Casper F. van Koppenhagen, MD, PhD1,2, Sonja de Groot, PhD3,4, Marcel W. M. Post, PhD1,2, Floris W. A. van Asbeck, MD, PhD2, Dorien Spijkerman, MD5, Willemijn X. M. Faber, MD6, Eline Lindeman, MD, PhD1,2† and Lucas H. V. van der Woude, PhD4,7
From the 1Department of Rehabilitation Medicine and Rudolf Magnus Institute for Neuroscience, University Medical Center Utrecht, 2De Hoogstraat Rehabilitation, Utrecht, 3Amsterdam Rehabilitation Research Center, Reade, Amsterdam, 4University of Groningen, University Medical Center Groningen, Center for Human Movement Sciences, 5 Rijndam Rehabilitation Center, Rotterdam, 6Heliomare Rehabilitation Centre, Wijk aan Zee and 7University of Groningen, University Medical Center Groningen, Center for Rehabilitation, Groningen, The Netherlands
OBJECTIVE: To elucidate the course and determinants of wheelchair exercise capacity in spinal cord injury up to 5 years after discharge from inpatient rehabilitation, and to describe loss to follow-up.
DESIGN: Prospective cohort study, with measurements at the start and discharge from inpatient rehabilitation, 1 and 5 years after discharge.
SUBJECTS: A total of 225 wheelchair-dependent persons with spinal cord injury.
METHODS: Random coefficient analysis of the course and determinants of peak aerobic power output (POpeak) and peak oxygen uptake (V.O2peak).
RESULTS: A total of 162 participants attended one or more peak exercise tests and were analysed. Significant changes were found for both V.O2peak and POpeak between start and 5 years after discharge, and discharge and 5 years after discharge. No significant changes were found for V.O2peak and POpeak between 1 year and 5 years after discharge. Age, gender, level and completeness of lesion were determinants for level of V.O2peak and age, gender, and level of lesion for level of POpeak. No significant determinants were found for the course of wheelchair exercise capacity. The 63 participants who were not analysed were older, and showed more persons with a tetraplegia.
CONCLUSION: Wheelchair exercise capacity of persons with spinal cord injury stabilizes between 1 and 5 years after discharge. The participants appear to be a positive selection of the total study group.
Key words: spinal cord injuries; wheelchair; physical fitness; cohort; lost to follow-up.
J Rehabil Med 2013; 45: 00–00
Correspondence address: Casper F. van Koppenhagen, Rehabilitation, Rehabilitation Centre De Hoogstraat, 3583TM Utrecht, The Netherlands. E-mail: casper.koppenhagen@planet.nl
Accepted Jan 17, 2013; Epub ahead of print Maj 17, 2013
Introduction
A spinal cord injury (SCI) is one of the greatest physical calamities that one can overcome (1), and dramatically impacts exercise capacity and activity level of persons involved (2–5). The majority of persons with SCI are wheelchair users and depend on arm work for mobility and activities of daily living. Wheelchair exercise capacity is the combined ability of the cardiovascular, respiratory and musculoskeletal systems to attain a certain level of wheelchair activity (6, 7). The different components of wheelchair exercise capacity are influenced by personal and lesion-related factors, exercise mode, expertise and training (3, 7–9). Wheelchair exercise capacity is diminished in many persons with SCI because of muscle weakness, loss of autonomic control below the level of injury and subsequent changes in metabolic and vascular function. Wheelchair exercise capacity is an important determinant of health status of subjects with SCI, especially in the long-term (10), because a low wheelchair exercise capacity exposes them to increased risk of developing medical conditions, such as metabolic syndrome and cardiovascular disease (11), and is related to a reduced level of functioning, a reduced level of activities and participation and quality of life (11–14).
Most studies on exercise capacity in the SCI population have been cross-sectional, and include healthy, young and active male persons with a long time since injury (> 6 years) (15, 16). A recent longitudinal study with 20 years follow-up (n = 7 persons with SCI) revealed a stable exercise capacity (17). In our own SCI cohort, on which the present study is based, recovery of wheelchair exercise capacity during inpatient rehabilitation up to 1 year after discharge was positively associated with lower age, male gender, low level and incompleteness of the lesion (7), as was found in other cross-sectional studies on determinants and the level of exercise capacity (2, 11). No other longitudinal studies on this topic were found.
