Anita Heeren, MD1,2, Mariëlle W. van Ooijen, MSc5,6, Alexander C. H. Geurts, MD, PhD1,2,3, Brian L. Day, DPhil4, Thomas W. J. Janssen, PhD5,6, Peter J. Beek, PhD5, Melvyn Roerdink, PhD5 and Vivian Weerdesteyn, PT, PhD1,2,3
From the 1Radboud University Medical Centre, Nijmegen Centre for Evidence Based Practice, Department of Rehabilitation, 2Sint Maartenskliniek, Centre for Rehabilitation, 3Sint Maartenskliniek Research, Development & Education, Nijmegen, The Netherlands, 4UCL Institute of Neurology, London, UK, 5MOVE Research Institute Amsterdam, Faculty of Human Movement Sciences, VU University Amsterdam and 6Amsterdam Rehabilitation Research Center | Reade, Amsterdam, The Netherlands
OBJECTIVE: To evaluate the concept of C-Mill gait adaptability training.
DESIGN: Pre- and post-intervention assessments.
SUBJECTS: Sixteen community-dwelling persons in the chronic phase after stroke (mean age 54.8 years).
METHODS: Participants received 10 sessions (1 h per session, for 5–6 weeks) of gait adaptability training on an instrumented treadmill augmented with visual targets and obstacles (C-Mill). Pre- and post-intervention assessments included: (i) clinical assessments of balance and gait: 10-m walking test, Timed Up-and-Go test, Berg Balance Scale, obstacle sub-task of the Emory Functional Ambulation Profile, and the Trunk Impairment Scale; (ii) physical activity level, assessed with a pedometer; (iii) success rate of accurate step adjustments towards a displacing target, assessed with an instrumented Target-Stepping Task; (iv) participant’s experience with the training.
RESULTS: All clinical assessments improved significantly after training (all p < 0.05), except for the Trunk Impairment Scale (p = 0.584). Physical activity increased by 19.6% (p < 0.05). Improvements in Target-Stepping Task success rates depended on the specific testing condition (time × body support × step direction, χ2(1) = 3.884, p < 0.05). All participants appreciated the training.
CONCLUSION: The concept of C-Mill gait adaptability training in the chronic phase after stroke is promising and warrants future research involving a randomized controlled trial.
Key words: stroke; gait; rehabilitation; exercise therapy; cues.
J Rehabil Med 2013; 45: 616–622
Guarantor’s address: Vivian Weerdesteyn, Radboud University Medical Centre, Department Rehabilitation, 898, PO Box 9101, 6500 HB Nijmegen, the Netherlands. E-mail: V.Weerdesteyn@reval.umcn.nl
Accepted Mar 7, 2013; Epub ahead of print Jun 24, 2013
Introduction
After stroke, 64% of survivors regain independent walking (1). However, post-stroke hemiplegic gait is often characterized by impaired standing balance on the paretic leg, reduced propulsion at paretic push-off, and decreased hip, knee and ankle flexion during the paretic swing phase resulting in inadequate foot clearance. These stroke-related balance and gait impairments are important risk factors for falling (2). In addition, community-dwelling people in the chronic phase after stroke most frequently mention walking as the activity associated with falling (2).
For independent and safe community ambulation, one needs to be able to adjust gait to environmental demands and constraints (e.g. while walking in crowded environments or on uneven terrain). Previous studies have demonstrated that gait adaptability is often impaired in people after stroke (3–9). This includes impairments in anticipatory control, as evidenced by placement of the affected lead foot closer to the obstacle before clearance (3), as well as impaired motor control of the leg to execute online step adjustments (e.g. more errors in avoiding a sudden obstacle) (4–6). Furthermore, people after stroke demonstrated different movement strategies, presumably to compensate for difficulties in balance control during the execution of a step adjustment (10). Gait adaptations impose large demands on balance control, and Nonnekes et al. (7) have demonstrated previously that reduced balance capacity in people after stroke significantly underlies their inability to perform accurate online step adjustments. In their study, participants stepped to a target that could suddenly jump medially or laterally, while the stepping foot was in the air, requiring a mid-step adjustment. This so-called Target-Stepping Task was executed both with and without external body support. People with stroke were found to suppress mid-step adjustments of foot placement when balance demands were greatest. Hence they sacrificed task performance for stability, whereas the healthy controls hardly ever needed to do so.
