Donald G. Grosset, MD, Elaine G. Tyrrell, MPhil and Katherine A. Grosset, MD
From the Institute of Neurological Sciences, Southern General Hospital, Glasgow, UK
OBJECTIVE: To explore the dose equivalence ratio and treatment costs for abobotulinumtoxinA and incobotulinumtoxinA for patients with focal dystonias.
DESIGN: Patient chart review.
Subjects/Patients: Adult patients with blepharospasm (n = 19), cervical dystonia (n = 122), hemifacial spasm (n = 91) or segmental/generalized dystonia (n = 19) at a neurology outpatient clinic.
METHODS: Patients were switched from established abobotulinumtoxinA therapy to incobotulinumtoxinA at a ~4:1 unit ratio. Dose requirements, injection intervals, treatment efficacy, and adverse events were evaluated for a period of ≥ 1 year.
RESULTS: Patients were switched from abobotulinumtoxinA to incobotulinumtoxinA with a mean dose ratio of 4.07 (standard deviation (SD) 0.50). After switching, incobotulinumtoxinA dose requirements remained stable; the mean (SD) dose ratio at the end of the review period (52–219 weeks after switching) was 3.89 (SD 0.58). Injection intervals also remained stable after switching. Adverse events were injection site pain (n = 45) and bruising (n = 4). Five patients (2.0%) discontinued incobotulinumtoxinA treatment: 4 stopped receiving injections, and 1 reverted to abobotulinumtoxinA. Switching to incobotulinumtoxinA reduced the mean toxin expenditure to 76.7% of the cost of abobotulinumtoxinA.
Conclusion: For patients with conditions commonly treated in dystonia clinics, switching from abobotulinumtoxinA to incobotulinumtoxinA, given at equivalent doses (~4:1 unit ratio) at similar intervals, was effective, well tolerated and achieved cost savings.
Key words: botulinum toxins, type A; incobotulinumtoxinA; abobotulinumtoxinA; blepharospasm; dystonia; torticollis; hemifacial spasm.
J Rehabil Med 2014; 46: 00–00
Correspondence address: Donald G. Grosset, Institute of Neurological Sciences, Southern General Hospital, 1345 Govan Road, Glasgow G51 4TF, UK. E-mail: donaldgrosset@gmail.com
Accepted Jul 7, 2014; Epub ahead of print Oct 16, 2014
Introduction
Intramuscular injections of botulinum toxin are the first-line treatment for most types of focal dystonia, including blepharospasm, hemifacial spasm and cervical dystonia (CD) (1). Three botulinum toxin type A products are currently available in Europe and the USA: abobotulinumtoxinA (Dysport®, Ipsen), incobotulinumtoxinA (Xeomin®, Merz Pharmaceuticals) and onabotulinumtoxinA (Botox®, Allergan). All products contain neurotoxin type A, derived from the Hall strain of Clostridium botulinum, but differ in their exact molecular composition and excipients (2). In abobotulinumtoxinA and onabotulinumtoxinA, the active 150 kDa neurotoxin is part of a larger complex with other proteins, which are not thought to affect the biological activity, diffusion profile or stability of the neurotoxin (3). IncobotulinumtoxinA contains the purified neurotoxin without complexing proteins (2).
The availability of several botulinum toxin type A products raises the opportunity to explore conversion between agents. The potency of all botulinum toxin type A formulations is measured in units based on the median lethal intraperitoneal dose in mice, but methodological details of potency assays differ between manufacturers. Hence, unit labelling is specific to each formulation and universal unit conversion factors between formulations do not exist. In controlled clinical trials, abobotulinumtoxinA and onabotulinumtoxinA have been reported to have similar efficacy and tolerability when used at a 4:1 unit ratio in treating blepharospasm or hemifacial spasm (4) and at a 3:1 unit ratio in CD (5). In retrospective studies, dose equivalence ratios between 1.7:1 and 6:1 have been described (6–8). IncobotulinumtoxinA and onabotulinumtoxinA have shown therapeutic equivalence in treating blepharospasm and CD at a 1:1 unit ratio in controlled clinical studies (reviewed in 9). Controlled comparison studies of incobotulinumtoxinA and abobotulinumtoxinA have not been published.
We have reviewed data from patients with focal dystonias in our neurology outpatient clinic who were switched from established therapy with abobotulinumtoxinA to incobotulinumtoxinA.
