Jens Bak Sommer, MSc1,2,3, Anne Norup, PhD1, Ingrid Poulsen, PhD1 and Jesper Mogensen, PhD3
From the 1Research Unit on BRain Injury rehabilitation, Copenhagen (RUBRIC), Department of Neurorehabilitation, Traumatic Brain Injury Unit, Copenhagen, University Hospital, Glostrup, 2Department of Drug Design and Pharmacology and 3The Unit for Cognitive Neuroscience, Department of Psychology, University of Copenhagen, Denmark
OBJECTIVE: To examine cognitive activity limitations and predictors of outcome 1 year post-trauma in patients admitted to sub-acute rehabilitation after severe traumatic brain injury.
SUBJECTS: The study included 119 patients with severe traumatic brain injury admitted to centralized sub-acute rehabilitation in the Eastern part of Denmark during a 5-year period from 2005 to 2009.
METHODS: Level of consciousness was assessed consecutively during rehabilitation and at 1 year post-trauma. Severity of traumatic brain injury was classified according to duration of post-traumatic amnesia. The cognitive subscale of Functional Independence MeasureTM (Cog-FIM) was used to assess cognitive activity limitations. Multivariate logistic regression analyses were performed to identify predictors of an independent level of functioning.
RESULTS: The majority of patients progressed to a post-confusional level of consciousness during the first year post-trauma. At follow-up 33–58% of patients had achieved functional independence within the cognitive domains on the Cog-FIM. Socio-economic status, duration of acute care and post-traumatic amnesia were significant predictors of outcome.
CONCLUSION: Substantial recovery was documented among patients with severe traumatic brain injury during the first year post-trauma. The results of the current study suggest that absence of consciousness at discharge from acute care should not preclude patients from being referred to specialized sub-acute rehabilitation.
Key words: traumatic brain injury; TBI; acute brain injury; vegetative state; minimally conscious state; confusional state; consciousness disorders; rehabilitation outcome.
J Rehabil Med 2013; 45: 749–755
Correspondence address: Jens Bak Sommer, Department of Drug Design and Pharmacology, Faculty of Health and Medical Sciences, University of Copenhagen, Universitetsparken 2, DK-2100 Copenhagen, Denmark. E-mail: jens.sommer@sund.ku.dk
Accepted June 13, 2013
Introduction
Advances in neurocritical care have led to a significant reduction in mortality after severe traumatic brain injury (TBI) (1), and despite national differences TBI mortality rates have declined substantially in the Nordic region (2). As a result, an increasing proportion of patients with the most severe injuries now survives TBI and is subsequently in need of specialized treatment and rehabilitation (3). From a Danish perspective, an epidemiological study has shown that the number of patients admitted to hospital for more than 3 months after TBI has increased by 64% from 1994 to 2002 (4).
Some of these patients remain in a state of severely disordered consciousness, such as coma, vegetative state (VS) (5), also known as unresponsive wakefulness syndrome (UWS) (6) and minimally conscious state (MCS) (7), for an indeterminate period of time post-injury.
Treatment and rehabilitation of patients with post-traumatic disorders of consciousness (DOC) have long been regarded with scepticism and nihilism concerning prognosis and outcome, which may be ascribed to the fact that loss of consciousness in these disorders has often been considered irreversible and similar to the end-stage of severe degenerative disorders (8). Furthermore, many patients with DOC have been deemed ineligible for rehabilitation as they have not been able to participate actively in standard rehabilitation interventions, and only a few specialized facilities have existed for this group of patients (9–11).
Despite significant progress in the understanding of pathophysiology and assessment (12, 13), knowledge of DOC among the general medical community still appears to be limited, even among professionals who are most likely to be responsible for the rehabilitation of patients with acquired brain injury. According to a Swedish survey from 2011, encompassing more than 1,000 physicians working within acute or rehabilitation care of patients with brain injury, only approximately half of the responding physicians knew the definition of VS/UWS, and nearly a quarter of respondents thought that patients in VS/UWS should never be admitted to rehabilitation or should be given lower priority than other patients (14). This is, moreover, complicated by the fact that misdiagnosis of VS/UWS has been found to be common, with studies estimating that evidence of consciousness is missed in approximately 40% of these patients (15).
