OBJECTIVES: To evaluate the effects of a home-based balance training programme using visual feedback (Nintendo Wii Fit game with balance board) on balance and functional abilities in subjects with Parkinson’s disease, and to compare the effects with a group of paired healthy subjects.
SUBJECTS: Ten subjects with moderate Parkinson’s disease and 8 healthy elderly subjects.
METHODS: Subjects participated in a 6-week home-based balance training programme using Nintendo Wii Fit and balance board. Baseline measures were taken before training for the Sit-to-Stand test (STST), Timed-Up-and-Go (TUG), Tinetti Performance Oriented Mobility Assessment (POMA), 10-m walk test, Community Balance and Mobility assessment (CBM), Activities-specific Balance and Confidence scale (ABC), unipodal stance duration, and a force platform. All measurements were taken again after 3 and 6 weeks of training.
RESULTS: The Parkinson’s disease group significantly improved their results in TUG, STST, unipodal stance, 10-metre walk test, CBM, POMA and force platform at the end of the 6-week training programme. The healthy subjects group significantly improved in TUG, STST, unipodal stance and CBM.
CONCLUSION: This pilot study suggests that a home-based balance programme using Wii Fit with balance board could improve static and dynamic balance, mobility and functional abilities of people affected by Parkinson’s disease.
Key words: Parkinson’s disease; Wii; balance training; rehabilitation; physical therapy; neurorehabilitation; falls.
J Rehabil Med 2012; 00: 00–00
Correspondence address: Louis E. Tremblay PT, PhD, Université du Québec à Chicoutimi, 555 boulevard de l’Université,Chicoutimi, QC, G7H 2B1, Canada. E-mail: Louis1_Tremblay@uqac.ca
Submitted March 20, 2011; accepted October 21, 2011
Introduction
Parkinson’s disease (PD) is a degenerative neuropathology characterized by motor slowdown, rigidity and tremors, with decreased muscular strength and balance. These deficits are thought to be provoked by decreased activity of the basal ganglia, and of the motor cortex as a direct consequence (1, 2). Persons affected by PD are predisposed to long-term postural instabilities, thus increasing the likelihood of falls (3, 4). Even with optimal medication, falls are a significant problem (40–70% of PD-affected persons) (4). It has been suggested that a combination of disease-specific balance- and mobility-related measures could effectively prevent falls in individuals with PD (5, 6). Recent advances suggest that abnormal functioning of the pedunculopontine nucleus might be implicated in locomotion and axial deficits of PD. These studies suggest that even if dopa therapy has positive effects on motor symptoms, it may have insufficient effects on postural instability and impaired locomotion in PD, since dopaminergic drugs may not address the pedunculopontine nucleus because it uses cholinergic synapses (7–9). Interventions using exercise would be a good combination with dopa-therapy (2). Unfortunately, most healthcare systems provide only late physical interventions to persons affected by PD, who are in advanced stages of the disease when deficits are well established (10).
In the first stages of PD, physical exercise appears to be an effective solution to slow down the psychological, social and physical progression of this pathology by stimulating neuroplasticity and improving functional abilities (2, 11). Many studies have demonstrated that a conventional physiotherapy programme combining lower limb strength, endurance and flexibility exercises, as well as balance training, is efficient in improving balance of persons affected by PD (2, 12–14). The opposite has also been demonstrated; balance training can improve strength along with equilibrium of people affected by this disease (2, 14). It has been suggested that a home-based self-supervised exercise programme could be as beneficial as a physiotherapist-supervised programme in improving motor symptoms in patients with PD (15).
In recent years, virtual reality (VR) and visual feedback training are being used more frequently to improve many deficits, such as decreased balance and function, for different populations (16, 17). Butler & Willett (17) define VR as a technology that allows the user to interact directly with a computer-simulated environment. They include the Wii console and balance board within this definition as a recent technology that can be used in rehabilitation (17). Since the Wii does not provide the immersive environment of VR, the expression “visual feedback training” would be more appropriate. The Wii Fit balance board interacts by collecting data from the user’s body movements to effect a specific action in the game, thus constantly re-sending visual feedback to the user using a sensory-enriched environment. Studies have shown that balance training using visual feedback can improve postural responses and improve the balance of healthy elderly subjects post-stroke (16, 18, 19). According to previous research, individuals affected by PD show an increased dependence on visual information to control equilibrium (20, 21). Some new VR technologies and video games require the use of visual feedback, but most important is that some of these can easily be used independently at home for a relatively affordable price.