Persons with SCI endure a process of adaptation to their new life situation in the first few years after injury (18). Once they have reintegrated in society, they have to deal with many threats and barriers to maintaining an adequate exercise capacity (10). Therefore, it is important to study the course of exercise capacity at the mid- and long-term and to identify determinants that influence exercise capacity over a longer follow-up period. Based on the limited longitudinal literature on this subject we hypothesize that the exercise capacity will remain stable up to 5 years (5Y) after discharge (3, 7, 17, 19, 20).
In a longitudinal study, especially including a voluntary and physically demanding peak wheelchair exercise test, loss to follow-up is inevitable. As the literature reveals, loss to follow-up is clearly related to age and disability and the perceived benefit of the test (21, 22). In long-term follow-up, the ability to trace and contact subjects is difficult and accounts for a higher level of loss to follow-up, as already described in our study population (23). Therefore, we expected the loss to follow-up to be higher at 5Y for the older persons and for those with a more severe spinal cord lesion. In order to control for the effects of loss to follow-up in the best possible way, we used random coefficient analyses, which allows the inclusion of those participants who (temporarily) dropped out at some point during the longitudinal study (24).
The aims of the current study are: (i) to determine the course of wheelchair exercise capacity of persons with SCI and its determinants up to 5 years after discharge from inpatient rehabilitation; and (ii) to describe the loss to follow-up.
Methods
Subjects
This study is part of the research programme “Restoration of mobility in the rehabilitation of persons with a SCI” (25). Eight rehabilitation centres that specialize in SCI participated in the programme. Subjects were eligible to enter the study if they had an acute SCI; were between 18 and 65 years of age; were classified as A, B, C or D on the American Spinal Injury Association (ASIA) Impairment Scale (26); and were expected to remain wheelchair-dependent, at least for community use. Exclusion criteria were: SCI due to malignancies, progressive disease, known cardiovascular disease or psychiatric problems; insufficient command of the Dutch language to understand the goal of the study and the testing methods. The medical ethics committee of the Stichting Revalidatie Limburg/Institute for Rehabilitation Research in Hoensbroek approved the research protocol in 1999, and the medical ethics committee of the University Hospital of Utrecht approved for the follow-up research protocol in 2006. All subjects gave written informed consent.
Procedure
Measurements were performed at the start of active inpatient rehabilitation (start, defined as the moment that a person could sit for 3–4 h),at discharge from inpatient rehabilitation (discharge), 1 year after discharge (1Y), and 5Y. These 4 occasions comprised, amongst others, a medical history and physical examination by a rehabilitation physician and a wheelchair peak exercise test (9, 27).
Peak exercise test
In this study exercise capacity was defined as wheelchair exercise capacity, being the peak results of the peak exercise wheelchair test in external power output (POpeak (W)) and oxygen uptake (V.O2peak (l/min)). To determine POpeak and V.O2peak, subjects performed a graded maximal wheelchair exercise test on a motor-driven treadmill. The testing protocol and equipment have previously been described by Kilkens et al. (28) and Haisma et al. (7). Before testing, subjects were asked to eat a light meal only, to refrain from smoking and drinking coffee or alcohol before testing, and to void their bladder. For each subject, and at every occasion, a drag test was used to determine the drag force and concomitant external power output for the wheelchair-user system on the treadmill at increasing inclinations and the actual testing speed (29). Subjects performed two blocks of submaximal exercise of 3 min each, separated by a 2-min rest. The treadmill incline was horizontal during the first block and then set at 0.36º during the second block. Treadmill velocity was set at 0.55 m/s for subjects with tetraplegia and at 1.1 m/s for subjects with paraplegia. In some subjects with a low cervical lesion, we used a protocol with a velocity of 0.89 m/s. After 2 min of rest, the peak exercise test followed at the same constant velocity, and the inclination was increased by 0.36º every minute. The test was terminated when the subject was exhausted or could no longer keep pace with the speed of the treadmill. The individual testing protocol was identical for each of the testing occasions.
The V.O2peak was defined as the highest value of oxygen consumption recorded during a period of 30 s. The POpeak was defined as the power output at the highest inclination that the subject could maintain for at least 30 s. Participants were tested at consistent time settings for every measurement.
Demographic characteristics collected at the first test occasion were age, gender, body weight and height. At each subsequent test occasion body mass (kg) was measured by the trained research assistant. Body mass index (BMI) was calculated in kg/m².
