OBJECTIVE: To assess the effect of arm motor impairment on actual arm use in the early post-stroke period and explore its association with self-care dependency.
SUBJECTS: Thirty-one patients recruited within the 30 first days after stroke.
METHODS: Motor impairment of the upper extremity was measured with Fugl-Meyer Motor Assessment (FMA) and arm use was measured with accelerometry. Arm movement ratio (AMR), the ratio of arm use duration between the more and less affected arm, was calculated. Self-care dependency was defined as needing personal assistance in primary self-care activities.
RESULTS: FMA of the more affected arm was strongly associated with AMR (Spearman’s correlation coefficient –0.851, p > 0.001), although some patients deviated considerably from the regression line. Covariates did not have any influence on this relation. Both arm motor function and actual arm use related to self-care dependency, but were no longer significant when we controlled for lower extremity motor function.
CONCLUSION: FMA and AMR correlated highly in the early post-stroke period. These measures relate to different dimensions of the International Classification of Functioning and Health, and could be supplementary measures to reveal non-use of the affected arm. Arm use and arm impairment were not significantly associated with self-care dependency in our sample.
Key words: stroke; accelerometer; motor impairment; ICF.
J Rehabil Med 2011; 43: 299–304
Correspondence address: Gyrd Thrane, University of Tromsø, Faculty of Health Sciences, Department of Health and Care Sciences, NO-9037 Tromsø, Norway. E-mail: gyrd.thrane@uit.no
Submitted August 27, 2010; accepted December 3, 2010
Introduction
Stroke commonly leads to impaired muscle function and is a leading cause of physical disability (1). One-fifth of stroke survivors do not regain functional activity in both arms, and half of patients with initial severe paresis do not gain any important function of the more affected arm (2). However, after hemiplegic stroke, actual use of the affected arm is reported to be less than potential use, and it is suggested that learned non-use of the paretic arm reduces the level of functioning (3, 4). It is unclear whether the discrepancy between arm use and motor impairment is present only in the chronic stages after stroke or appears in the early post-stroke period.
To assess the function properly, it is crucial to analyse different aspects of arm mobility within a conceptual framework. The International Classification of Functioning and Health (ICF) provides a multidimensional framework for health and disability suited for the classification of outcome instruments (5). Motor function (bodily function/impairment), arm use (mobility-activity), and dependency (activity) represent different dimensions of the ICF (6). Suitable outcome measures for these dimensions have been recommended in subjects with stroke (4, 7). Motor impairment, as measured in the clinic or laboratory, normally represents the patient’s maximum motor ability and is considered to be different from the functional activity outside the testing situation (8). Although earlier reports have outlined a gap between measures of arm motor impairment and actual real-world arm use, these investigations have been restricted to the chronic post-stroke period (9). A study of Lang et al. (10) on early post-stroke patients found a relationship between motor impairment and duration of more affected arm movement. A possible more sensitive parameter to describe the actual use of the more affected arm is the arm movement ratio (AMR), which is less influenced by general activity (11), but so far, this has not been investigated in the early phases after stroke.
The Fugl-Meyer upper extremity motor assessment (FMA) (12) is a recommended disease-specific impairment index (13). The actual use of the impaired arm can be estimated from patient interviews; however, these may be affected by recall bias, subjectivity, aphasia, and cognitive impairment (3). Most interview methods that have been validated for arm use in home activities, however, are not suitable for inpatient measurement (14). An alternative is to use wrist-worn accelerometers, which may provide an accurate, reliable, and stable measure of the duration of arm movement in real life, outside the examination room in a hospital setting (10,11).
The aims of the study were therefore: (i) to investigate the correlations between arm motor impairment (FMA) and real-world arm use measured with accelerometry, and reveal if there are indications of learned non-use of the affected arm in early post-stroke patients; (ii) to analyse whether AMR is more or less correlated with impairment than pure duration of arm use; and (iii) to assess the influence of arm motor impairment and arm activity/real use on dependency in self-care activities.