From the above, it follows that task-specific gait training focusing on gait adaptability may be an important target for improving safe community ambulation in people after stroke. Although there is convincing evidence that gait training in the chronic phase after stroke can improve walking velocity (11–16), only a few studies have specifically evaluated training of gait adaptability and associated dynamic balance demands (12, 15, 17), and none of these studies have evaluated training effects on pertinent gait adaptability and dynamic balance outcomes. Given that community walking is a frequent therapeutic target, this is surprising, particularly in light of a recent systematic review highlighting the importance of task specificity of exercise interventions (18). Nevertheless, a falls prevention programme focusing on gait adaptability in healthy elderly people improved obstacle-avoidance abilities (19). In people after stroke, there is preliminary evidence for the beneficial effects of gait adaptability training on walking speed (12, 15) and balance (17). These studies employed virtual reality to train gait adaptability, for example by presenting an obstacle in a virtual reality environment that needs to be crossed by making a step adjustment in the real environment. However, obstacle avoidance in virtual reality is distinct from obstacle avoidance in real environments, where the step adjustment can be directly monitored relative to the obstacle, allowing for accurate foot positioning relative to the real obstacle.
In the present study we used augmented reality to train gait adaptability. The benefit of augmented reality is that it allows for direct foot positioning relative to augmented objects presented on the walking surface; that is, in the real environment. In other words, with augmented reality, gait-environment interactions are direct (one can step directly onto a virtual obstacle), whereas with virtual reality, gait-environment interactions are indirect (step adjustments in the real world are detached from the obstacles in the virtual environment).
The aim of this pilot study was to evaluate this new augmented-reality concept of gait adaptability training in a group of community-dwelling persons in the chronic phase after stroke. A novel instrumented treadmill C-Mill (ForceLink, Culemborg, The Netherlands)1 (Fig. 1A) was used, in which the surface of the belt was augmented with visual context (targets, obstacles) via a projector. In addition to a direct interaction of foot placement and augmented visual context, the so-elicited gait adjustments continuously probe dynamic balance during walking. Hence, the implicit training of balance responses integrated into the actual, challenging gait tasks are a main asset of C-Mill gait adaptability training. We hypothesized that, after training, participants would demonstrate improvements in clinical balance and gait tests and physical activity, as well as more accurate step adjustments on the Target-Stepping Task. We further evaluated the feasibility of this training protocol from the participant’s perspective.

1Roerdink M, Beek PJ, inventors; ForceLink BV, assignee. Device for displaying target indications for foot movements to persons with a walking disorder. US2009246746-A1; EP2106779-A1; JP2009240775-A; NL1035236-C2. 2010.

Fig. 1. (A) C-Mill, showing a pattern of stepping targets projected on the belt. A computer is positioned in the desk next to the belt, which is coupled to the embedded force platform. The online-detected gait data are transformed by custom-made software into the visual context, and projected onto the belt’s surface by a wide-angle projector underneath the computer. (B–E) Various gait adaptability exercises on the C-Mill: (B) visually guided stepping; (C) bilateral obstacle avoidance; (D) speeding-up/slowing-down; (E) fun and functional game (see Appendix I for more details).
METHODS
Participants
A total of 16 persons after stroke who were referred to the participating centres for gait adaptability training by their rehabilitation physician between January and September 2011 were included in the study. Inclusion criteria were: (i) more than 6 months after first unilateral supratentorial stroke; and (ii) ability to walk independently (Functional Ambulation Categories 4–5 (20)). Exclusion criteria were: (i) other disorders influencing walking; (ii) serious cognitive impairments influencing comprehension of instructions; (iii) use of psychotropic medication; and (iv) serious visual impairments that limit the correct perception of the direct environment.