Interest among rehabilitation professionals and the general public has created a demand for research into the use of the Wii console in rehabilitation. A recently published study suggests that this new equipment would be a valid tool to assess standing balance compared with a laboratory force platform (22). A pilot study of Wii Fit training in a population of healthy women showed improvement in standing balance and lower limb strength (23).
The goals of this pilot study were: (i) to observe through objective testing whether PD subjects’ balance and functional abilities were improved by a 6-week home-based balance training programme using the Nintendo Wii and Wii Fit game with balance board; and (ii) to compare the effects on PD subjects with a paired sample of healthy subjects.
Methods
Subjects
The study protocol was approved by the Research Ethics Committee of the University of Ottawa, Canada. Participants were recruited on a voluntary basis throughout PD meeting groups in the greater Ottawa region. Two groups were determined, one composed of PD subjects and the other of paired healthy subjects, the majority being life partners of the PD subjects. Exclusion criteria were the same for both groups: cognitive inability to participate, the presence of at least one lower limb problem, including musculoskeletal, neurological and vascular; any history of falls; or the presence of any other health problem except PD. Eleven PD subjects (PD group) and 9 healthy subjects (HS group) were recruited (Table I). All subjects signed a detailed consent form before entering the study.
| Table I. Subject characteristics |
| Subjects | Healthy subjects (n = 9) 4 women, 5 men | | Parkinson’s disease (n = 11) 5 women, 6 men |
| |
| Mean (SD) | Min–Max | | Mean (SD) | Min–Max |
| Age, years | 63.5 (12.0) | 49–81 | | 61.9 (11.0) | 48–80 |
| Folstein Mini-Mental/30 | 29.9 (0.35) | 29–30 | | 29.5 (1.0) | 27–30 |
| Duration of PD, years | | | | 8.5 (3.6) | |
| UPDRS Motor score | | | | 18.4 (5.4) | |
| UPDRS: Unified Parkinson’s Disease Rating Scale; PD: Parkinson’s disease; SD: standard deviation. |
Procedures
Each subject was invited to 3 different evaluation sessions in order to gather a range of balance and functional data. The initial session took place during the week preceding the start of the training programme. The motor section of the UPDRS was administered to PD subjects to determine the level at which they were affected. Only in this first evaluation all subjects were asked to complete a health questionnaire to verify the presence of any contra-indication and had to score at least 24 on the Folstein Mini-Mental Test (MMT) (24) to ensure a sufficient level of cognitive ability to participate in the programme. The baseline values for all balance and functional tests were then taken. The second session was conducted after 3 weeks of training and the third within a week after the completion of the 6-week programme. All balance and functional tests were repeated in these two sessions. All sessions were performed at the same time of day and while the drugs prescribed for PD were having their optimal effect.
Functional balance and mobility
Several commonly used measuring scales were chosen to evaluate functional balance and mobility. Subjective perception of balance level and fear of falling were measured with the Activities-specific Balance and Confidence (ABC) scale (25). Several objective tests were administered during the 3 evaluation sessions; the timed up and go (TUG), in which the subject was asked to stand from a chair, walk 3 m, turn around and return to sit on the chair (26, 27), the sit-to-stand test (STST), in which the subject was asked to stand and sit back on a chair as many times as possible during 30 s (28), the Tinetti’s Performance-Oriented Mobility Assessment (POMA), containing several static and dynamic tasks for a maximum score of 28 (29), the Community Balance and Mobility scale (CBM), reflecting different functional tasks of the everyday life with a maximum score of 96 (30, 31) and the 10-m walking speed (mean of 3 trials) (32). Since functional balance and mobility improvement were the main goals of training, the objective testing focused on this aspect. We believe that improving results on tests such as the TUG, STST, walking speed and CBM would reflect on functional balance and mobility in the subjects’ activities of daily living (ADL).