Lesion characteristics were assessed according to the International Standards for Neurological Classification of Spinal Cord Injury (26). The ASIA Impairment Scale classifications A and B were considered motor complete, and the classifications C and D were considered motor incomplete. Neurological lesion level was defined as the highest motor level. We clustered the cervical, thoracic and lumbar lesions for presentation. Neurological levels below T1 were defined as paraplegia, and neurological lesion levels at or above T1 were defined as tetraplegia. Cause of injury was dichotomized in traumatic vs non-traumatic (e.g. spinal cord infarction, benign tumours, infections). Time since injury was counted in years.
Statistics
Descriptive statistics (means and standard deviations) of personal and lesion characteristics and at all test occasions were calculated with SPSS 16.0.
Random coefficient analysis (MlwiN version 1.1; Centre for Multilevel Modelling, Institute of Education, London, UK) was used to study the course of wheelchair exercise capacity up to 5 years after inpatient rehabilitation and its determinants (24). The benefits of this method are: (i) that it accounts for the dependency of repeated measures within the same person; (ii) that it accounts for the hierarchical nature of the longitudinal data of the present study (3 levels of hierarchy are present: the repeated measurements are nested within the participants and the participants are nested with rehabilitation centres); and (iii) that, in contrast to traditional methods of longitudinal data analysis (i.e. multivariate analysis of variance (MANOVA) for repeated measures), the number of observations per individual may vary, so repeated measures as well as cases with missing values can be included in this multiple regression analyses. Therefore, we included the persons who performed one or more peak exercise tests during the test period (STUDY), which allows us to evaluate the course over time as well as the role of different determinants (24). STUDY was divided into a group with tetraplegic lesions and a group with paraplegic lesions, and the course of exercise capacity for these groups was studied separately.
Possible determinants of the course of wheelchair exercise capacity were examined in a random coefficient regression model with the POpeak and V.O2peak at start, discharge, 1Y and 5Y as the dependent variables. Time was included in the basic model as a set of 3 dummy variables, representing the long-term period after the start of active inpatient rehabilitation (start–5Y), the period after inpatient rehabilitation (discharge–5Y) and the period from 1 to 5 years after discharge (1Y–5Y). The 5Y occasion was chosen as the reference and was estimated by the intercept. The independent variables selected for further analysis were personal: age (years), gender (woman = 1, man = 0); and lesion characteristics: level of SCI (tetraplegia = 0; paraplegia = 1) and completeness of SCI (complete = 1; incomplete = 0). These were added 1 by 1 to the basic model to study their individual relationship with the wheelchair exercise capacity scores. Finally, all independent variables that were significantly (p < 0.10) related to the POpeak or V.O2peak score were simultaneously entered in the multivariate random coefficient regression model using the backward elimination method, leading to a final model for POpeak and V.O2peak (p < 0.05).
Furthermore, the interaction of the personal and lesion characteristics with time was studied by adding them to the basic model.
The consequences of loss to follow-up on the study outcomes were studied in different ways. First, we entered a variable yes/no5Y-test into the multivariate random coefficient regression model for the STUDY group in order to study the effect of loss to follow-up on the course of POpeak and V.O2peak. The consequences of loss to follow-up on the study outcomes were studied in different ways. First, we entered a variable yes/no5Y-test in the multivariate random coefficient regression model for the STUDY group to study the effect of loss to follow-up on the course of POpeak and V.O2peak. Secondly, we identified and compared several subgroups: the participants who performed one or more peak exercise tests during follow-up (STUDY) and whose data were used in the random coefficient regression model. The participants who performed no peak exercise test (EXCLUDED) were identified and not used in the model. We performed t-tests and Chi-square tests to compare the STUDY versus EXCLUDED groups on available personal and lesion characteristics at the start and at 5Y (p < 0.05). Further, we identified the participants who attended all the peak exercise tests (ALLTESTS) and studied their personal and lesion characteristics and their POpeak and V.O2peak scores at all occasions. We performed t-tests and on available POpeak and V.O2peak scores at all measurements (p < 0.05) for ALLTESTS and STUDY.