At an intake visit, a rehabilitation physician conducted a standardized medical history and performed a physical examination to check the inclusion and exclusion criteria and to record descriptive participant characteristics. Physical examination included Functional Ambulation Categories (20) (0–5), Motricity Index (20) (0–100), Fugl-Meyer Assessment of the lower extremity (21) (0–100%), assessment of muscle tone with the Modified Ashworth Scale (22) (0–4), assessment of the lowest perceived vibration in the lower extremities with the Rydel Seiffer tuning fork (Arno Barthelmes & Co., Tuttlingen, Germany) (23) (0–8) to assess deep sensibility, and visual confrontation tests for visuospatial hemi-neglect and visual field deficits (presented in Table I for the included participants). Finally, a familiarization session of the Target-Stepping Task (see below) was performed. Participants were instructed not to participate in other therapies aimed at improvement in balance or gait during the study period. All participants gave written informed consent. Study approval was obtained from the regional medical ethics committee.
|
Table I. Demographic and clinical characteristics of participants (n = 16) |
||
|
Characteristics |
mean (SD) [range] (unless noted otherwise) |
|
|
Age, years |
54.8 (10.8) [38–68] |
|
|
Gender, male/female, n |
9/7 |
|
|
Time post-stroke, months |
17 (11) [6–40] |
|
|
Type of stroke, ischaemic/haemorrhagic, n |
12/4 |
|
|
Hemisphere of stroke, right/left, n |
9/7 |
|
|
Functional Ambulation Category, 4/5, n |
2/14 |
|
|
Walking aid, n |
||
|
None |
12 |
|
|
Ankle-foot orthosis |
4 |
|
|
Cane |
1 |
|
|
Motricity Index Leg (0–100) |
81 (11) [57–100] |
|
|
Fugl Meyer Lower Extremity (0–100%) |
80 (10) [65–100] |
|
|
Modified Ashworth Scale (0–4)* |
||
|
Long hip adductors/short hip adductors |
0 (0–1)/0 (0–1) |
|
|
Knee flexors/extensors |
0.5 (0–1+)/0 (0–1+) |
|
|
Ankle plantar flexors/dorsiflexors |
1+ (0–2)/0 (0–1+) |
|
|
Quantitative Vibration threshold (0–8)* |
||
|
Interphalangeal joint hallux |
5 (0–7.5) |
|
|
Medial malleolus |
4.75 (0–7.5) |
|
|
Visuospatial hemi-neglect, n |
0 |
|
|
Hemianopia, n |
0 |
|
|
Values are noted as scores of paretic side. *Median [range]. SD: standard deviation. |
||
Intervention
Participants received 10 sessions of C-Mill gait adaptability training (1 h per session) over a period of 5–6 weeks. The embedded force platform of the C-Mill allowed for online gait event detection and presentation of augmented-reality on the belt’s surface, such as visual stepping targets and obstacles, attuned to the participant’s gait. Training sessions were performed at comfortable gait speed (as assessed in training sessions 1, 2 and 6) and participants were instructed not to use the handrails. Each training session comprised a predefined, sequence of 6 blocks of gait adaptability exercises. With exception of the first and last exercise block, the sequence of exercises in each training session is counterbalanced. The content and duration of the exercises was predefined in a protocol (see Appendix I). Training was given on an outpatient basis in two rehabilitation centres by physical therapists familiar with the C-Mill. Therapists progressively increased the difficulty level depending on the abilities of the participant.
Each training session started with a warm-up at comfortable walking speed (block 1). In block 2, visually guided stepping exercises were performed to practice foot positioning relative to a projected pattern of irregular stepping targets (Fig. 1B). In block 3, obstacles were presented on the belt’s surface, such that participants had to adjust their step length in order to avoid stepping on them (Fig. 1C). In block 4, so-called speeding-up and slowing-down exercises were performed using a projected target area that moved back and forth over the entire length of the treadmill at various, increasing rates of acceleration (Fig. 1D). In exercise block 5, target-stepping exercises that elicited tight-rope walking were performed, and to probe balance during walking even more, several targets suddenly turned into obstacles, requiring a step adjustment of an already planned step. Finally, each training session ended with a fun and functional gait adaptability game, that also combined visually guided stepping and obstacle avoidance, with immediate feedback on performance (Fig. 1E).