Static balance
First, the one-leg stance duration, with a maximum of 45 s, was measured (mean of 3 trials) (26). Then, the antero-posterior and medio-lateral displacements of the centre of pressure (COP) were recorded and analysed using a Kistler force platform (Kistler Instrument Corporation, Amherst, NY, USA) and AMTI Netforce programme (Advanced Mechanical Technology Inc., Watertown, MA, USA). Subjects were asked to stand as still as possible, maintaining their arms by their side while keeping their head straight and feet together. A total of 6 trials were chosen in a random order: 3 trials with eyes open, with the subject looking at a red dot on the wall 2 m in front of the platform, and 3 trials with eyes closed. The resultant root mean square (RMS) from antero-posterior and medio-lateral velocity of COP was calculated and used as the measure for static postural stability.
Training programme
Home installation of the Wii console, detailed explanations of the programme, and supervision of the first complete training session was carried out by two members of the research team to ensure the safety of the subjects in their homes. Subjects also received a logbook, describing the experimental programme and additional precautions. The logbook was used to gather scores given by the Wii Fit game and to record subjective satisfaction with the programme for every training session by answering questions and completing Likert-type scales from 0 (not satisfied) to 5 (very satisfied).
Participants performed a specific programme of approximately 40 min for 3 days per week during 6 weeks (18 total training sessions). The programme consisted of a pre-determined number of repetitions for different exercises in the game, which was identical for all subjects. Each training day included 30 min with the Wii Fit game using the balance board and 10 min with the Wii Sports game (Golf or Bowling). The Wii Fit game contains 4 exercise sections. Balance games used were Table Tilt, Ski Slalom, Balance Bubble, Ski Jump and Penguin Slide. For these exercises either there is a marker on the screen that tracks the user’s movement and the challenge is the keep it within a certain area to win points, or a character/object moves according to weight shifting on the balance board. Only one yoga exercise was used (Deep Breathing), where the subject was asked to maintain the centre of pressure as still as possible. Only the Hula-Hoop was selected from the aerobics category. No exercise was chosen in the strength training section. No changes were made in the games played through the 6 weeks, to maintain uniformity among subjects. Training progression within the activities was carried out by the Wii Fit game itself. Increased difficulty levels could be permanently unlocked secondary to good performances, as well as temporary levels according to daily results. At the point where deep breathing was too easy, subjects were asked to close their eyes. PD subjects were instructed to always train at times when their medication was effective. A motivational telephone call was made by one of the researchers every week and to ensure that the subjects were doing well. Participants were encouraged to contact the research team to ask any questions about the programme or to request assistance with technical problems.
Statistical analysis
Because of the relatively small number of participants included in this study (n < 30) and since visual inspection of the histograms did not allow us to assume that the data were normally distributed, non-parametric tests were employed. Specifically, the Friedman’s test was used to compare the multiples comparison’s conditions and the Wilcoxon’s test to compare two conditions. Significant results were followed with Wilcoxon signed-rank tests. Differences were considered to be significant if p <0.05 was obtained. All tests were performed using Graphpad Prism 5 (Graphpad Software Inc., La Jolla, CA, USA).
Results
The results of the different tests are shown in Figs 1–7 as well as p-values for significant results, always compared with the baseline score. Power analysis using Mann-Whitney tests based on CBM results indicate a 67% statistical power. Effect size is 0.74 for PD and 0.48 for HS.
Exclusions
One subject in each group did not finish the training programme. The PD subject stopped for personal health reasons after only 9 training sessions and had to cancel the mid-programme evaluation session, and his wife (HS) also decided to stop. Therefore, 10 PD and 8 HS subjects were considered for all results.