Methods
Patients and settings
Our neurology outpatient clinic serves a regional catchment area in the west of Scotland with a population of 2 million, and provides botulinum toxin treatment to all cases of CD and hemifacial spasm, and most cases of blepharospasm, in that catchment area. From 2009, we switched patients from established abobotulinumtoxinA treatment to incobotulinumtoxinA. In the current study, we retrospectively reviewed patient chart data to evaluate a potential impact of switching on dose requirements, treatment intervals and treatment cost for patients with a follow-up period of at least one year after switching. Treatments were administered in our clinic by 4 experienced injectors.
Switching from abobotulinumtoxinA to incobotulinumtoxinA
Switching was generally initiated at a 4:1 unit ratio of abobotulinumtoxinA to incobotulinumtoxinA. Following the standard procedure in our clinic and in accordance with the prescribing information, 500 unit vials of abobotulinumtoxinA were reconstituted to a final volume of 2.5 ml and 100 unit vials of incobotulinumtoxinA to 2.0 ml. Therefore, switching at a 4:1 unit ratio did not affect the injection volume.
Injection intervals
The standard initial treatment interval for botulinum toxin injections at our clinic is 13 weeks. Appointments for the next injection were made at each injection visit; however, intervals were reviewed at each visit and increased or decreased according to clinical need. Adjustments were made stepwise, usually in 1-week steps. The minimum injection interval at our clinic is 10 weeks.
Assessments
At each clinic visit, the effects of the previous botulinum toxin treatment were captured using a standard assessment form. Treatment efficacy was recorded as described by patients on a 7-point scale: excellent, very good, good, fairly good, fair, poor, or negligible. The duration of treatment effect during the previous treatment cycle was rated by patients on a 4-point scale as: excellent duration, good duration, a few weeks or short-lived, where a few weeks was defined as approximately 5 weeks. Patients were encouraged to report any adverse events (AEs) and actively questioned about any occurrences of swallowing difficulties, bruising at the injection site, dry eye, eyelid ptosis, facial weakness and diplopia, which were graded as mild, moderate or severe, as reported by patients. Based on these assessments, botulinum toxin doses, injection sites and injection interval for the next treatment were adjusted to patients’ clinical needs.
Results
Patients
Botulinum toxin treatment was switched in 257 patients with blepharospasm, CD, hemifacial spasm, or segmental or generalized dystonia. Of these, 251 (97.7%) received incobotulinumtoxinA for one year or longer and were included in this analysis. The most commonly treated disorders were CD and hemifacial spasm. Further patient characteristics are summarized in Table I. Six patients stopped receiving incobotulinumtoxinA within one year of switching. Of these, 3 patients stopped treatments because their symptoms settled, 1 patient with hemifacial spasm stopped when going for surgery, 1 patient stopped as they felt the treatment was not working and 1 patient was lost to follow-up.
|
Table I. Patient characteristics and abobotulinumtoxinA dosing before switching (n = 251) |
|||||
|
Diagnosis |
Patients n (%) |
Age, years Mean (SD) |
Female gender % |
AbobotulinumtoxinA dose, U |
|
|
1 year pre-switch Mean (SD) |
Last pre-switch Mean (SD) |
||||
|
Blepharospasm |
19 (7.6) |
63.4 (12.0) |
47.4 |
80 (30) |
79 (26) |
|
Cervical dystonia |
122 (48.6) |
57.2 (13.7) |
73.8 |
379 (166) |
402 (171) |
|
Hemifacial spasm |
91 (36.3) |
62.5 (12.6) |
73.3 |
46 (38) |
47 (43) |
|
Segmental or generalized dystonia |
19 (7.6) |
55.4 (14.7) |
73.7 |
288 (227) |
326 (246) |
|
SD: standard deviation. |
|||||
The mean follow-up period after switching was 91 weeks (standard deviation (SD) 31; range 52–219 weeks). During this period, patients received a mean of 8.3 (SD 1.8) incobotulinumtoxinA treatment cycles. Five patients stopped receiving incobotulinumtoxinA injections more than 52 weeks after the switch. Of these, 4 patients stopped because they felt they did not benefit from receiving any botulinum toxin treatment. One patient reverted to abobotulinumtoxinA due to a perceived lack of efficacy and preference for abobotulinumtoxinA.
Dose requirements
The mean abobotulinumtoxinA dose patients received one year before switching was 229 units (SD 207). At the treatment just prior to switching, the mean dose was 243 units (SD 220). Doses for patients with different conditions are summarized in Table I. At the first incobotulinumtoxinA treatment, the mean dose administered was 60.6 units (SD 54.8). The mean (SD) switch dose ratio of abobotulinumtoxinA units to incobotulinumtoxinA units was 4.07 (SD 0.50).