Pre- and post-intervention assessments
Pre-intervention (i.e. in the week prior to the first training) and post-intervention (i.e. in the week after the last training) assessments consisted of a set of complementary outcome measures. First, a series of clinical balance and gait tests were conducted (if necessary with orthosis, pre- and post-intervention): the 10-m walking test (20) at comfortable speed, the obstacle sub-task of the Emory Functional Ambulation Profile in which two real wooden obstacles (20 [l] × 10 [w] × 5 [h] cm) were placed in the walkway (24), the Timed Up-and-Go test (25), the Berg Balance Scale (26), the Trunk Impairment Scale 2.0 (27) and the 6-item Activities Specific Balance Confidence scale (28).
The second domain of outcome measures pertained to participants’ level of physical activity, which was monitored with a pedometer (Yamax Digiwalker; Yamax Health & Sports Inc., San Antonio, TX, USA). Participants wore the pedometer on their waist for 7 consecutive days to assess the number of steps taken per day, both in the week before and the week after the training (not including any assessments or training sessions).
The third outcome domain evaluated participants’ experience with and appreciation of C-Mill gait adaptability training after completing the intervention period. To this aim, an 11-item evaluation questionnaire was purpose-designed by the authors. This questionnaire evaluated satisfaction with duration and intensity, the perceived difficulty of the trained items and perceived training effects on confidence, gait speed and physical fitness. Free space was left for additional remarks.
Lastly, participants’ ability to make step adjustments during standing with and without balance support was assessed by means of an instrumented Target-Stepping Task (Fig. 2) (29). Participants stood barefoot (without orthosis) with their feet placed parallel in front of an electroluminescent panel (GeniuZ Netherlands BV, Kaatsheuvel, The Netherlands). Six stepping targets (30 [l] × 14 [w] cm) were embedded in the panel, situated on top of a dual-plate force platform (AMTI, Watertown, MA, USA). Every trial started with illumination of the target in front of the affected foot. Participants were instructed to step with their affected foot as accurately as possible on the illuminated target. However, the illuminated target could jump to either the adjacent medial or lateral side during the step and an adjustment of the ongoing step was required in order to land on the target. Target jumps were controlled by custom-written software (Matlab R2008a, Mathworks, Natick, MA, USA) and occurred 95 ms after the foot was lifted from the floor. After every trial, participants stepped back to the initial position indicated by chalk-marked footprints.
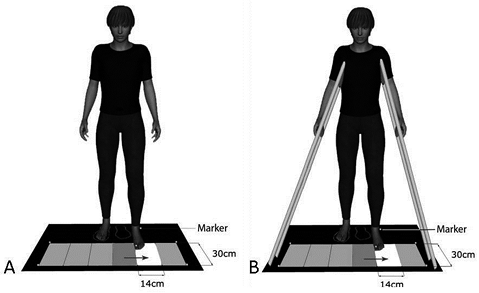
Fig. 2. Set-up of the Target-Stepping Task, demonstrating a lateral target jump (A) without support and (B) with the frame providing body support.
Both pre- and post-intervention, the Target-Stepping Task started with a series of 15 practice trials (6 jumps, 9 non-jumps) during which participants received auditory feedback on the intended step duration (450 ms). The step duration was standardized to minimize differences in accuracy as a result of varying stepping speeds. After these 15 practice trials, 2 test series of 40 experimental trials were conducted. Both test series contained 16 target jumps (8 medial, 8 lateral) and 24 non-jumping control trials, presented in random order. The first series was performed without balance support (Fig. 2A) and participants were instructed to step on the target with the affected foot and then place the unaffected foot alongside. In the second series, participants were supported by two large padded frames (Fig. 2B) and were instructed to step on the target with the affected leg, while keeping the stance foot in place. Stepping movements were recorded using an 8-camera 3D-motion registration system (Vicon, Oxford, UK) (sampling rate: 100 Hz) with 3 retro-reflective markers attached to the lateral malleolus, heel and hallux. During the Target-Stepping Task participants wore a safety harness fixed to the ceiling. Series were separated by a 10-min break.