Static balance
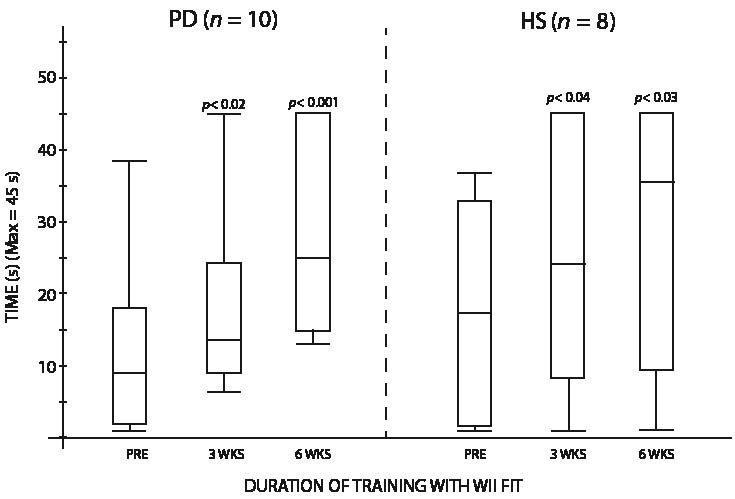
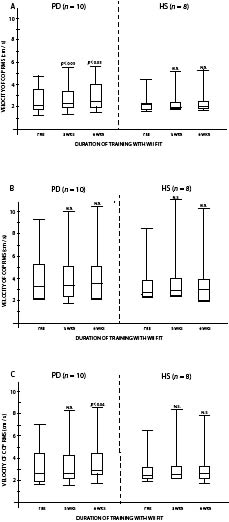
One-leg stance duration increase was significant for PD, with a 15.2 s median value improvement, and for HS, where the median value increase was 18.1 s (Fig. 1). On the force platform (Figs 2 A–C), PD increased their COP RMS velocity after 6 weeks by 0.42 cm/s with eyes open (p <0.05), by 0.41 cm/s with eyes closed (not significant (N.S.)) and by 0.43 cm/s when combining the two conditions (p <0.04). HS increased their COP RMS velocity by 0.17 cm/s with eyes open (N.S.), by 0.24 cm/s with eyes closed (N.S.), and by 0.21 cm/s when combining the two conditions (N.S.).
Fig. 1. Duration of one-leg stance by both groups. PD: Parkinson’s disease; HS: healthy subjects.
Fig. 2. Results for both groups for the centre of pressure root mean square (COP RMS) velocity with: (A) eyes open; (B) eyes closed; and (C) combining eyes open and eyes closed. PD: Parkinson’s disease; HS: healthy subjects; N.S.: not significant.
Dynamic balance and functional abilities
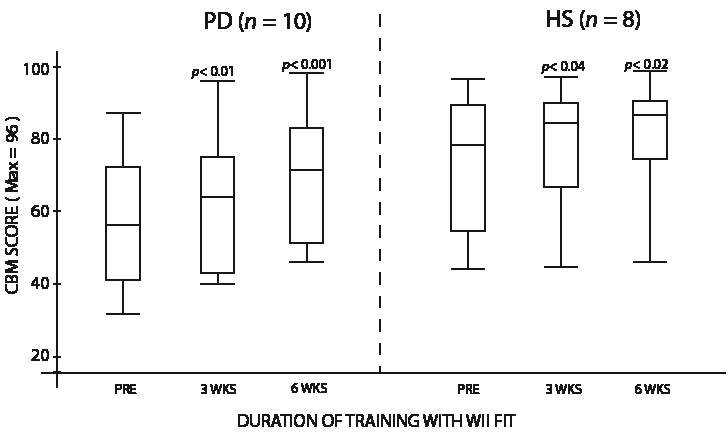
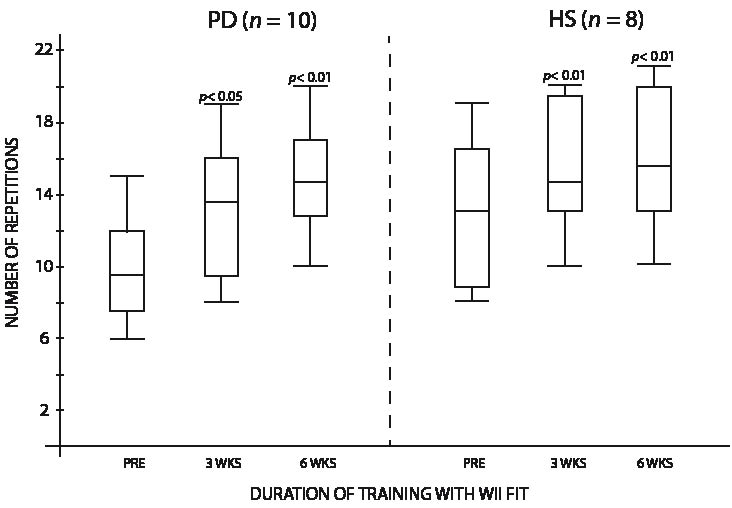
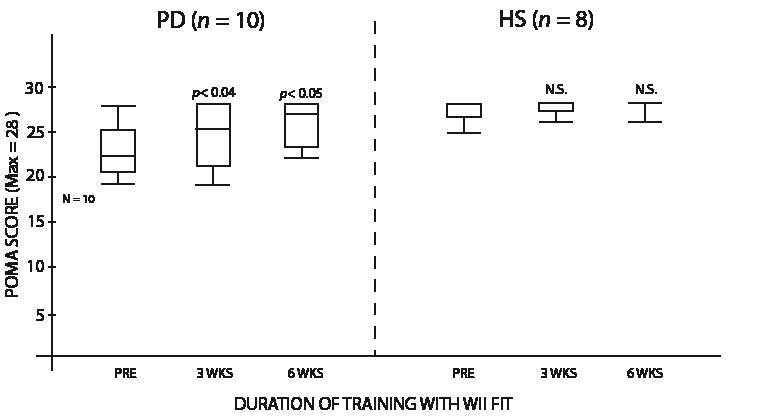
Improvement in the scores to the CBM test was observed in both groups, as PD subjects increased their median score by 15.0 points (p <0.001) and HS subjects by 7.5 points (p <0.02) (Fig. 3). For the STST, significant