Overall, dose requirements were stable over time; one year after switching the mean incobotulinumtoxinA dose was 62.5 units (SD 55.8). Follow-up data over a longer period of more than one year were available for 119 patients, with a mean dose requirement of 64.8 units (SD 59.6) of incobotulinumtoxinA at the end of year 2. The mean dose ratio at the end of the review period (52–219 weeks after switching) was 3.89 (SD 0.58). Dose requirements varied for the conditions treated in our clinic; requirements according to diagnosis are shown in Fig. 1.
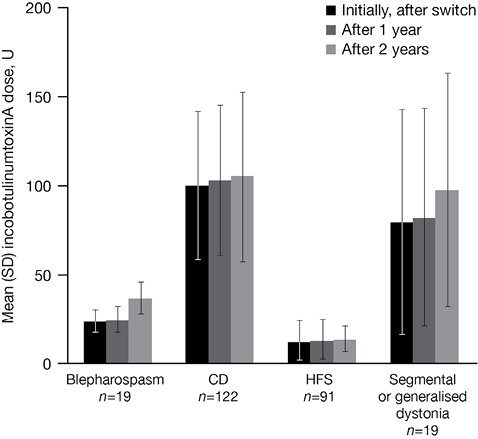
Fig. 1. IncobotulinumtoxinA dose requirements by diagnosis over time. SD: standard deviation.
Injection intervals
Prior to switching, abobotulinumtoxinA injection intervals were relatively stable over time. For all patients, the mean abobotulinumtoxinA injection interval one year before switching was 12.9 weeks (SD 2.7) and the interval directly before switching was 13.0 weeks (SD 3.0). After switching to incobotulinumtoxinA, injection intervals remained stable during the follow-up period; the last mean incobotulinumtoxinA treatment interval for all patients at the end of their follow-up period (52–219 weeks after switching) was 12.9 weeks (SD 3.3). Overall, mean injection intervals were similar for patients with different diagnoses. Injection intervals before and after switching according to diagnosis are shown in Fig. 2.
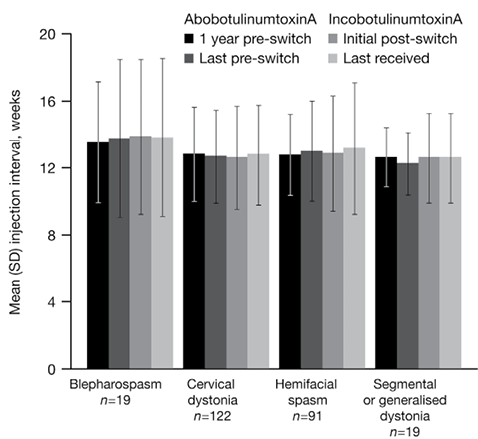
Fig. 2. Injection intervals with abobotulinumtoxinA and incobotulinumtoxinA by diagnosis over time. SD: standard deviation.
Patient-rated efficacy and duration of treatment effect
The majority of patients (84.1%) rated the efficacy of their incobotulinumtoxinA injections as excellent or very good (excellent, n = 10; very good, n = 201; good, n = 29; fairly good, n = 11). No patients reported efficacy ratings of fair or less.
The duration of treatment effect was comparable for abobotulinumtoxinA and incobotulinumtoxinA injections. The majority of patients (93.6%) rated the duration of incobotulinumtoxinA treatment effect as excellent or good (excellent duration, n = 8; good duration, n = 227; a few weeks, n = 14; short-lived, n = 2).
Tolerability
IncobotulinumtoxinA injections were generally well tolerated. Throughout the observation period, and over a mean of 8.3 incobotulinumtoxinA injection cycles, 45 patients reported pain at the injection site (mild, n = 4; moderate, n = 34; severe, n = 7) and 4 patients experienced bruising (mild, n = 3; moderate, n = 1).
Expenditure
In our clinic, based on the mean expenditure for abobotulinumtoxinA per patient per year before the switch and the mean expenditure for incobotulinumtoxinA per patient per year after the switch, switching from abobotulinumtoxinA to incobotulinumtoxinA resulted in a mean reduction in the expenditure for botulinum toxin per patient per year to 76.7% (SD 14.8) of the cost before switching.