|
Table II. Results of clinical tests pre- and post-intervention (n = 15) |
||||
|
Clinical tests |
Pre-intervention Mean (SD) |
Post-intervention Mean (SD) |
Differences pre- vs post-intervention |
95% Confidence Intervals |
|
10MWT, s |
9.55 (1.72) |
8.45 (1.36)** |
1.1 |
0.33 to 1.9 |
|
EFAP, s |
15.00 (3.19) |
13.10 (2.63)* |
1.9 |
0.40 to 3.4 |
|
TUG, s |
11.38 (1.97) |
9.81 (2.24)** |
1.5 |
0.52 to 2.6 |
|
TIS total, points |
11.4 (3.1) |
11.9 (1.9) |
–0.5 |
–1.9 to 0.89 |
|
BBS, pointsa |
53.6 (2.8) |
55.0 (1.4)* |
–1.4 |
–2.5 to –3.0 |
|
ABC, % |
56.5 (24.4) |
63.1 (17.0) |
–6.6 |
–19.2 to 6.1 |
|
Pedometer, steps/dayb |
5,738 (1,395) |
6,864 (1,645)* |
–1,126 |
–2,213 to –20 |
|
*p < 0.05, **p < 0.01. aAnalysed with Wilcoxon signed-rank test. bn = 11. SD: standard deviation; 10MWT: 10-m walking test; EFAP: Obstacle sub-task of the Emory Functional Ambulation Profile; TUG: Timed Up-and-Go task; TIS: Trunk Impairment Scale 2.0; BBS: Berg Balance Scale; ABC: 6-item Activities-specific Balance Confidence Scale. |
||||
Data analysis
Target-Stepping Task marker data were low-pass filtered (15 Hz) and processed in Matlab R2008a (Mathworks). Instants of foot-off and foot-landing were determined from filtered marker data combined with the ground reaction forces, yielding the step duration. Step adaptability was assessed by calculating foot placement errors in the jump trials. The foot placement error was calculated as the mediolateral distance from the centroid of the foot to the centre of the target. Foot placement errors were not normally distributed; 90.2% of the errors were either small (< 35 mm) or large (> 105 mm). Therefore, trials were considered successful when the absolute foot placement error was smaller than 35 mm, trials with foot placement errors larger than 35.0 mm were classified as failures. Success rates, defined as the percentage of successful target jump trials, were calculated for each participant and each condition.
Statistical analysis
To establish the effects of C-Mill gait adaptability training, paired-samples t-tests were used to compare pre- and post-intervention scores of clinical balance and gait tests and physical activity levels. The data of the Berg Balance Scale were not normally distributed and, therefore, Wilcoxon signed-rank test was used to compare its pre- and post-intervention scores. Target-Stepping Task success rates were analysed using a linear Generalized Estimated Equation analysis with “time” (pre-intervention/post-intervention), “support condition” (supported/unsupported) and “step direction” (medial/lateral) as within-subjects factors, and step duration as a covariate. We adopted the conservative generalized χ2 statistic in view of the small sample size. The level of significance (α) was set at 0.05. All statistical analyses were performed with SPSS 18 (SPSS Inc., IBM Corp., New York, NY, USA).
RESULTS
A total of 15 participants completed all 10 h of C-Mill gait adaptability training within 5–6 weeks. One participant dropped out because of an increase in lumbago. Three participants reported delayed-onset muscle soreness early in the training period. No other adverse events occurred. The distance walked per session increased significantly (p = 0.000) from 1.3 km (standard deviation; SD 0.4) at the first session to 2.0 km (SD 0.6) in the last training session of the protocol. The increase in distance was paralleled by a progressive increase in the difficulty of the gait adaptability training.