The exact epidemiology of VS/UWS is not known; however, the prevalence of the condition in hospital cases in Europe is estimated to be approximately 0.5–2 per 100,000 of the population per year (16). VS/UWS is thus a relatively rare condition, and limited knowledge of VS/UWS in the medical community may be ascribed to the fact that few physicians have had direct clinical experience with such patients (17). In addition, treatment of patients is often distributed over a broad range of institutions limiting specialization and accumulation of expertise among healthcare professionals (10, 14).
To improve the assessment and treatment of severe TBI and DOC in Denmark, early sub-acute rehabilitation of these patient groups was centralized to 2 specialized hospital units in the year 2000 (18). Thus, a continuous chain of care from accident site to trauma centre and to centralized specialized rehabilitation has been established for most of these patients in Denmark. Analogous centralized treatment programs for patients with severe TBI have subsequently been implemented or proposed in other Nordic countries (14, 19, 20).
Centralization of sub-acute rehabilitation has allowed for the prospective collection of data regarding demography, progress and outcome of patients with DOC and severe TBI in Denmark (18). The systematic registration of data during early rehabilitation may contribute to better prediction modelling, which may be of value in rehabilitation planning, counselling of relatives, and the identification of targets for intervention trials (3). As data now are available for larger cohorts of patients with DOC and severe TBI, and recent research suggests that further knowledge about the rehabilitation potential and long-term outcome of this patient population is strongly needed (14), the authors decided to undertake the current study.
The purpose of the study was to examine cognitive activity limitations 1 year post-trauma in patients with DOC and severe TBI admitted to sub-acute rehabilitation in the Eastern part of Denmark. Furthermore, demographic and clinical predictors of an independent level of functioning were investigated.
Methods
Participants
Data were analyzed for patients consecutively admitted to the TBI Unit, Department of Neurorehabilitation, Copenhagen University Hospital, Glostrup, Denmark.
The TBI Unit is a highly specialized sub-acute rehabilitation unit that receives patients with severe TBI early after injury. The unit has an uptake area of approximately 2.5 million inhabitants covering the Eastern part of Denmark as well as the Faroe Islands and Greenland. At referral to the unit, highest priority is given to patients with a Glasgow Coma Scale score (GCS) (21) in the range 3–9 one day after cessation of sedation. Patients with a higher GCS score may also be admitted to the unit provided that severe focal neurological deficits, severe cognitive disorders and/or pronounced agitation are present. The rehabilitation regimen has been described elsewhere (18).
As a standard procedure, demographic and clinical data are prospectively registered for all patients admitted to the TBI Unit. The database of the unit is approved by the Danish Data Protection Agency. For the current study, we considered patients admitted during a 5-year period from 2005 to 2009. Inclusion criteria were: (i) TBI as cause of admission; (ii) a minimum age of 16 years; and (iii) participation in a follow-up assessment 1 year post-trauma. Patients with comorbidities that could interfere with the assessment of TBI-related cognitive disability were excluded, including patients with: (i) congenital or previously acquired brain injury; (ii) neurodegenerative disorders; and (iii) psychiatric disorder or substance abuse affecting daily functioning at time of injury. Individuals with no follow-up data and/or with missing data about comorbid disorders and cognitive disability prior to injury were also excluded.
Measures
Demographic variables. Age, sex and socio-economic status (SES) were registered at admission to the TBI Unit. SES was rated on a 5-level ordinal scale adopted from the Danish Head Trauma Database, a national clinical database for patients with head trauma. Level of SES was classified according to occupational achievement in combination with educational level and number of sub-ordinates in the workplace. In the statistical analyses, the 5-level scale was dichotomized into lower (level 4–5) and higher SES (level 1–3). Persons, who had never had a job or completed formal education, were assigned the lowest level of SES.