increase was observed, with a total 6-week median improvement of 5.0 repetitions for PD subjects (p <0.01) and 2.5 for HS subjects (p <0.01) (Fig. 4). Significant increase was also obtained for the TUG, with a median value of 1.9 s less needed to do the test for the PD group (p <0.04), while HS subjects median value improved by 1.2 s (p <0.001) (Fig. 5). The 10-m walking speed also increased significantly after the 6-week training, with a 0.7 second median value decrease for PD subjects to complete the distance (p <0.001). HS variation was not significant, the median value decreasing by 0.4 s (Fig. 6). Results variation on the Tinetti’s POMA was significant for PD subjects, with a 4.0 point median increase out of 28 (p <0.05), but not for HS subjects, where the median value was already at the maximum score pre-training and stayed at this same perfect score of 28 (Fig. 7). There was no significant change in the ABC scale, where PD increased by 1.0 point and HS decreased by 1.0 point.
Fig. 3. Scores for both groups on the Community Balance and Mobility scale (CBM). PD: Parkinson’s disease; HS: healthy subjects.
Fig. 4. Repetitions performed by both groups on the Sit-to-Stand test. PD: Parkinson’s disease; HS: healthy subjects.
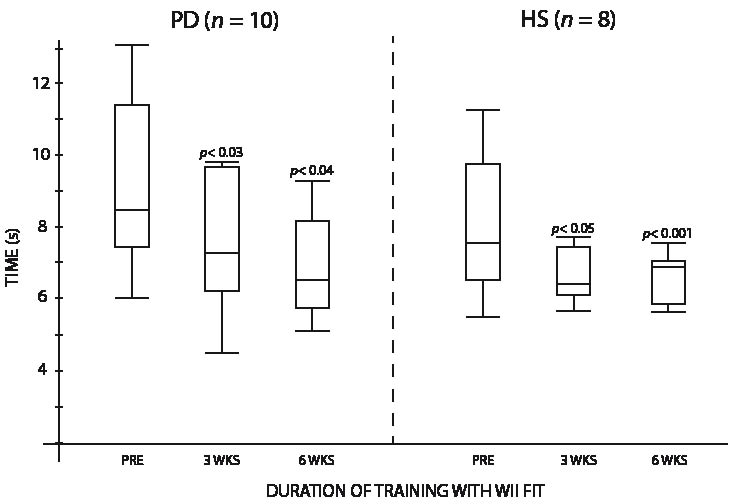
Fig. 5. Time to complete the Timed-Up-and-Go by both groups. PD: Parkinson’s disease; HS: healthy subjects.