Clinical balance and gait tests
The results of the clinical balance and gait tests are summarized in Table II. The time to complete the 10-m walking test, obstacle sub-task of the Emory Functional Ambulation Profile and Timed Up-and-Go test all decreased significantly from pre- to post-intervention (–1.1, –1.9 and –1.5 s, respectively). The Berg Balance Scale showed a significant improvement of 1.4 points (p = 0.017), whereas post-intervention Trunk Impairment Scale and Activities-specific Balance Confidence scores did not differ from pre-intervention scores.
Physical activity level
Physical activity increased significantly after C-Mill gait adaptability training, by 1,126 steps/day (Table II; note that activity data of 4 participants were omitted; 2 because of a malfunctioning pedometer and 2 because of inconsistent reporting).
Participant’s perspective
Evaluation questionnaires indicated that all participants were enthusiastic about C-Mill gait adaptability training and would recommend it to peers. Most participants were satisfied with the duration of the training sessions (87%) and its intensity (80%), whereas 20% would have liked more sessions. Walking with a narrow base of support was rated as the most challenging element of C-Mill gait adaptability training (53%). All participants reported to have perceived an increase in confidence in walking abilities in daily life, both indoors (e.g. manoeuvrability) and outdoors (e.g. uneven terrain, walking in crowds). Furthermore, all participants experienced an increase in walking speed and fitness. Finally, 3 participants additionally mentioned generalization of training effects to other activities of daily life, such as cycling and walking stairs.
Target-Stepping Task
In total, 3.5% of the target-jump trials were omitted from analysis (e.g. registration errors, trials not performed as instructed; such as handrail support in an unsupported trial).
The Generalized Estimated Equation analysis yielded a significant “time” × ”support condition” × ”step direction” interaction effect on Target-Stepping Task success rates (χ2 (1) = 3.8, p = 0.049). Post-intervention, higher mean success rates were observed for both supported conditions (medial 9.7%, lateral 4.7%) as well as for the lateral unsupported condition (6.9%). In the medial unsupported condition, in contrast, success rates decreased by 5.9%. This is also shown in Fig. 3; the quartiles of improved conditions were reduced post-intervention.
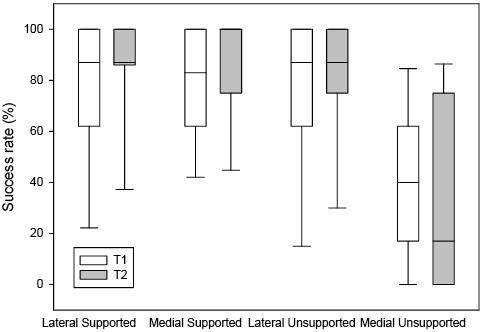
Fig. 3. Box-plot showing the pre-intervention (T1, white bars) and post-intervention (T2, grey bars) success rates of step adjustments on the Target-Stepping Task for all 4 conditions. Note the ceiling effect in the conditions in which participants improved. Furthermore, note that the median of the medial supported condition at T2 was 100%
Discussion
This study aimed to evaluate the concept of a new augmented-reality gait adaptability training, in which visual context was projected onto an instrumented treadmill (C-Mill) to trigger step adjustments. This was assessed in a group of 16 community-dwelling persons after stroke using a set of complementary outcome measures. The training was feasible, well tolerated and appreciated by the participants. Post-intervention, we observed improvements in clinical balance and gait tests and participants were physically more active. In addition, we found improved success rates (in all but one condition) on the Target-Stepping Task, which required online step adjustments during stance.
The improvements on clinical balance and gait tests were observed in the 10-m walking test, Timed Up-and-Go test, Berg Balance Scale, as well as in the obstacle sub-task of the Emory Functional Ambulation Profile. Gait speed (extracted from the 10-m walking test) improved from 1.08 ± 0.18 to 1.21 ± 0.18 m/s, which is sufficient for unrestricted participation (30). This improvement is comparable to walking speed gains observed after 4–26 weeks of non-body-weight-supported aerobic treadmill training (12–14, 31, 32). The presently observed improvements on the Timed Up-and-Go test are in line with the results of Silver et al. (32), who demonstrated that aerobic treadmill training in a similar population improved performance on a modified Timed Up-and-Go test. They attributed the increased performances on the modified Timed Up-and-Go test to the increased walking speed. Hence, gains in gait speed appear to be rather universal after relatively short periods of treadmill training.