Duration of acute care. The duration of acute care was measured as the number of days from trauma to admission to the TBI Unit. During this period patients were primarily admitted to neurosurgical wards and intensive care units.
Post-traumatic amnesia (PTA). The duration of PTA was measured prospectively by neuropsychologists with Galveston Orientation and Amnesia Test (GOAT) (22). GOAT is a 10-item questionnaire that assesses orientation and memory after TBI. A score of ≥ 76 on 2 consecutive ratings marks the resolution of PTA. The duration of PTA was calculated as the number of days from trauma to the day criteria was met on GOAT. A minor proportion of the patients had not cleared PTA at discharge, and in these cases duration of PTA was estimated retrospectively at follow-up.
Level of consciousness (LOC). LOC was assessed prospectively by neuropsychologists from admission to discharge and at follow-up with the Rancho Los Amigos Levels of Cognitive Functioning Scale (RLA) (23). RLA is an ordinal scale comprising the following 8 levels: No response (RLA 1); Generalized response (RLA 2); Localized response (RLA 3); Confused – agitated (RLA 4); Confused – inappropriate (RLA 5); Confused – appropriate (RLA 6); Automatic – appropriate (RLA 7); Purposeful – appropriate (RLA 8). For descriptive purposes, the RLA was collapsed into 5 categories consisting of coma (RLA 1), VS/UWS (RLA 2), MCS (RLA 3), confusional state (RLA 4–6) and post-confusional state (RLA 7–8). These categories describe the stages of recovery typically seen following TBI in a more traditional neurological nomenclature (11, 24). MCS was assessed in accordance with the diagnostic criteria of the Aspen Workgroup (7), and patients fulfilling these criteria were categorized as RLA 3. LOC at admission was based on several neuropsychological assessments performed over the first days of admission and corroborated by behavioral observations from the interdisciplinary rehabilitation team. At follow-up a neuropsychological assessment of LOC was performed as part of an interdisciplinary examination. Family members or other care providers familiar with the patient were invited to participate in these examinations.
Cognitive activity limitations. The Cognition subscale of Functional Independence Measure (FIMTM) (25) (Cog-FIM) was used to evaluate limitations in cognitive activities. Cog-FIM includes 5 items that assess functional independence within the domains of comprehension, expression, social interaction, problem-solving and memory. Each item is rated on a 7-point scale from “total assistance” to “complete independence”. The Cog-FIM total score ranges from 5 to 35, with higher scores indicating greater independence. In the analyses scores on each item were dichotomized into a dependent (scores 1–5) vs an independent cognitive activity level (scores 6–7). These are the 2 broad levels of functioning recognized by the scale defined by whether help from another person is required for performance of the task in question. The interdisciplinary team of each patient consisting of nurses, physio- and occupational therapists rated patients consecutively with Cog-FIM from admission to discharge and at follow-up. Nurses and therapists were trained in rating Cog-FIM, and the department is certified for using FIMTM by the Uniform Data Set for Medical Rehabilitation (UDSMR). Cog-FIM has been recommended as a core measure of cognitive activity limitations by the TBI Outcomes Workgroup (26).
Data analysis
Descriptive data are presented as 25th, 50th and 75th percentiles, ranges and percentages when appropriate. Kruskal-Wallis tests (H) and Mann-Whitney tests (U) were used to compare differences between subgroups of patients in scores on Cog-FIM and on continuous variables (age, duration of acute care and PTA) when parametric assumptions were not met. Bivariate correlations were calculated with Spearman’s rho (rs). Pearson’s χ2 and Fisher’s exact test were used to evaluate differences in categorical variables.