Methods
Design
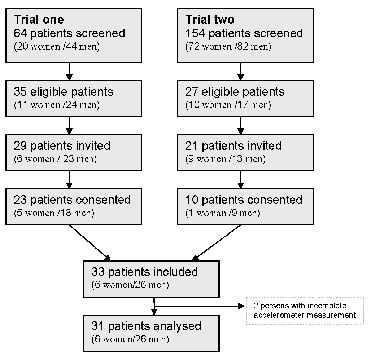
In this cross-sectional study, subjects were recruited from two trials from the stroke units at the University Hospital of North Norway. Fig. 1 illustrates the recruitment process. Patients from trial 1 were recruited between January and July 2008 and included stroke patients (first or second stroke) older than 18 years old who were medically stable and expected to survive for 1 year without other neurological diseases. Trial 2 was a constraint-induced movement therapy intervention trial. Patients were recruited from October 2008 to November 2009 with the following inclusion criteria: (i) first or second stroke, without motor upper extremity motor deficit after the first stroke; (ii) Modified Ranking Scale (mRS) of 0–2 prior to admission; (iii) age over 18 years; (iv) persistent unilateral paresis of one upper extremity; (v) extension movement in wrist or fingers, (vi) ability to follow a two-step command and a Mini Mental State score above 20. Patients were excluded if they were not able to consent, their mRS was 5 or 6, or they had substantial hemispatial neglect, short life-expectancy, other neurological impairment or injury of the affected arm. Among the 218 patients screened for this study 62 satisfied the criteria for inclusion. Twelve of these were not invited, mainly because they were discharged from the hospital before the trial coordinator was able to contact them. Thirty-three patients were included, of whom 31 had complete measurements and were analysed for the study. Subjects were examined between day 1 and day 30 post-stroke. The mean time since stroke onset was 10.6 days (standard deviation (SD) = 6.0). The study was reviewed and approved by the Regional Committee of Medical Ethics and the Commission of Privacy Rights at the University Hospital of North Norway.
Fig. 1. Study population.
Measurement
Arm motor function/impairment. The FMA of the hemiplegic side was used to measure arm motor impairment after stroke. The FMA is a 226-point multi-item Likert-type scale developed as an evaluative measure of recovery from hemiplegic stroke (12). Each item is scored on a 1–3-point ordinal scale (0 = cannot perform, 1 = performs partially, 2 = performs fully). The upper extremity motor part includes 33 items measuring movement, coordination, and reflex action of the shoulder, elbow, forearm, wrist, and hand, with a possible score ranging from 0 to 66. A trained physical therapist administered the test. Previous studies have reported excellent test-retest (r = 0.99) and inter-rater reliability (r = 0.89–0.95/intra class correlation (ICC) = 0.97) for this measurement (13). Validity to activities of daily living (ADL) capacity, Barthel Index, and other upper extremity motor tests have been demonstrated (13).
Activity/disability. Arm use/arm activity outside the examination room was measured using ActiGraph GT1M Accelerometers (ActiGraph Inc., Pensacola, USA). The patients were asked to wear one accelerometer on each arm for 24 h. Accelerometer measurements were taken within 24 h before or after the FMA examination. The GT1M is a uniaxial accelerometer that counts acceleration over a given period called an epoch. One count is equal to 0.004 G/s (15). To calculate the duration of arm movement, we used the transformation technique described by Uswatte et al. (11). Epoch time was set to 2 s. When a summarized epoch count was 1 or more, the period was defined as “in movement”. If the count was zero, the period was defined as “not in movement”. Epoch defined as ‘in movement’ was then summed and converted to seconds to represent the duration of movement of the arm measured. Participants kept a diary, in which they noted when they removed their accelerometers, when they were travelling by motor vehicle, and when they were sleeping. Periods of sleeping and car driving are not included in the analyses. The AMR, which is the ratio between the duration of movement in the least affected and the more affected arm, was used as measurement of the actual arm use. Several investigations validated this method in chronic phases after stroke and in subacute phases 3–9 months after stroke (4, 11, 16).