Results
Respondent characteristics
A total of 225 subjects were included in the study. Of these, 162 performed 1 or more peak exercise tests and were included in the random coefficient analyses (STUDY). The remaining group with no peak exercise tests (n = 63) were treated as lost to follow-up (EXCLUDED). The characteristics of persons in the STUDY and EXCLUDED groups are shown in Table I.
|
Table I. Personal and lesion characteristics at start, and outcomes of wheelchair exercise test at different measures of the participants (STUDY) and non-participants (EXCLUDED) and participants that attended all 4 peak exercise tests (ALLTESTS) in means and standard deviations (SD) or percentages |
||||
|
Characteristics |
ALLTESTS n = 31 |
STUDY n = 162 |
EXCLUDED n = 63 |
p-value |
|
Age, years, mean (SD) |
38.0 (6.5) |
39.3 (13.8) |
43.8 (13.9) |
0.00* |
|
Gender, male, % |
|
76 |
72 |
0.63 |
|
BMI, kg/m, mean (SD) |
23.7 (3.6) |
22.8 (3.8) |
22.9 (3.9) |
0.56 |
|
AIS, % |
||||
|
A |
69 |
51 |
36 |
0.00* |
|
B |
8 |
14 |
23 |
|
|
C |
14 |
18 |
25 |
|
|
D |
9 |
17 |
16 |
|
|
Type of lesion, % |
||||
|
Cervical |
10 |
49 |
37 |
0.00* |
|
Thoracic |
81 |
104 |
18 |
|
|
Lumbar |
9 |
18 |
4 |
|
|
Type of injury, % |
||||
|
Tetraplegic Paraplegic |
10 90 |
27.1 62.9 |
62.7 37.3 |
0.00* |
|
Cause of injury, % |
||||
|
Trauma Non-trauma |
87 13 |
76 24 |
67 33 |
0.32 |
|
Time since injury at 5Y, years, Mean (SD) |
6.6 (0.7) |
6.4 (2.3) |
6.6 (0.9) |
0.14 |
|
POpeak, mean (SD) |
||||
|
Start (W) |
35.2 (16.3) |
33.1 (18.8) |
||
|
Discharge (W) |
51.5 (20.2) |
44.1 (23.7) |
||
|
1Y (W) |
54.9 (23.0) |
51.5 (27.4) |
||
|
5 Y (W) |
56.3 (22.2) |
53.1 (24.7) |
||
|
VO2peak, mean (SD) |
||||
|
Start (l/min) |
1.00 (0.3) |
1.03 (0.4) |
||
|
Discharge (l/min) |
1.24 (0.4) |
1.21 (0.4) |
||
|
1Y |
1.31 (0.4) |
1.32 (0.5) |
||
|
5Y |
1.38 (0.5) |
1.36 (0.5) |
||
|
*Significance set at p < 0.05 for t-test and χ2 tests between STUDY and EXCLUDED. Personal and lesion characteristics were counted at start of inpatient rehabilitation. ALLTESTS: persons who attended all 4 peak exercise tests; STUDY: persons who attended 1 or more out of 4 peak exercise tests; EXCLUDED: persons who attended no peak exercise test; BMI: body mass index; PO: peak power output; VO2: peak oxygen intake; Start: start of active rehabilitation; Discharge: discharge from inpatient rehabilitation; 1Y: test occasion at 1 year after discharge from inpatient rehabilitation; 5Y: test occasion at 5 years after discharge from inpatient rehabilitation; AIS: American Spinal Injury Association Impairment Scale. |
||||
Course of wheelchair exercise capacity in STUDY
Figs 1 and 2 show the course of wheelchair exercise capacity over time, as estimated with the basic regression model. We found a significant improvement in POpeak and V.O2peak in the intervals start–5Y and discharge–5Y. In addition, we found no change in the course of POpeak between 1Y (51.5 W) and 5Y (53.1 W), or in the course of V.O2peak between 1Y (1.32 l/min) and 5Y (1.36 l/min).
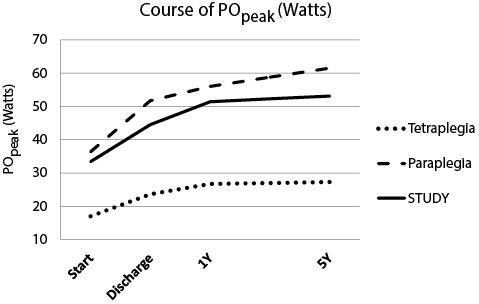
Fig. 1. Course of POpeak (Watts) as calculated from the regression model with lesion level added. Estimated with the basic regression model (constant + 3 time dummies). Tetraplegia and Paraplegia figures were estimated with regression model including level of lesion as determinant (constant + time dummies + lesion level + interaction term lesion level × time dummies). The improvement in intervals Start–5Y and Discharge–5Y were significant for STUDY, tetraplegia and paraplegia. p < 0.05. POpeak: peak power output; Start: start of clinical rehabilitation; Discharge: discharge from clinical rehabilitation; 1Y: 1 year after discharge; 5Y: 5 years after discharge; SCI: spinal cord injury; STUDY: total group (tetraplegia and paraplegia) models.