Direct multiple logistic regression was conducted to identify predictors of an independent cognitive activity level within each domain on Cog-FIM at follow-up. Analyses were performed to ensure that the assumption of linearity of the logit was not violated. Multicollinearity was found to be insignificant, and the number of expected frequencies was adequate in all cells. Multivariate outliers in the regression solutions were identified by examining residuals and deviance statistics. Three patients with extreme values were identified, however analyses indicated that they neither had erroneous scores nor exerted an undue influence on regression coefficients, and consequently all of these patients were included in the final models.
A univariate attrition analysis was conducted to investigate systematic bias between patients with and without follow-up as well as between the total group of included and excluded patients.
All statistical tests were 2-sided and considered significant at p < 0.05. In case of multiple comparisons the significance level was adjusted with Bonferroni corrections. All analyses were conducted with the statistical software package PASW Statistics for Windows, version 18 (SPSS Inc., Chicago, IL, USA).
Results
Study population
From 2005 to 2009 a total of 207 patients over the age of 16 years were admitted to the unit with a TBI diagnosis, and of these patients 157 (75.8%) participated in a 1 year follow-up examination. Reasons for patients being lost to follow-up were mortality (n = 17), refusal to participate (n = 3) and migration (n = 6). For the remainder of patients (n = 24) causes were unknown.
Due to previous brain injury and cognitive disability 35 (22.3%) patients with follow-up data were excluded from the study, and another 3 patients were excluded due to missing data on 1 or more predictor variables. Hence, a total of 119 patients (57.5%) fulfilled the inclusion criteria and were included in the statistical analyses.
Univariate analyses were conducted to investigate whether patients with follow-up differed systematically from patients without follow-up on study variables. Patients without follow-up were found to be significantly older (median 53.6 years) than patients with follow-up data (median 41.2 years, U = 2703.00, p = 0.001). Moreover, the former group had significantly lower scores on Cog-FIM at discharge from the TBI Unit (median = 7.0) compared with the latter (median = 23.0, U = 1519.00, p < 0.001). No differences were found concerning gender, SES, proportion of patients in VS/UWS at admission to rehabilitation or duration of acute care and PTA. The same differences were found when the total group of excluded patients was compared with included patients.
In the study group, Cog-FIM total scores at follow-up were significantly correlated with total scores at discharge from the TBI Unit (rs = 0.83, p < 0.001) and patient age (rs = –0.26, p = 0.005).
Table I provides demographic data and injury-related information for patients included in the study. Patients were primarily male (70.6%) and approximately one-quarter (26.8%) was 25 years old or younger. The vast majority of patients (89.9%) was admitted to the TBI Unit within 1 month post-trauma, and at admission to rehabilitation almost half of patients (48.7%) were still in VS/UWS or MCS (Fig. 1). Based on duration of PTA, 103 patients (86.6%) were classified as having extremely severe TBI (PTA > 4 weeks), whereas the remaining 16 patients (13.4%) had very severe TBI (PTA = 1–4 weeks) (27).
|
Table I. Demographic and injury data (n = 119) |
||
|
Characteristics |
25th/50th/75th percentiles |
Range |
|
Age, years |
24/39/54 |
16–78 |
|
Acute care stay, days |
12/18/22 |
6–59 |
|
Rehabilitation stay, days |
74/113/182 |
25–349 |
|
Cog-FIM at admission, total |
5/5/6 |
5–34 |
|
RLA at admission |
3/4/5 |
2–8 |
|
PTA, days |
32/64/160 |
12–365 |
|
Trauma to follow-up, months |
13/13/15 |
11–23 |
|
VS/UWS at admission, % |
23.5 |
|
|
Lower SES, % |
72.3 |
|
|
Sex, male, % |
70.6 |
|
|
Cause of injury, % |
||
|
Vehicular |
72.3 |
|
|
Fall |
20.2 |
|
|
Other |
7.5 |
|
|
RLA: score on Rancho Los Amigos Levels of Cognitive Functioning Scale; PTA: duration of post-traumatic amnesia; SES: socio-economic status; VS/UWS: vegetative state/unresponsive wakefulness syndrome; Cog-FIM: Cognitive subscale of Functional Independence MeasureTM. |
||
Level of consciousness and cognitive outcome 1 year post-trauma
The median time from trauma to follow-up was 13.1 months. LOC at follow-up is depicted in Fig. 1 for the 117 patients who were assessed with RLA. The vast majority of patients (84.6%) had progressed to a post-confusional state (PCS). Of the remainder, no patients were in VS/UWS, whereas 5 were in MCS (4.3%) and 13 (11.1%) in a confusional state (CS).