Personal (self-care) activities of daily living were measured with the Sunnaas ADL-index. (17). Subjects were considered dependent if they reported dependency on at least 1 of the 8 items: eating, continence, indoor mobility, toilet management, transfer, dressing/undressing, personal care, or preparation of food (18).
Covariates. Age, gender, time since stroke, stroke severity, hemi-neglect, sensory deficit, apraxia, and 5 Times Sit to Stand Test (5STS) was used as covariates. National Institute of Health Stroke scale (NIHSS) (19) was used to score stroke severity at admission. The 11-item version of Lyden et al.’s (20) measure was used. The NIHSS was scored retrospectively from the patient’s medical record based on Williams et al.’s algorithm (21). Information about initial sensory deficit and initial hemi-neglect was drawn from the admission NIHSS score. The examiner classified apraxia after examination based on the patients’ ability to understand instructions for the test battery. The 5STS was used for examining functions of the lower extremities. The patients were asked to move from the sitting to the standing position 5 times, and the time to complete the task was recorded by a stopwatch. The test has not been validated in a stroke population, but is a valid and cost-effective tool for quantifying lower extremity function in healthy adult populations. The validity of the 5STS test is supported by the correlation to sit to stand performance along with other relevant measures as knee extension strength and gait performance (22). The reliability has been reported adequate (ICC 0.67–0.94) (23–25). The test has an obvious floor effect because some patients are not able to perform the test. Quartile groups are therefore used for analyses.
Statistical analyses
SPSS for Windows release 15.0.1 (SPSS Inc., Chicago, IL, USA) was used for statistical analyses. Histograms were used to explore the distribution of all variables and Kolmogorov-Smirnov test for normality was used to decide whether the variables were normally distributed. In addition, the skewness (g) of the sample was calculated. Means and SDs were used to describe normally distributed variables. Median, range, and interquartile ranges (IQRs) were used to describe other variables. For descriptive purposes, continuous variables were categorized and cross-tabled with the dependent variable. The association between AMR and continuous variables were analysed using scatter plots and Spearman’s correlation coefficient (rho). A correlation below 0.20 was considered poor, 0.21–0.40 fair, 0.41–0.60 moderate, 0.61–0.80 good, and above 0.81 very good (26). Box-plots were used to examine the associations with categorical variables. Criteria for regression analyses were examined by visually inspecting the histograms and scatter plots of the variables and residuals. Possible correlations among the cofactors and FMA, duration of more affected arm movement, and the AMR were investigated. A regression model of two variables was used to control for the influence of covariates, showing a univariate association with the AMR. The association between FMA/AMR and dependency was analysed by logistic regression. Due to the low number of participants only two variable logistic models was used to control for the influence of covariates on the associations of dependency with FMA and AMR. A p-value below 0.05 was considered statistically significant.
Results
The age of the 31 participants ranged from 35 to 86 years. Women were older 75.5 (SD = 9.5) years compared with men 62.7 (SD 13.6) (p = 0.031). The age and gender distribution of patients did not differ significantly from screened or eligible cases. The admission NIHSS ranged from zero to 20, with a median of 3. Fourteen persons experienced sensitivity loss at admission. Table I summarizes the characteristics of the subjects.