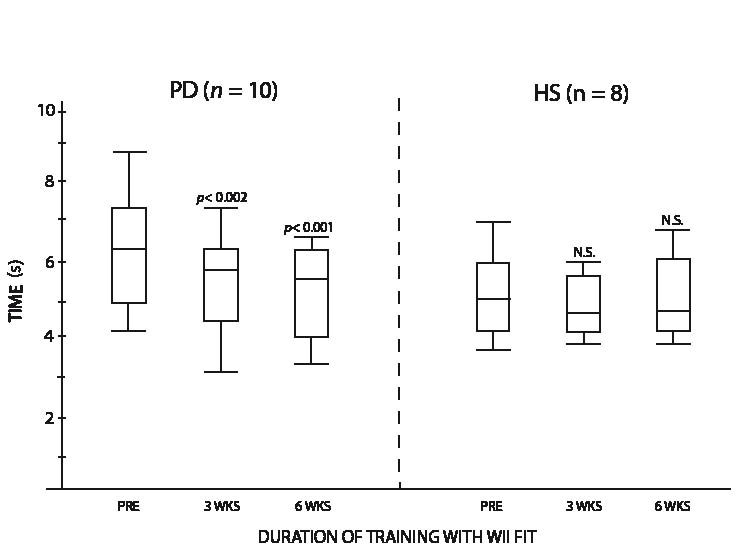
Fig. 6. Time to complete 10-m walk test by both groups. PD: Parkinson’s disease; HS: healthy subjects.
Fig. 7. Scores for both groups on the Performance Oriented Mobility Assessment (POMA). PD: Parkinson’s disease; HS: healthy subjects; N.S.: not significant.
Subjective appreciation
A customized questionnaire was completed by participants at the end of the 6-week training programme. Fifty percent of participants liked the training programme very much, 33% liked it, 17% were neutral and no subject disliked the programme. Most of the PD subjects reported having fun while exercising, since the majority trained with their spouse. Some even exercised with their children or grandchildren. Favourite games were Ski Jump, Ski Slalom and Table Tilt.
Discussion
Both PD and HS demonstrated an improvement in most of the static, dynamic and functional balance aspects, as shown in Figs 1–7. Better scores occurring through the whole programme suggest that subjects were still improving after 6 weeks. Outcome measures used in this study evaluated static and dynamic balance, which were trained while playing the Wii Fit game, and functional tests to determine if the training had an effect on mobility and functional abilities. Improvements in the range of tests used suggest that progress occurred for both groups.
Increased one-leg stance duration indicates that subjects reached the end of this programme with much better static balance. As shown in Fig. 1, the evolution of improvement for the 1-leg stance in PD started more significantly from the mid-point of the programme, suggesting a slower learning process. As for the force platform, results with eyes open and combining the two conditions lead to a significant increase in COP RMS velocity only for PD; our main hypothesis is that PD had a better internal representation of their balance and COP after training, thus improving their motor control to help maintain COP within normal limits, even if its velocity increased. We hypothesize that using a more complex task while standing on the platform (foam interface, dynamic task) would have been more sensitive to balance changes induced in HS by the training program. During initial testing, we decided to use the force platform alone, as using advanced testing was too difficult and less safe for PD. Bisson et al. (16) come to the same conclusion while using a similar force platform protocol with healthy elderly subjects.