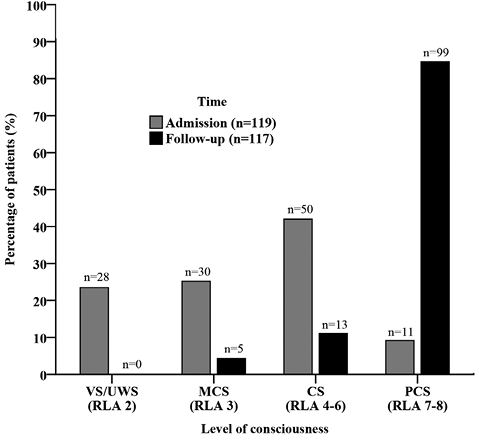
Fig. 1. Level of consciousness at admission to rehabilitation and at follow-up 1 year post-trauma. VS/UWS: vegetative state/unresponsive wakefulness syndrome; MCS: minimally conscious state; CS: confusional state; PCS: post-confusional state.
In the study group, median total scores on Cog-FIM improved from 5 at admission to 27 at follow-up (Fig. 2). Moreover, 6 patients (5%) had achieved the maximum score of 35 indicating complete independence within all of the assessed cognitive domains. A significant association was found between LOC at admission to rehabilitation and total scores on Cog-FIM 1 year post-trauma (H(3) = 28.80, p < 0.001). Post-hoc analyses with Bonferroni correction of the significance level to 0.017 indicated that patients admitted in VS/UWS had significantly lower scores than patients with MCS (U = 203.50, p = 0.001) or a higher LOC upon admission. However, outcomes for patients admitted in VS/UWS were extremely varied, with Cog-FIM total scores at follow-up ranging from 5 to 33.
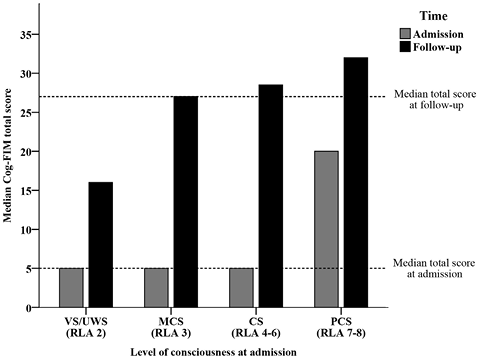
Fig. 2. Cognitive subscale of Functional Independence MeasureTM (Cog-FIM) total scores at admission to rehabilitation and at follow-up 1 year post-trauma according to level of consciousness at admission (n = 119). VS/UWS: vegetative state/unresponsive wakefulness syndrome; MCS: minimally conscious state; CS: confusional state; PCS: post-confusional state.
Predictors of an independent cognitive activity level
The proportion of patients that achieved an independent cognitive activity level at follow-up varied from 32.8% to 58.0% between the 5 domains on Cog-FIM (Fig. 3).