| Table I. Characteristics of the 31 patients after stroke included in the study |
| Characteristics | |
| Age, years, mean (SD) | 65 (14) |
| Gender – females, n (%) | 6 (19) |
| First stroke, n (%) | 29 (94) |
| Diagnosis of haemorrhage, n (%) | 3 (10) |
| Left-sided lesion, n (%) | 15 (48) |
| Right-sided lesion, n (%) | 16 (52) |
| Bilateral lesion, n (%) | 1 (3) |
| Days post-stroke, mean (SD) | 10.6 (6.0) |
| Admission NIHSS, median (interquartile range) | 3 (1–6) |
| Sensory disorder present, n (%) | 14 (45) |
| Hemineglect present, n (%) | 5 (16) |
| Apraxia present, n (%) | 1 (3) |
| FMA score, median (interquartile range) | 50 (40–60) |
| Accelerometer AMR (interquartile range) | 1.5 (1.1–2.0) |
| 5STS, median (interquartile range) | 18 (11–25) |
| Participants dependent in personal ADL, n (%) | 12 (39) |
| SD: standard deviation; NIHSS: National Institute of Health Stroke Scale; FMA: Fugl-Meyer Motor Assessment upper extremity; AMR: arm movement ratio; 5STS: 5 Times Sit to Stand Test. |
The FMA of the affected upper extremity ranged from 4 to 66. The distribution was skewed toward higher values (g = –1.2, SD 0.4). Accelerometer recordings showed that the less affected arm movement ranged from 1.6 to 8.7 h, with a mean of 4.5 h (SD 1.7). The more affected arm movement ranged from 0.8 to 8.0 h, with a mean 3.0 (SD 1.7). The more/less affected AMRs ranged from 0.9 to 4.7. The distribution was skewed towards the lower values (g = 1.68, SD 0.42), with a median of 1.5. Four persons were not able to complete the 5STS test. The results from those who completed ranged from 7 to 38 s. We found a significant correlation of the 5STS with FMA (rho = –0.529, p = 0.002), movement of the more affected arm (rho = –0.627, p < 0.001), and the AMR (rho = –0.643, p < 0.001). No other statistical significant relations appeared, but the relation with NIHSS score at admission had a near significant p-value of 0.06.
Association between arm motor impairment and actual arm use
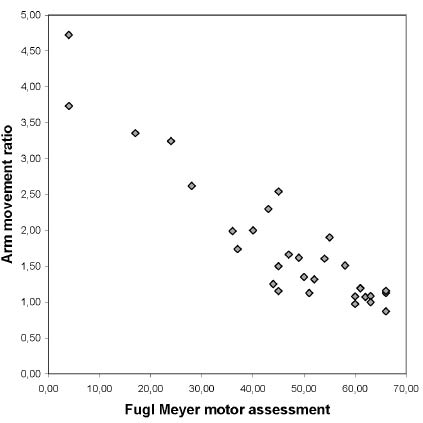
The correlation rho between arm motor impairment assessed with the FMA of the more affected upper extremity and the duration of the more affected arm movement measured with the accelerometer was 0.601 (p < 0.001). Fig. 2 shows the scatter-plot illustrating the relationship between the FMA of the affected arm and the AMR. The correlation between AMR and FMA was rho = –0.851 (p < 0.001). Although there was a very good correlation between FMA and AMR, Fig. 2 illustrates an important variation in AMR, especially in the upper range of the FMA. In the FMA range of 43–47, the AMR varied from 1.2, which is normal, to 2.5, which indicates 2.5 times greater movement of the less-affected arm. The association between FMA and AMR met the assumptions for regression analyses (linearity, homoscedasticity and normally distributed residuals). The regression analyses supported the statistically significant relationship between the FMA and AMR (β = –0.05, p < 0.001). None of the cofactors (age, gender, days since stroke, initial stroke severity, initial sensory deficit, initial hemi-neglect, apraxia, and lower extremity function) interfered with this relationship in the two-factor regression models.
Fig. 2. Scatter-plot showing the relation between the Fugl-Meyer upper extremity motor assessment and the arm movement ratio between the more and less affected arm, in 31 subjects.
Motor function and dependence in self-care activities
Overall, 17 subjects were independent in self-care activities, i.e. they did not use aids. Two were independent but used aids, and 12 were dependent on personal assistance. During analyses, the two independent outcomes were noted in the same group. The FMA scores of independent subjects ranged from 43 to 66, with a median of 58, while FMA scores of the dependent persons ranged from 4 to 63, with a median of 36.5 (p = 0.001, Mann–Whitney 2-sample test). Median AMRs were lower in the independent group (median 1.2, range 0.9–2.5) than in the dependent group (median 2.0, range 1.0–4.7) (p = 0.001 with Mann–Whitney 2-sample test). Table II categorizes FMA, AMR, and the covariates in relation to being self-care dependent. As seen, dependency was significantly associated with the scores on the FMA and AMR. Being dependent in self-care activities was also closely related to lower extremity function, as indicated by the 5STS test. Tables III and IV show the results obtained from introducing one covariate at the time into the Dependency/FMA and Dependency/AMR models. When the odds ratio (OR) was corrected for the 5STS, the relationship between dependency and FMA or AMR was no longer significant.