Fig. 2. Course of VO2peak (l/min) as calculated from the regression model with lesion level added. Estimated with the basic regression model (constant + 3 time dummies). Tetraplegia and Paraplegia figures were estimated with regression model including level of lesion as determinant (constant + time dummies + lesion level + interaction term lesion level × time dummies). The improvement in intervals Start–5Y and Discharge – 5Y were significant for STUDY, tetraplegia and paraplegia. p < 0.05. VO2peak: peak oxygen uptake; Start: start of clinical rehabilitation; Discharge: discharge from clinical rehabilitation; 1Y: 1 year after discharge; 5Y: 5 years after discharge; SCI: spinal cord injury; STUDY: total group (tetraplegia and paraplegia) models.
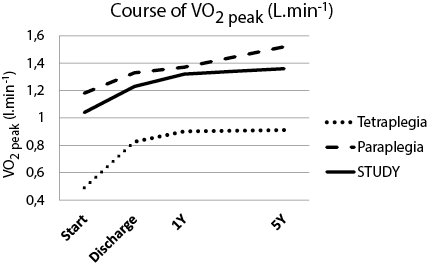
Model outcomes for the subjects with paraplegia and tetraplegia are presented separately in Figs 1 and 2, showing an improvement during inpatient rehabilitation and a stabilization after discharge up to 5Y. There are no differences in the course of POpeak and V.O2peak for persons with tetraplegia or paraplegia up to 5 years after discharge from inpatient rehabilitation. We found no significant difference in the level and course of POpeak and V.O2peak from start up to 5Y between the subgroup of STUDY that attended the 5Y measurement and the subgroup that did not attend the 5Y measurement.
Determinants
Table II presents the outcomes of the random coefficient analysis on the association between the wheelchair exercise capacity (POpeak and V.O2peak) and the time intervals (basic regression model) and the association between wheelchair exercise capacity, the determinants personal and lesion characteristics and the time intervals (final backward regression model).
|
Table II. Data on the longitudinal relationship between subject and lesion characteristics and the (change in) physical capacity in STUDY. Basic model and final backward regression model presented |
|||||||
|
Independent variable |
POpeak (W) |
V.O2peak (l/min) |
|||||
|
Beta |
SE |
p-value |
Beta |
SE |
p-value |
||
|
Basic model |
|||||||
|
Constant (reference 5Y) |
53.10 |
3.95 |
1.36 |
0.08 |
|||
|
D 5Y–Start |
–19.86 |
3.47 |
0.00* |
–0.32 |
0.07 |
0.00* |
|
|
D 5Y–Discharge |
–12.64 |
3.60 |
0.03* |
–0.14 |
0.07 |
0.04* |
|
|
D 5Y–1Y |
–1.60 |
3.63 |
0.42 |
–0.04 |
0.07 |
0.26 |
|
|
Final model |
|||||||
|
Constant (reference 5Y) |
61.81 |
4.11 |
1.87 |
0.12 |
|||
|
D 5Y–Start |
–20.81 |
2.74 |
0.00* |
–0.38 |
0.06 |
0.00* |
|
|
D 5Y–Discharge |
–7.11 |
2.63 |
0.04* |
–0.15 |
0.05 |
0.03* |
|
|
D 5Y–1Y |
–3.83 |
2.86 |
0.23 |
–0.11 |
0.06 |
0.03* |
|
|
Gendera |
–15.01 |
1.88 |
0.00* |
–0.31 |
0.04 |
0.00* |
|
|
Age (10 years) |
–3.10 |
0.06 |
–0.07 |
0.00 |
0.00* |
||
|
Levelb |
–29.04 |
1.93 |
0.00* |
–0.46 |
0.04 |
0.00* |
|
|
Completec |
– |
– |
–0.23 |
0.04 |
0.00* |
||
|
All results are regression coefficients (b) and standard errors (SE) for the regression model (constant + time dummies + determinant). The regression coefficients represent the change in outcome associated with an increase in the independent variable of 1 unit. For the peak power output the interaction term 5Y–Start, for example, indicates the difference between 5Y and Start was –20.81 W, i.e. power output improved from start up to 5 years after discharge 20.81 W. *p < 0.05. a0: men; 1: women; b0: paraplegia; 1: tetraplegia; c0: incomplete; 1 = complete; 1Y/5Y: test occasion, respectively 1 and 5 years after discharge from inpatient rehabilitation; POpeak: peak power output; VO2: peak oxygen intake. |
|||||||
The regression coefficients represent the change in POpeak and V.O2peak with an increase in the independent variable of 1 unit. Age, gender and level of lesion were significantly related to POpeak and VO2peak. An increase of 10 years in age was associated with a decrease of 3 W in POpeak and a decrease of 0.07 l/min in V.O2peak. Men had a significantly higher POpeak (+15.0 W) and V.O2peak (+0.31 l/min) than women. Paraplegia was associated with a higher POpeak (+29.0 W) and V.O2peak (+0.46 l/min) than tetraplegia. Subjects with complete lesions had 0.23 l/min lower V.O2peak than persons with incomplete lesions.