Balance capacities were less frequently assessed to evaluate the effects of aerobic treadmill training. Due to the critical importance of balance control for accurate step adjustments, however, measures of balance capacity are expected to be more sensitive to reflect the specific nature of C-Mill gait adaptability training. Although improvements in Berg Balance Scale scores have been demonstrated previously after aerobic treadmill training (14), the presently observed improvements were obtained in a group of high-functioning participants with near-optimal Berg Balance Scale performance at baseline and after only 10 training sessions. In contrast, the study of Globas et al. involved an intensive programme with 39 sessions over a period of 3 months (14).
Improved balance performance has been demonstrated previously for short-duration (3–4 weeks) virtual-reality based treadmill training interventions in people in the chronic phase after stroke (17). Virtual-reality based treadmill training and the presently investigated intervention using augmented reality have important commonalities, in that they both require participants to adjust their gait in the real world in response to the virtual-reality or augmented-reality visual context, thereby implicitly challenging dynamic balance control while making step adjustments. These types of interventions may therefore be more effective than “traditional” aerobic treadmill training in improving the specific gait capacities required for safe community ambulation; namely the ability to adjust gait to environmental hazards on the walking surface.
The present study further adds to the body of literature on gait training in the chronic phase after stroke, by demonstrating that the C-Mill gait adaptability training intervention, which implicitly targeted dynamic balance during walking, proved to be responsive on both clinical and laboratory tests of dynamic balance and targeted stepping. We observed gains on the obstacle-avoidance sub-task of the Emory Functional Ambulation Profile that were greater than those observed in the 10-m walking test. This finding suggests that treadmill training with a focus on gait adaptability (including “real-virtual” obstacle avoidance) may also generalize to real obstacle negotiation, despite the fact that toe clearance (as an important parameter of obstacle crossing) was not specifically trained.
Furthermore, improved accuracy of online step adjustments during standing was observed on the Target-Stepping Task. We suggest that the higher success rates post-intervention in both supported conditions (lateral and medial adjustments) indicate training-related amelioration of the motor control of the paretic leg. In the unsupported conditions, improvements were observed only for lateral step adjustments, whereas the performance on medial step adjustments became slightly less accurate. Hence, when balance demands were added to the task, the participants only benefited from improvements of leg motor control in lateral step adjustments. The lack of improvement in medial unsupported steps may indicate that this condition is too difficult for people after stroke. Indeed, our participants were unsuccessful in the vast majority of the trials (comparable to Nonnekes et al. (7)). The difficulties in executing medial step adjustments are probably related to the fact that it is almost impossible to implement mid-step medial adjustments of the centre of mass trajectory prior to foot landing (33). For persons after stroke, whose balance problems are most prominent in the frontal plane (34), the imposed narrow base of support at foot landing after medial steps further challenges their balance. It has been suggested that these persons may suppress medial step adjustments in anticipation of the unstable end position, thereby sacrificing task accuracy for stability (7). Considering the high balance demands associated with unsupported medial adjustments, the slight decrement in performance post-intervention might therefore indicate that participants became more aware of their limitations and tended to rely on a safer strategy (i.e. a wider base of support). Note, however, that people rarely take medial steps under more natural circumstances that offer a choice of potential foot landing positions (35).
Study limitations
A limitation of the present study was that we did not include a control group. Hence, it cannot be excluded that similar results could be achieved with an intervention of equal intensity, but of a different modality (e.g. aerobic treadmill training, circuit class training or virtual reality gait training).
Although participants were familiarized with the Target-Stepping Task at baseline (i.e. 1 week before the pre-intervention assessment), we cannot rule out the possibility that differences between the pre-intervention and post-intervention assessments may be (partly) caused by the effects of learning on the task. However, it seems unlikely that a (second) exposure to a measurement of approximately 30 min would greatly influence re-test performance 7 weeks later. Future studies involving a control group are needed to provide conclusive evidence.