| Table II. The influence of Fugl-Meyer upper extremity motor assessment (FMA), arm movement ratio (AMR) and covariates on dependency in 31 post-stroke patients. Continuous variables are categorized in the third column for better visualization of the results. Odds ratios in fourth column are calculated by logistic regressions, were FMA, AMR, age and National Institute of Health Stroke Scale (NIHSS) are treated as continues variables |
| | n | Dependent n (%) | Dependent OR (95% CI) |
| FMA 0–39 40–49 50–60 61–66 | 7 8 8 8 | 7 (100) 3 (38) 1 (13) 1 (13) | 0.86 (0.77–0.96) (p = 0.009) |
| AMR 0.00–1.13 1.13–1.51 1.51–2.00 > 2.00 | 8 8 8 7 | 1 (13) 1 (13) 5 (63) 5 (71) | 8,34 (1.55–44.80) (p = 0.013) |
| Age, years 35–60 61–70 > 70 | 10 10 11 | 4 (40) 2 (20) 6 (55) | 1.03 (0.98–1.093) (p = 0.262) |
| Gender Male Female | 25 6 | 8 (32) 4 (66) | 4.25 (0.39–28.25) (p = 0.134) |
| Admission NIHSS 0–1 2–3 4–6 7–20 | 8 8 8 7 | 2 (25) 1 (13) 6 (75) 3 (43) | 1.27 (0.94–1.35) (p = 0.191) |
| NIHSS Arm Motor > 0 No Yes | 16 15 | 5 (31) 7 (47) | 1.95 (0.45–8.33) (p = 0.381) |
| NIHSS Sensory > 0 No Yes | 17 14 | 7 (41) 5 (36) | 0.79 (0.19–3.41) (p = 0.756) |
| NIHSS Neglect > 0 No Yes | 26 5 | 10 (39) 2 (40) | 1.07 (0.15–7.54) (p = 0.948) |
| 5 Times Sit to Stand 0–11 12–17 18–24 > 25 | 8 7 8 8 | 0 (00) 0 (00) 5 (63) 7 (88) | 15.13 (2.15–106.14) (p = 0.006) |
| OR: odds ratio; CI: confidence interval. |
| Table III. Adjusted odds ratio for the association between Fugl-Meyer upper extremity motor assessment (FMA) and dependency in self-care corrected for covariates in a 2 variable model in 31 post-stroke patients |
| Model | OR | p-value | 95% CI |
| FMA | 0.86 | 0.009 | 0.77–9.96 |
| FMA Age | 0.84 1.09 | 0.011 0.067 | 0.72–0.96 0.99–1.19 |
| FMA Gender – woman | 0.87 1.91 | 0.018 0.630 | 0.77–0.98 0.14–26.85 |
| FMA Days post-stroke | 0.87 1.057 | 0.011 0.522 | 0.77–0.97 0.89–1.25 |
| FMA Admission NIHSS | 0.86 0.99 | 0.009 0.959 | 0.77–0.96 0.78–1.26 |
| FMA Arm Motor > 0 | 0.86 0.71 | 0.008 0.733 | 0.77–0.96 0.10–5.20 |
| FMA Sensory > 0 | 0.86 0.53 | 0.009 0.627 | 0.77–0.96 0.12–6.28 |
| FMA Neglect > 0 | 0.86 0.52 | 0.009 0.627 | 0.77–0.96 0.39–7.09 |
| FMA 5STS (quartiles) | 0.89 9.22 | 0.104 0.035 | 0.78–1.02 1.18–72.29 |
| NIHSS: National Institute of Health Stroke Scale; 5STS: 5 Times Sit to Stand Test; OR: odds ratio; CI: confidence interval. |
| Table IV. Adjusted odds ratio (OR) for the relationship between arm movement ratio (AMR) and dependency in 31 post-stroke patients |
| Model | OR | p-value | 95% CI |
| AMR | 8.34 | 0.013 | 1.55–44.80 |
| AMR Age | 9.99 1.01 | 0.009 0.096 | 1.76–56.77 0.99–1.16 |
| AMR Gender – woman | 6.88 2.54 | 0.022 0.414 | 1.32–35.72 0.27–23.76 |
| AMR Days post-stroke | 8.66 1.09 | 0.016 0.225 | 1.50–50.23 0.94–1.26 |
| AMR Admission NIHSS | 8.37 1.00 | 0.015 0.976 | 1.52–45.98 0.77–1.28 |
| AMR Admission NIHSS Arm motor > 0 | 8.60 0.77 | 0.012 0.784 | 1.60–46.32 0.12–4.93 |
| AMR Admission NIHSS Sensory > 0 | 8.28 0.69 | 0.013 0.666 | 1.56–44.00 0.11–4.19 |
| AMR Admission NIHSS Neglect > 0 | 8.98 0.41 | 0.014 0.545 | 1.57–51.29 0.02–7.36 |