We found no significant differences in the course of wheelchair exercise capacity between persons with different personal or lesion characteristics (interaction terms of personal or lesion characteristics with time) and these outcomes are therefore not presented.
Determinants of loss to follow-up
In comparison with the STUDY group, the EXCLUDED group was older and included relatively more persons with tetraplegia (Table I). Only 31 persons attended the peak exercise tests at all occasions (ALLTESTS). The characteristics of this group are also presented in Table I. We found no significant differences in the outcomes of the wheelchair exercise test for the STUDY and ALLTESTS.
Discussion
Course of wheelchair exercise capacity
Random coefficient analysis revealed a significant change in both V.O2peak and POpeak for the intervals start–5Y and discharge–5Y. No significant changes in V.O2peak and POpeak were found for the 1Y–5Y interval. Age, gender, level and completeness of lesion were determinants for VO2peak scores and age, gender and level of lesion and for POpeak scores. No significant determinants were detected for the course of wheelchair exercise capacity in the 1Y–5Y interval. The loss to follow-up group was older and included more persons with tetraplegia, probably leading to an overestimation of the model outcome for wheelchair exercise capacity.
Our study is the first published longitudinal study of wheelchair exercise capacity in a general cohort of SCI patients with measurements taken both during inpatient rehabilitation and at follow-up to 5 years after discharge. We included all subjects in the multilevel regression analyses with one or more wheelchair exercise tests from the start of active inpatient rehabilitation. This gives us the important advantage of including more subjects with missing values during follow-up, which improves the power of our analyses (24).
In our previous study (7) the mean wheelchair exercise capacity, expressed in POpeak and V.O2peak, improved during inpatient rehabilitation and V.O2peak improved even further up to 1 year after discharge. In the current study we underlined these findings by showing that POpeak and V.O2peak improved from start up to 5Y and from discharge up to 5Y. We did not find significant changes in POpeak and V.O2peak between 1 year and 5 years after discharge. This is in line with our hypothesis, based on our previous study (7) and with other small prospective studies, all performed with a shorter follow-up period after onset (3, 17, 19). Our findings are in contrast with the conclusions of Hoffman’s review (20), which, however, deals with older study material from a possibly different clinical area. Overall, it appears that persons with SCI in the Netherlands with a mean time since injury of 6.4 years, might be able to maintain stable wheelchair exercise capacity over time.
The mean wheelchair exercise capacity in our study group did not change during the 4-year interval from 1Y to 5Y, despite the observations by many authors that aerobic capacity declines with ageing in the able-bodied population (30), as well as in the SCI population (31). Nevertheless, in accordance with our study, Janssen et al. (19) found no decline in a 3 year follow-up of a group of SCI patients, and Shiba et al. (17) recently demonstrated almost no change in V.O2peak in approximately 20 years follow-up in a group of 7 subjects with SCI. The mean time after injury of 6.4 years in our study population may be too short to reflect an ageing decline effect, and the (increased) level of activity of the participants might have balanced the age-related decline in V.O2peak. Another explanation might be that the participants were allowed to use their own wheelchair, which might contribute to a higher wheelchair exercise efficiency.