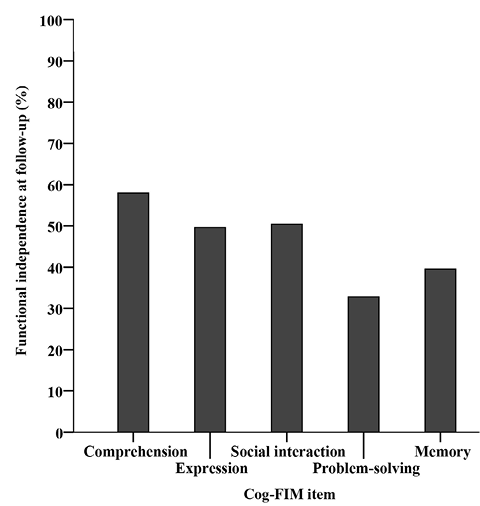
Fig. 3. Percentage of patients with an independent cognitive activity level within each Cognitive subscale of Functional Independence MeasureTM (Cog-FIM) domain 1 year post-trauma (n = 119).
Omnibus tests indicated that each of the 5 logistic regression models were statistically significant (p < 0.001 for all models), and outcome was in total predicted correctly for 76.5–81.5% of patients. Odds ratios (OR) for predictors within each domain on Cog-FIM are shown in Table II.
|
Table II. Odds ratios (OR) for an independent cognitive activity level one year post-trauma. Significant odds ratios (OR) for each predictor and domain on Cognitive subscale of Functional Independence MeasureTM (Cog-FIM) are depicted below in bold. An OR of less than 1 indicates a reduced probability of functional independence. Confidence intervals for significant predictors are presented in the text |
|||||
|
n = 119 |
Comprehension |
Expression |
Social interaction |
Problem-solving |
Memory |
|
Age |
0.974 |
0.966 |
0.976 |
0.981 |
0.997 |
|
Sex (male) |
0.593 |
0.499 |
0.546 |
0.723 |
0.392 |
|
Lower SES |
0.590 |
0.937 |
0.607 |
0.287* |
0.292* |
|
Acute care |
0.968 |
0.913* |
0.966 |
0.943 |
0.970 |
|
VS/UWSa |
0.823 |
2.395 |
0.488 |
0.886 |
1.023 |
|
PTA |
0.982*** |
0.977*** |
0.988** |
0.979** |
0.977** |
|
*p < 0.05, **p < 0.01, ***p < 0.001. aVS/UWS at admission to rehabilitation. RLA: score on Rancho Los Amigos Levels of Cognitive Functioning Scale; PTA: duration of post-traumatic amnesia; SES: socio-economic status; VS/UWS: vegetative state/unresponsive wakefulness syndrome. |
|||||
A longer duration of PTA was a significant negative predictor of an independent cognitive activity level within all domains on Cog-FIM (OR = 0.977–0.988, 95% confidence interval (95% CI) = 0.963–0.996). Lower SES was a significant negative predictor within the domains of problem-solving (OR = 0.287, 95% CI = 0.086–0.953) and memory (OR = 0.292, 95% CI = 0.087–0.982), while a longer duration of acute care was associated with a reduced probability of functional independence with regard to expression (OR = 0.913, 95% CI = 0.845–0.985).
Discussion
A substantial proportion of patients with severe TBI and prolonged DOC was found to recover during the first year post-trauma. At admission, almost half of patients in the study group were in VS/UWS or MCS. However, at follow-up 1 year post-injury no patients were in VS/UWS and only 5 were in MCS. Moreover, 32.8% to 58.0% of the total number of patients had achieved functional independence within the 5 domains on Cog-FIM. SES, duration of PTA and acute care were significant predictors of cognitive activity limitations 1 year post-trauma with PTA being the only predictor that was significant across all domains.
Cognitive outcome 1 year post-trauma
As emergence from VS/UWS more than 1 year after TBI has been found to be infrequent and associated with severe residual disability (28, 29), a particularly encouraging finding in the current study was that no patients were in VS/UWS at follow-up. However, patients admitted in VS/UWS did have significantly lower scores on Cog-FIM at follow-up compared with patients in MCS or with a higher LOC upon admission to rehabilitation. Nevertheless, during the first year post-trauma median scores improved from 5 to 16 among patients admitted in VS/UWS, and outcomes at follow-up were extremely varied with scores on Cog-FIM ranging from total assistance to modified and complete independence.