| AMR 5 Sit to Stand | 5.16 10.80 | 0.165 0.019 | 0.51–52.39 1.48–79.01 |
| NIHSS: National Institute of Health Stroke Scale; OR: odds ratio; CI: confidence interval. |
Discussion
The main finding of this study was a high correlation between motor impairment and the patient’s actual use of the more affected arm. The correlation was stronger between the FMA and calculated AMR than between the FMA and raw accelerometer data measuring duration of the movement. The finding verifies the importance of correcting for the general movement when measuring arm movement with accelerometry. Considering that these two measurements (FMA and AMR) measure different concepts of arm mobility and relate to different aspects in the ICF, the association is remarkably good. Further findings are that the scores on the FMA and the accelerometer values are both related to dependency in self-care activities, and that general mobility seems to be more important for self-care independence.
Lang et al. (10) studied the correlation between the use of the affected upper extremity and Action Research Arm Test (rho = 0.40), Wolf Motor Function Test (rho = –0.65), and some arm-activity aspects of the Functional Independence Measure (rho=0.67). The correlation between FMA and AMR in our study is larger (rho = 0.85). There may be several explanations for this difference. First, the FMA is different from the measures in Lang’s study (10). Wolf Motor Function Test and Action Research Arm Test are considered measures of arm motor function or laboratory measurements of activity (27). The FMA is typically described as a motor impairment measure, with items that are intended to assess recovery within the context of the motor system. Functional tasks are not incorporated into the evaluation (28). Several authors recommend the instrument as the primary choice for assessing motor impairment in subjects with stroke (5,13) and our findings support these recommendations. In addition, we used the calculated AMR from the accelerometer measures, correlating it with activity in the less-affected arm or the patient’s level of activity. The correlation between more affected arm movement duration and FMA in our study was rho = 0.601, which is closer to the correlations measured by Lang et al. (10), mentioned above. Based on these findings, we agree with Uswatte et al. (4) and recommend the use of AMR in arm accelerometer studies.
The association between FMA and AMR was not influenced by the cofactors introduced in our models. It is possible that other cofactors or a higher number of participants could reveal other confounding sources that might affect this association. In our study, we have controlled only for initial stroke severity. Neurological impairment at the time of measurement (NIHSS) could have explained more of the variation in both FMA and AMR. As demonstrated, there were variations in actual use of the affected arm, measured as AMR, in subjects with the same level of motor function, assessed with FMA. Concordance to hand dominance could have explained some of the variation in the AMR. Unfortunately, incomplete data on hand dominance prevented us from performing such analyses.