Another interesting observation was the increase in physical activity following the intervention. Physical activity levels have been demonstrated to be related to balance and mobility performance in community-dwelling individuals after stroke, with walking speed being a strong predictor (36). Hence, the presently observed improvement in physical functioning may have enabled the participants to become more physically active in daily life. However, it remains for future studies to determine whether this increase reflects improved participation in daily life. Furthermore, it must be pointed out that pedometers tend to undercount the number of steps in people with gait disorders, and this is particularly true for those with low walking speeds (37). Hence, it cannot be excluded that the increase in the number of steps measured may have been an artefact of the higher post-intervention gait speed. This possibility, however, does not seem very likely, as the average gait speeds at baseline (1.08 m/s) and follow-up (1.21 m/s) were within the range of speeds with less than 10% known registration inaccuracy (38) for the pedometers used in the present study.
Finally, inconsistent results were obtained for confidence. On the evaluation questionnaire, all participants reported having perceived improved confidence, which was not confirmed by the Activities-specific Balance Confidence scores. It is possible that the perceived improvements in confidence were too gait-specific to be captured by the Activities-specific Balance Confidence scale, which also pertains other activities of daily living than walking.
In conclusion, this pilot study evaluated the concept of C-Mill gait adaptability training in community-dwelling individuals after stroke. This augmented-reality intervention was feasible and well accepted by the participants. Clinical balance and gait tests, physical activity level, and the ability to make step adjustments during standing with and without balance support, were all responsive outcome parameters. These findings are promising with respect to improvement in community ambulation, and warrant future research, preferably involving a randomized controlled trial contrasting C-Mill gait adaptability training with another training modality (e.g. aerobic treadmill training, circuit class training, virtual reality gait training). The relative efficacy of the training interventions should then not only be determined directly following the training period, but also with follow-up assessments to examine whether the training effects, and potential differences between interventions, prevail.
ACKNOWLEDGEMENTS
The authors would like to thank Judith Vloothuis, rehabilitation physician at Reade, Centre for Rehabilitation and Rheumatology, for conducting the intake visits. Futhermore, they thank the physiotherapists Jasper den Boer, Peter Elich, Sifra Broeder and Sjoerd Heubers for their feedback on the training protocol. From the department of rehabilitation medicine at the Radboud University they thank Coen Bongers, Digna de Kam and Jorik Nonnekes for their help in the data collection and Roland Loeffen, Mark de Niet and Roos van Swigchem for their technical support.
This project was funded by the Dutch Brain Foundation (Hersenstichting Nederland) Grant 2010(1)-25.
MR and VW are financially supported by the Netherlands Organisation for Scientific Research (NWO Veni grants 451.09.024 and 916.10.106, respectively). ForceLink (producer of the C-Mill) was not involved in this study, financially or otherwise, and had no influence on the interpretation of data and the final conclusions.
REFERENCES
Appendix I. Training protocol | ||
Block | Contents | Duration (min) |
1 | Warming up at comfortable walking speed | 5 |
2 | Visually-guided stepping: continuous projection of a pattern of stepping targets of varying stride length, step width, step-length asymmetry and regularity. Difficulty increased by changing the irregularity in the sequence of stepping targets (Fig. 1B) | 7 |
3 | Obstacle avoidance: projection of unilateral or bilateral obstacles. Task difficulty was manipulated by changing the obstacle’s size and available response time (Fig. 1C) | 7 |
4 | Speeding-up and slowing-down: projection of a target area that moved back and forth over the entire length of the treadmill at various, increasing rates of acceleration (Fig. 1D) | 7 |
5 | Tight-rope walking: projection of a pattern of stepping targets with a narrow base of support while several targets turned unpredictably into obstacles. The number of targets turning into obstacles increased, in parallel with decreasing reaction times | 7 |
6 | Fun and functional game: projection of targets (e.g. footballs) and obstacles (e.g. puddles of water). Participants received points for successful target hits, but lost points when hitting obstacles. Points appeared immediately on the belt’s surface, accompanied by visual and auditory feedback on performance. Available in 3 difficulty levels, which varied in the number of augmented-reality objects per metre (Fig. 1E) | 7 |
