.117
|
0.978
|
|
|
1
|
Race: whiteb
|
–1.540
|
5.391
|
0.776
|
0.062 (0.062, 0.232)
|
|
|
Education: more than high schoolb
|
–1.242
|
3.450
|
0.720
|
|
|
|
Gender: maleb
|
–0.570
|
4.239
|
0.893
|
|
|
|
Age
|
–0.177
|
0.193
|
0.360
|
|
|
|
Country: USAb
|
5.663
|
3.972
|
0.157
|
|
|
2
|
Medication: DMARDSb
|
1.289
|
3.334
|
0.700
|
0.399 (0.336, < 0.001)
|
|
|
Medication: anti-inflammatoriesb
|
1.736
|
6.727
|
0.797
|
|
|
|
Surgical group
|
23.520
|
3.522
|
< 0.001
|
|
|
3
|
Grip strength
|
–0.347
|
0.345
|
0.317
|
0.432 (0.033, 0.797)
|
|
|
Key pinch strength
|
1.022
|
1.162
|
0.381
|
|
|
|
Mean degree of ulnar drift
|
0.027
|
0.115
|
0.813
|
|
|
|
Mean degree of extensor lag
|
0.126
|
0.123
|
0.310
|
|
|
|
Mean arc of motion – MCP joint
|
0.210
|
0.148
|
0.161
|
|
|
|
Mean arc of motion – PIP joint
|
–0.060
|
0.083
|
0.469
|
|
|
|
AIMS – Affect
|
–0.647
|
1.120
|
0.565
|
|
|
|
AIMS – Symptom
|
–0.470
|
0.846
|
0.580
|
|
|
|
AIMS – Comorbidity
|
0.943
|
1.604
|
0.558
|
|
|
4
|
AIMS – Social
|
–1.277
|
1.083
|
0.241
|
0.450 (0.019, 0.392)
|
|
|
AIMS – Physical
|
1.246
|
1.096
|
0.259
|
|
|
|
Jebson-Taylor score
|
0.069
|
0.149
|
0.644
|
|
|
aTesting for significant change in R2.
bThe reference categories are as follows: for Race – other non-white; for Education – high school or less; for Gender – female; for Country – UK; for Medication – biologics; for Surgical group – non-surgical group.
SE: standard error; DMARDS: disease-modifying anti-rheumatic drug; AIMS: Arthritis Impact Measurement Scales; MCP: metacarpophalangeal; PIP: proximal interphalangeal.
|
For both models, collinearity diagnostics revealed a high correlation between extensor lag and arc of motion (r = –0.8). The models were rerun without one of the variables, resulting in a negligible change to the model R2 (range 0.001–0.011), regardless of the variable excluded. For example, change in R2 was 0.007 when arc of motion was excluded from the baseline MHQ model and 0.012 when arc of motion was excluded from the one-year MHQ model. Furthermore, the exclusion of the variables resulted in no change in the predictors that were found to be significant in the model.
The magnitude of the changes in R2 and significance of the individual predictors from the analysis of imputed data in which missing predictors were imputed were similar to those for the analysis of complete data. For the baseline MHQ model using imputed data (n = 142), the same predictors that were significant in the model based on complete data (n = 130) were found to be significant in predicting the baseline MHQ scores. For the analysis of change score in MHQ at one year based on imputed data (n = 125), only the surgical group indicator was found to be a significant predictor of the change score, as was the case in the analysis of complete data (n = 111). Similarly, the changes in R2 from the imputed data-set model of one-year change in MHQ were consistent with those from the complete data model.
Discussion
The results presented in this paper have demonstrated the value of the ICF Core Set for RA when assessing hand outcomes in patients with RA. Linking our study measurements to the Core Set gave us an indication of how well our study measurements assess hand function. We found that the ICF blocks explained 70% of the variation in hand outcomes for all patients with RA enrolled in the study. This result indicates that the variables included in our study do a good job of covering “what to measure.” Our analysis of ICF blocks indicated which measurements were better predictors of function. The analysis highlighted the importance of Body function and Activity/Participation factors when evaluating functioning and disability in patients with RA. In particular, pain (Body function) and physical functioning (Activity/Participation) were significant predictors of poor hand function.
Personal factors, such as age and education, have been found in other studies to be predictors of outcomes for physical functioning in patients with RA (29–33). These studies measured overall physical functioning in patients with RA using health-related questionnaires. Although personal factors were included in this study, country of origin was the only personal factor identified as a predictor of hand function at baseline. Subjects from the USA reported worse hand function at baseline compared with subjects from the UK. In the USA, because of the insurance structure, it is possible that patients with severe deformity and more disability are being seen by hand surgeons. This may be especially true for younger patients. The lack of universal health insurance may deter patients from consulting physicians until it becomes absolutely necessary
The ICF was less effective in assessing changes in hand function for patients with RA over time in this study. Only 45% of the variation in the one-year change in MHQ score was explained by the ICF groups for all patients. Unlike the baseline analysis, most of the variation at one year after surgery was explained by the ICF environment, specifically, the surgical group indicator. This finding is consistent with a previous analysis of SMPA data, which found improvement in hand function at one year for surgical patients, but non-surgical patients report no change (34). Additionally, because patients with RA with hand problems may have multiple other problems in their hands and in other parts of their body that can affect their functioning, more variables may need to be considered and collected in order to identify other predictors.
A limitation of our study was that it was not designed using ICF Core Set categories. For this reason, many Core Set categories were not included as study variables. This lack of coverage of some categories probably explains much of the variance in hand function that is not explained by our model. Another limitation is that the ICF provides a detailed list of “what to measure”, but translating the list into “how to measure” is not always straightforward. For example, sensation of pain is a category for the body function component, but there is no specified way to assess pain.
Our results demonstrate the importance of using the ICF RA Core Set when designing a comprehensive study assessing outcomes of patients with RA. Outcome studies of the RA hand have traditionally relied on physical measures, such as grip and pinch strength, to measure patient function following treatment. The results show the importance of patient-reported outcomes, such as pain and the ability to perform ADL, when assessing hand function in patients with RA. The ICF framework has given us a better understanding of how multiple factors should be considered when assessing function and disability.
AcknowledgEments
The authors acknowledge the assistance of the following participants in the SMPA Study Group: Sandra V. Kotsis, MPH, (University of Michigan), Lorraine A. Zellers, CRC (Curtis National Hand Center), Mary J. Bradley, MSc (Pulvertaft Hand Centre) and the referring rheumatologists in Michigan, Derby and Baltimore. The authors also greatly appreciate the assistance of Jeanne M. Riggs, OTR, CHT, Kurt Hiser, OTR, Carole Dodge, OTR, CHT, Jennifer Stowers, OTR, CHT, Cheryl Showerman, OTR, (University of Michigan), Jo Holmes, OTR, Victoria Jansen, PT and Helen Dear, OTR (Pulvertaft Hand Centre) in taking measurements for the study patients.
Supported in part by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (R01 AR047328) and a Midcareer Investigator Award in Patient-Oriented Research (K24 AR053120) (to Dr Kevin C. Chung).
References