Hence, the results of the present study indicate that VS/UWS at admission to rehabilitation is not uniformly associated with a poor cognitive outcome, and VS/UWS was, moreover, not a significant predictor of cognitive activity limitations 1 year post-trauma in any of the multivariate logistic regression models. In agreement with previous research (11, 30), the current study accordingly suggests that absence of consciousness at discharge from acute care should not preclude patients from being referred to specialized sub-acute rehabilitation. Furthermore, behavioral evidence of consciousness may be missed in the acute care setting due to a short duration of stay, sedation and limited access to specialized assessment methods (31), and referral to a specialized rehabilitation facility may thus be pivotal to a proper diagnosis and patient management.
At follow-up 5 patients were still in MCS in the study cohort; however, compared with recovery from VS/UWS recovery from MCS 1 year or longer after brain injury has been found to be more frequent. Luauté et al. (29) reported that approximately one-third of patients remaining in MCS 1 year after traumatic or non-traumatic brain injury exhibited functional improvement during the following 4 years. In this regard studies have shown some inconsistencies regarding the prognostic value of duration of MCS. Lammi et al. (32) did not find any significant associations between duration of MCS and most measures of functional and psychosocial outcome 2–5 years after TBI. However, as noted by the authors a small sample size may have attenuated statistical relationships, and in a larger cohort of patients Katz et al. (11) reported that duration of MCS and age were significant predictors of level of disability 1 year post-trauma. Interestingly, a relationship between duration of VS/UWS and the probability of emerging from MCS after traumatic and non-traumatic brain injury was found in the latter study. Out of a total of 23 patients with a follow-up period of at least 1 year, 5 failed to emerge from MCS, and all of these patients had been in VS/UWS for more than 8 weeks. Unfortunately, it was not possible to determine the duration of VS/UWS for the 5 patients in our sample who remained in MCS at follow-up. However, it seems reasonable to expect that at least some of these patients will improve over the months and years to come.
Previous research has suggested that Cog-FIM may be of limited value in the measurement of long-term outcome of patients with TBI due to a ceiling effect (33). In contrast, only 5% of patients had obtained the maximum score of 35 on Cog-FIM 1 year post-trauma in our study, which may be explained by the fact that only patients with the most severe TBI were included. In a similar manner, Hammond et al. (34) found that merely 16% of patients with moderate to severe TBI were at ceiling on Cog-FIM 1 year post-trauma. Altogether, this suggests that Cog-FIM may have utility as a measure of long-term cognitive activity limitations after more severe injuries, whereas ceiling effects may be a concern in studies primarily including patients with mild to moderate TBI.
The median Cog-FIM total score at follow-up in the present study is comparable to the results of a recent investigation of the longitudinal outcome of patients with DOC (30). In that study a median score of 25 was reported 1 year after TBI, and, interestingly, significant change in Cog-FIM total scores was seen from 1 to 5 years post-injury. During the same time interval Hammond et al. (34) also noted that scores only remained stable in 61% of patients with TBI, which was explained by improvement as well as decline in the functional level of patients. Thus considerable change is to be expected beyond 1 year post-trauma, and consequently the follow-up period in the present study may be too short to document long-term cognitive activity limitations.