The improvement in the TUG, CBM and POMA provide indications of the increased balance and functional mobility of the PD group. Indeed, the mean decreased time to complete the TUG (2.11 s, median 1.9 s) is a notable change (minimal detectable change is 2–5 s for PD-affected people) (25). According to Inness & Howe (31), the minimal clinically important difference on CBM was easily obtained by PD, with a mean increase of 12.1 points (median 15.0 points). This important change suggests that functional balance in ADL could be improved, as the CBM reproduces common functional situations (30). As for walking speed during the 10-m walking test, PD subjects increased their performance by 0.31 m/s, a value higher than the minimal clinically important difference of 0.25 m/s (25). According to Wolfson et al. (33), this change suggests an improvement in mobility, fitness and dynamic balance. The higher number of repetitions noted on the STST suggests that this Wii Fit programme improved not only balance, but also the overall functional strength of the lower limbs (34). This could be explained in part by the Ski Jump activity, where the user had to squat for approximately 5 s and then quickly stand back up straight in order to make a longer jump.
Although changes to the ABC scale were not significant for either group, PD subjects reported increased balance and stability in their ADL, and reduced fear of falling at the end of the programme. It has been suggested that fear of falling could be an important function-limiting factor for individuals with PD (35).
Previous studies have suggested balance improvement in elderly people following visual feedback training (19), and in healthy middle-aged women using the Wii balance board (23). The Wii console and Wii Fit game offer visual feedback training as a direct application of VR, while being much less expensive and much more accessible than several programmes used in specialized clinics and research laboratories. Furthermore, it provides a safe way of training in the home setting, which is interesting from a clinical point of view. We believe that one of the most important successes of the programme lies in the pleasure component of training, with simple amusing games that can be performed at home with a life partner, hence maximizing compliance with the activity. To participate in such a programme, one does not necessarily need an intact intellect. In this study, the lowest score obtained on the MMT was 27 and subjects did not experience difficulty using the game after proper supervision and guidance of the first training session.
Our hypothesis for the good results achieved is that the Wii Fit offers a rich environment of sensory cues combining visual, auditory and proprioceptive feedback. It may provide persons affected by PD more ways to enhance attention and focus on the required moves in the game and make them more explicit by using the motor, oculomotor, cerebellar and limbic loops (36). Proprioceptive feedback is enhanced by the constant signal sent to the visual system by the game depending on weight shifting on the balance board. Users are also motivated by the results obtained in the game, trying to gain points and beat their scores. The reward circuitry may be activated by this process, and it has been stated that PD would benefit from such intervention (37, 38). Also, de la Fuente-Fernandez et al. (37, 38) suggest that the expectation of clinical benefit would be equivalent to the expectation of reward, hence stimulating the placebo effect by releasing dopamine in the ventral striatum.
The use of observation in PD rehabilitation has been a recent research interest. Some authors suggest that the use of observation while executing an action could facilitate motor re-learning in neuro-rehabilitation by the use of mirror neurones, which could prepare the individual for action imitation (39, 40). In activities such as Ski Slalom and Ski Jump, the user sees an avatar who mimics his or her movements while playing. This could be another factor supporting good results obtained in improvements through the 6-week programme.
Limitations
To our knowledge, no previous study has been carried out on PD subjects using applications of VR for balance training. This pilot study did not include a control group and evaluated an intervention with small groups. Given the small number of PD subjects available for the study, we decided to regroup them in the same group of Wii Fit exercise. The programme was performed with a 6-week training period with positive results. It would be interesting to study a longer period of 8–12 weeks, as improvements were still noted after 6 weeks for some tests. The absence of follow-up evaluation should also be noted as a limitation of this study, as improvements could not be evaluated as durable or not. The fact that the investigators administered the outcome measures should also be considered as another limitation of this study. Although the small number of subjects who participated in this pilot study indicate promising results, further research is needed with more subjects in order to generalize these findings to the PD population.
Results from this pilot study suggest that a home-based training programme using visual feedback with the Wii and Wii Fit could lead to improved static and dynamic balance, global mobility and functional abilities of PD-affected persons. Healthy elderly subjects also improved, but to a lesser extent. Clinical implications are significant for the PD population, who are most often neglected by the healthcare system during the first stages of the disease; as such a programme would permit an early and pleasant homecare plan. Further studies on the use of the Wii are needed in order to generalize these results.
Acknowledgements
The authors would like to thank all the subjects who participated in the study, the “Regroupement des personnes atteintes de la maladie de Parkinson de l’Outaouais“, Parkinson Society Ottawa and Étienne Bisson, PhD candidate, for his help in measuring force platform results.
References