In accordance with Viitanen et al. (29), we found that the degree of motor impairment and arm mobility influenced the ability to complete personal self-care activities of daily living. The high odds ratio (OR) of the AMR has to be interpreted with the caution, as it is calculated based on one unit of the AMR scale. Apparently small changes in the value represent considerable changes in arm function. A value of 2 on the AMR means that the less-affected arm is used twice as much as the affected, while a value of 1 reflects equal use. It is also necessary to consider the low power of all logistic regression analyses due to a small material when interpreting the results. Dependency was measured for activities such as eating, continence, indoor mobility, toileting, dressing/undressing, personal care, and food preparation. Unilateral upper limb paralyses may influence these activities, but because most of the activities can be completed with one hand, the influence can be limited. This assumption is supported by the fact that when we controlled for the lower extremity motor function, the association between AMR/FMA and dependency was no longer statistically significant. However, the relatively high OR of the AMR in relation to dependency (Tables III and IV) may also suggest that the insignificance may be due to low power. In the end, lower extremity impairment as an indication of general level of mobility probably has a larger effect on dependency in self-care activities than unilateral impairment of an upper extremity. Other factors that may be of significance, though not found in this study, are cognitive function, apraxia, hemi-neglect, and loss of sensibility (30).
Regarding information bias, the FMA has not been validated in a Norwegian setting, and this may interfere with the quality of measurement. To ensure uniformity of the testing, Axel R. Fugl-Meyer, the main author of the original protocol (12), reviewed and approved the Norwegian version. The measurement of arm movement with accelerometers is considered to be very accurate. The Actigraph GT1M accelerometer used in this study uses digital sensors. All previous work on the upper extremity arm use has used older versions containing piezoelectric sensors (4, 10, 11, 16). Although the piezoelectric models are no longer available and the manufacturer states that the data consistency is maintained with the digital sensors, we cannot automatically assume that the reliability and validity of the piezoelectric sensors are valid for the digital (31). A possible source of error when measuring the AMR is the registration form, which requires the patients to record the hours spent driving a car, sleeping, or not wearing the accelerometers. Our analyses did, however, show that there was a minimal difference between the estimates from the crude data and the corrected data, which indicates that error from poor self-reports is probably insignificant. Even with good measurement properties, it is still a question whether the AMR is really measuring functional activity. Accelerometers record any movement, even if it is meaningless to the patient’s life or functioning. Only limited data is available on the relationship between functional activity and the AMR and, because of this, results from our measurements need to be interpreted with caution. The NIHSS was scored retrospectively, which is less accurate than prospective assessment according to the NIH protocol. This may have influenced the analysis of confounding factors and the description of stroke severity in the study sample.
Regarding selection bias, the age and gender distribution of the enrolled subjects did not differ significantly from the eligible and screened cases. However, more men than women were enrolled. This was partly because more men than women were invited to participate in trial 1 and because more men than women were eligible and consented to participate. This means that our results generalize mostly to men. Men and women may have a different activity profile after stroke, which may affect the association between arm use and other variables, but these differences are probably less evident in a inpatient hospital setting were our measurements were done. Most of the participants had mild to moderate strokes and a relatively high arm motor functioning, making them healthier than the general stroke population. This must be taken into account when interpreting the results of our study.
The concept of learned non-use after stroke is derived from animal research (3, 32) and, to our knowledge, there is little empirical evidence for its existence in humans. Interventions for treating learned non-use in chronic stage after stroke have developed in the past two decades (33). Based on the present results, we suggest that rehabilitation during the early phase should aim to prevent the development of learned non-use. Moreover, future studies should look at suitable prevention strategies. In addition, there is a need for larger prospective studies that would include data on hand preference, and would attempt to determine the optimal period for identifying and treating persons with learned non-use motor disabilities.
Acknowledgements
We would like to thank Gunn Marte Vadset, Grete Jakobsen, Kristin Fiskaa, Anders Thommessen, Cecile Jakobsen Steiro, Ane Fadnes, Eva Grønning, Ingrid Kristoffersen and Richard Grytvik for taking part in the recruitment and examination of subjects in this study.
References