An important question is to what extent the observed recovery may be ascribed to the administered rehabilitation intervention. Previous research indicates that the centralization of sub-acute rehabilitation of patients with severe TBI in Denmark has resulted in better rehabilitation outcomes (35). When data from an earlier cohort of patients admitted to the TBI Unit were compared with historical data from patients treated before centralization, significantly better outcomes were found at discharge in the former group for equal injury severity and length of stay. In addition, a recent prospective study from Norway indicates that an early comprehensive rehabilitation intervention does improve functional outcomes 1 year after severe TBI compared with a delayed and broken chain of treatment (19). Collectively, this suggests that centralized sub-acute rehabilitation may contribute to better outcomes after severe TBI. Nevertheless, the current study cannot make any conclusive claims concerning the effects of rehabilitation on recovery, as such analyses would require a different research design and a comparison with patients treated in alternative settings.
Predictors of cognitive activity limitations
A longer duration of PTA was consistently found to be associated with a reduced probability of an independent cognitive activity level, and across Cog-FIM domains odds of functional independence were reduced by approximately 1–2% with each additional day of PTA. This is not surprising considering that PTA has also been found to be a predictor of Cog-FIM total scores at discharge from acute care (36) and inpatient rehabilitation (37) after mild to severe TBI. Duration of PTA is positively correlated with the volume of grey and white matter lesions on MRI, and may thus reflect the extent of organic brain damage after TBI (38).
SES and duration of acute care were significant predictors of outcome within specific cognitive domains. Higher SES was associated with increased odds of functional independence concerning problem-solving, and memory. SES may be considered an indicator of pre-morbid cognitive reserve (39) and was tentatively included as a measure of such. However, a crude dichotomous variable was used in the present study, and as confidence intervals for the calculated odds ratios were very large, results are to be interpreted with caution.
Study limitations
Important limitations in the current study were the risk of systematic bias caused by patients lost to follow-up and the use of the RLA and qualitative neuropsychological assessments in the classification of LOC. As shown by Schnakers et al. (15) qualitative behavioral assessments may increase the likelihood of misdiagnosis in patients with DOC relative to when a standardized assessment tool such as the Coma Recovery Scale-Revised is applied. The total rate of diagnostic error in the current study is unknown. However, none of the included patients were assessed to be in VS/UWS at follow-up, and hence it is unlikely that signs of consciousness were missed in patients at this time-point. Moreover, as specialized assessment tools for VS/UWS and MCS are currently not used on a wide scale in acute care facilities (14), the main message of this and previous studies to professionals working within acute care is the considerable rehabilitation potential of patients with DOC (11, 30).
Attrition has been found to be substantial in previous longitudinal studies of TBI (40), and systematic bias may occur when a variable associated with attrition is also associated with the outcome of interest. In the current study, univariate analyses indicated that patients lost to follow-up were significantly older and had lower Cog-FIM total scores at discharge from the TBI Unit. The same differences applied, when the total group of excluded patients was compared with patients included in the study group. Since age was negatively correlated with Cog-FIM total scores at follow-up, and patients lost to follow-up had lower scores on Cog-FIM at discharge, patients with poor outcomes appeared to be missing systematically from the study. Accordingly, outcomes in the present study may have been positively biased with consequences for the external validity and generalizability of results.
Future research
The current study was mainly descriptive and exploratory rather than hypothesis testing and thus further investigation of the effects of sub-acute rehabilitation and prognostic factors in patients with severe TBI is warranted. Future studies should assess patients consecutively in the years post-trauma, since ongoing recovery is to be expected, and an effort should be made concerning the follow-up of patients of older age and with greater functional dependence at discharge from rehabilitation, as these patients may be particularly vulnerable to attrition. Moreover, the association between pre-morbid cognitive reserve and cognitive activity limitations post-trauma may be investigated using more specific and sensitive measures of cognitive reserve.
Conclusion
Overall, substantial recovery was documented 1 year post-trauma in patients with DOC and severe TBI admitted to sub-acute rehabilitation in the Eastern part of Denmark. SES, duration of acute care and PTA were significant predictors of functional independence within important cognitive domains 1 year after TBI. In accordance with previous research, the current study suggests that absence of consciousness at discharge from acute care should not preclude patients from being referred to sub-acute rehabilitation.
References