Discussion
In our neurology outpatient clinic, patients with focal dystonias who received established abobotulinumtoxinA therapy were switched to incobotulinumtoxinA. We retrospectively reviewed the charts of patients who received incobotulinumtoxinA for at least one year after switching and found that a conversion at a ~4:1 unit ratio of abobotulinumtoxinA to incobotulinumtoxinA resulted in similar treatment effects in terms of efficacy, duration of treatment benefit and adverse events profile. Importantly, after switching at a 4:1 unit ratio, the overall incobotulinumtoxinA dose requirements and injection intervals remained stable at subsequent treatments over the observation period (52–219 weeks). The largest variations in dose requirements were seen for blepharospasm and segmental or generalized dystonia. However, few patients with these conditions were included in this analysis.
To our knowledge, this is the first report describing a switch from treatment with abobotulinumtoxinA to incobotulinumtoxinA in daily neurological practice; controlled head-to-head clinical trials comparing these botulinum toxin formulations in patients with neurological conditions have not been reported. IncobotulinumtoxinA has shown therapeutic equivalence to onabotulinumtoxinA in controlled clinical studies (9); hence, it would be expected that the clinical conversion ratio between abobotulinumtoxinA and incobotulinumtoxinA would be similar to the conversion ratio between abobotulinumtoxinA and onabotulinumtoxinA. Previous clinical studies and retrospective reviews have described abobotulinumtoxinA to onabotulinumtoxinA conversion ratios ranging from ~1.7:1 to 6:1 (4–8), in agreement with our findings.
Botulinum toxin doses used in our clinic are within the recommended dose range for each indication according to product prescribing information, but lower than doses used in controlled abobotulinumtoxinA and incobotulinumtoxinA clinical trials (10–13), and those described in many clinical practice reports (6, 7, 14). However, treatment approaches vary between different countries and clinics and will evolve over time. For instance, our treatment approach for hemifacial spasm was refined during the review period from injecting botulinum toxin into 3 sites to distributing the same total dose across 5 sites. Importantly, botulinum toxin doses received were regularly reviewed, individualized and adjusted throughout the review period based on efficacy and duration of treatment effect to optimize effectiveness, suggesting that dosing was appropriate for the patient population in our clinic.
Overall, incobotulinumtoxinA treatment was generally well tolerated. The only AEs reported by patients (pain and bruising) were restricted to the area around the injection site and were consistent with AEs expected from any injection procedure. Other AEs that have been observed in clinical trials of botulinum toxin treatment for focal dystonias (reviewed in (14)) were not reported, despite active questioning regarding the occurrence of swallowing difficulties, dry eye, eyelid ptosis, facial weakness or diplopia at each clinic visit. As the incidence of AEs after botulinum toxin injections has been reported to be dose-related (10, 14), this may in part reflect the lower doses used in our clinic compared with many clinical trials.
The number of patients who stopped receiving incobotulinumtoxinA injections 12 months post switching was low (2.0%, 5/251), and 4 of the 5 patients who stopped incobotulinumtoxinA injections stopped receiving any botulinum toxin treatment. Importantly, we did not observe a difference in the number of patients who stopped receiving botulinum toxin injections during treatment with abobotulinumtoxinA or incobotulinumtoxinA.
We found that, in our clinic, switching from abobotulinumtoxinA to incobotulinumtoxinA reduced the expenditure on botulinum toxin per patient per year to a mean 76.7% of the pre-switch cost. This is a substantial cost saving, since previous phamacoeconomic evaluations of botulinum toxin therapy for focal dystonias have shown that the overall treatment costs are largely made up of the cost of the toxin itself (15). Of note, the expenditure is based on the actual dose of botulinum toxin administered.
Potential limitations of this study are the retrospective nature of the analysis and the reliance on patient-reported outcomes to assess efficacy and treatment duration. However, reliance on patient-reported information is typical of routine clinical practice. The main strengths are the large number of patients, who were treated by 4 experienced injectors throughout the review period, and the low rate of treatment discontinuation.
In conclusion, switching from abobotulinumtoxinA to incobotulinumtoxinA at a 4:1 unit ratio resulted in good therapeutic effectiveness in terms of treatment efficacy, duration of treatment effect and adverse events profile, and reduced treatment costs for patients with a range of conditions commonly treated in our dystonia clinic.
Acknowledgements
Editorial support for developing this manuscript was provided by Dr Simone Boldt (Complete Medical Communications) and funded by Merz Pharmaceuticals GmbH.
Conflicts of interest and funding
The authors did not receive financial support for this study. Funding for editorial support for the development of this manuscript was provided by Merz Pharmaceuticals GmbH. The author’s department received support from Merz for the development of a dystonia database (in 2011–2012). Merz Pharmaceuticals had no influence on the interpretation of data and the final conclusions drawn.
References