Determinants
Gender and level of lesion were related to the level of POpeak and V.O2peak. Men show higher scores for wheelchair exercise capacity than women, and persons with paraplegia show higher scores than persons with tetraplegia; both findings conform with the results of previous studies (3, 4, 7, 11). In addition, as the literature reveals, lower scores were found for persons with complete lesions (V.O2peak) and for older persons (POpeak and V.O2peak) (3, 4, 11). In our previous study up to 1 year after discharge, completeness of lesion and age were not found as determinants of V.O2peak (7), which might have partially been caused by the loss to follow-up of older persons with SCI.
None of the personal and lesion characteristics were related to the course of wheelchair exercise capacity up to 5 years after discharge, which is partially in contrast with our previous study up to 1 year after discharge, in which gender and age were found to be determinants of the course of POpeak, and level of lesion was a determinant of the course of V.O2peak (7). Perhaps in the long term the beneficial circumstances, personality and coping strategies of the persons involved may be responsible for the maintenance of an active lifestyle (32) and indirectly to the maintenance of wheelchair exercise capacity, irrespective of the influence of age, gender and level of lesion (27).
Persons who attended no peak exercise test
The EXCLUDED group included older people and more subjects with tetraplegia. Both determinants ageing and severity of spinal cord lesion reduce functional outcome and are related to more secondary impairments (33, 34), reducing the chance of active involvement in the wheelchair exercise test. This, in turn, may lead to lower general participation and loss to follow-up (35–37). The loss to follow-up of relatively more persons with tetraplegia might have contributed to an overestimation at 1Y and 5Y of the level of the mean wheelchair exercise capacity in STUDY. It might be postulated that persons who were not able or willing to attend had a low level of physical fitness in the first place in comparison with the group that attended one or more tests.
Limitations and clinical implications of this study
Unfortunately, we had a high level of drop-out from the peak exercise test follow-up. The relatively large drop-out of old persons and of persons with a tetraplegia, who are prone to have lower physical fitness, might have resulted in an overestimation of wheelchair exercise capacity after SCI. However, our use of random coefficient analysis generated the best possible estimation of the course of exercise capacity during inpatient rehabilitation and the first 5 years after discharge.
In published data from our cohort a pattern of lower physical activity scales was found in the older and tetraplegic persons with a SCI (5). In order to prevent negative side-effects of a low exercise capacity in the long-term, persons with tetraplegia and older persons at onset of injury could be provided with intense and long medical supervision, as well as a structural follow-up rehabilitation opportunity in combination with exercise recommendations, such as low-intensity wheelchair training (38).
In random coefficient analysis, dichotomous variables add a significant degree of uncertainty to the outcomes compared with continuous variables. In our analysis only the variables age and change in peak exercise test are continuous, and this is a further limitation to this study (24).
Research recommendations
In order to unravel the complex long-term physical adaptation process of persons with SCI, the relations of wheelchair exercise capacity with the International Classification of Functioning Disability and Health domains activity level, participation, social support, general health and life satisfaction (39) need to be studied in more detail, and longitudinally, both in the early phase of rehabilitation and on the long-term after discharge. In particular, the influence of physical activity and having SCI-related secondary impairments as potential determinants of wheelchair exercise capacity, need to be studied in more detail in this cohort. In order to improve attendance for measurements, it may be advisable to include a less physically demanding submaximal exercise test, even though the maximal wheelchair exercise test is the gold standard.
CONCLUSION
The mean wheelchair exercise capacity performance of persons with SCI appears to stabilize between 1 year after discharge up to 5 years after discharge. However, because of the loss of the older persons and of persons with more severe lesions, the participating group appears to be a positive selection of the total study group.
AcknowledgEments
We thank the research assistants and physiatrists of the SCI units for collecting all the data and the following participating Dutch rehabilitation centres: De Hoogstraat Rehabilitation (Utrecht), Reade Centre for Rehabilitation and Rheumatology Amsterdam, Rehabilitation Center Het Roessingh (Enschede), Adelante (Hoensbroeck), Sint Maartenskliniek (Nijmegen), Rehabilitation Center Beatrixoord (Haren), Heliomare Rehabilitation Centre (Wijk aan Zee) and Rehabilitation Center Rijndam (Rotterdam).
References
