Deborah Severijns, MSc1, Ilse Lamers, MSc1, Lore Kerkhofs, BSc2 and Peter Feys, PhD1
From the 1REVAL Rehabilitation Research Center, BIOMED, Faculty of Medicine and Life Sciences, Hasselt University, Diepenbeek and 2Rehabilitation and MS Center, Overpelt, Belgium
BACKGROUND: Hand grip strength in both the dominant and non-dominant hands is often used to assess upper limb impairment. Excessive hand grip fatigability is another important measure, as fatigability may also influence activities of daily living.
OBJECTIVE: To investigate to what extent hand grip fatigability in multiple sclerosis is dependent on hand dominance, muscle strength and disease progression.
METHODS: Thirty persons with multiple sclerosis and 16 healthy controls performed 15 repeated maximal hand grip contractions and a 30 s sustained contraction in order to determine dynamic and static fatigue indices. Fatigability was compared between the dominant and non-dominant hands and between the more and less affected hands in a subgroup of persons with multiple sclerosis with asymmetrical hand grip strength impairment. Furthermore, fatigability was compared between controls and subgroups of persons with multiple sclerosis with different disease progression.
RESULTS: There was no difference in fatigability between dominant and non-dominant hands in healthy controls or in persons with multiple sclerosis. Similarly, there was no difference between the more and less affected hands in the subgroup of persons with multiple sclerosis with asymmetrical hand grip impairment. The dynamic fatigue index did not discriminate persons with multiple sclerosis from controls. While the static fatigue index was not different between healthy controls and persons with multiple sclerosis with low to moderate (< 6) Expanded Disability Status Scale (EDSS), it was significantly higher in persons with multiple sclerosis with high (≥ 6) EDSS scores. The static fatigue index was related to the EDSS score, but not to maximal grip strength.
CONCLUSION: Fatigability of hand grip strength in persons with multiple sclerosis is not influenced by hand dominance or muscle strength, but there is a correlation with disease progression. Differences in fatigability between healthy controls and, in particular, persons with multiple sclerosis with high EDSS, were found during sustained, but not during dynamic, contractions.
Key words: muscle fatigue; fatigability; multiple sclerosis; upper extremity; grip strength; hand dominance.
J Rehabil Med 2014; 46: 00–00
Correspondence address: Deborah Severijns, Hasselt University, REVAL-BIOMED, Agoralaan gebouw A, BE-3590 Diepenbeek, Belgium. E-mail: deborah.severijns@uhasselt.be
Accepted Jul 7, 2014; Epub ahead of print Sep 30, 2014
Introduction
Persons with multiple sclerosis (MS) frequently experience motor symptoms, such as muscle weakness, hypertonia and fatigue (1). Fatigue is considered one of the most disabling symptoms by 30–50% of persons with MS (2, 3). Fatigue is an umbrella term for different concepts, therefore Kluger et al. (3) propose using the term “fatigability” when referring to objective changes in performance. Fatigability during motor tasks is also referred to as motor or muscle fatigue and can be described as “an exercise-induced reduction in the ability of muscles to produce force or power, regardless of whether a task can be sustained” (4).
Upper limb impairment is present in a large proportion of persons with MS (5), causing disabilities in daily life (6). Grip strength is often used as a simple outcome measure to assess arm function or overall functional decline, since it is related to the perceived functional use of the hand (7, 8), manual dexterity (9) and overall functional status (10). During activities of daily living, such as lifting and holding a cup, not only short maximal force production, but also the ability to sustain a prolonged contraction or to produce repeated contractions is important. Many persons with MS have a lower grip strength than healthy persons and, moreover, many use excessive grip force when displacing an object (11), which could predispose them to higher fatigability. Fatigability in persons with MS has previously been assessed in both upper and lower limbs with dynamometry, documenting a decline in maximal force either during repeated maximal contractions or during a sustained contraction. However, these studies varied in the protocol and calculation of outcome parameters (12–16), which makes comparison of the results difficult. Reports on the psychometric properties of these various fatigability assessments are sparse in persons with MS. However, Surakka et al. (12) and Schwid et al. (14) reported that indices based on the area under the strength curve of maximal isometric contractions of 30 s are reliable in persons with MS. Literature on upper limb fatigability showed higher fatigability levels in persons with MS compared with healthy controls (14–16). These conclusions were based mainly on persons with MS with mild arm-hand dysfunction. However, upper limb function is expected to deteriorate as the disease progresses. Questions remain about the relationship between fatigability and degree of muscle weakness, and about the relationship between fatigability and accumulating overall disability related to disease progression. Research into the relationship between muscle weakness and fatigability has shown inconsistent results, although recent research has indicated an association (12, 17). One could furthermore hypothesize that fatigability is related to disease progression, with possible associated neuronal atrophy and reduced neural conduction times. In this regard, a significant relationship between subjective fatigue and Expanded Disability Status Scale (EDSS) scores has been reported (18, 19), but, to the best of our knowledge, no relationship between fatigability and EDSS score has yet been reported.
So far, upper limb fatigability studies in persons with MS have investigated only the dominant arm (14–16). It is known, however, that symptoms such as muscle weakness can occur asymmetrically in persons with MS, therefore a difference between the more and less affected hands can be expected. In addition, a recent study suggested that, in persons with MS, the non-dominant arm is likely to be more impaired than the dominant arm, perhaps due to de-training effects associated with reduced use in daily life given decreased functionality (8). One may hypothesize a higher fatigability in the non-dominant compared with the dominant hand in persons with MS with higher EDSS scores, since these persons with MS use their dominant arm more in daily life (8). However, if pathological muscle fatigue in persons with MS is mainly of central origin, one could also hypothesize that there would be no difference between dominant and non-dominant hands, nor between the more and less affected sides, which could be supported by reports in healthy persons, stating that fatigability did not seem to differ between dominant and non-dominant hands (20). To our knowledge, the difference in fatigability between dominant and non-dominant hands has not been assessed in persons with MS.
The aim of this study is to examine whether hand grip fatigability differs between the dominant and non-dominant arms in both healthy controls and persons with MS, and between the more and less affected hands in a subgroup of persons with MS, and to determine if hand grip fatigability is different between healthy controls and subgroups of persons with MS with different disease progression.
Material and Methods
Subjects
Persons with MS diagnosed according to McDonald’s criteria were recruited via existing databases in the research institute and the Rehabilitation and MS centre Overpelt (Belgium). Exclusion criteria were: (i) the clinical presence of tremor and ataxia; (ii) cognitive and orthopaedic problems that could interfere with hand grip testing; (iii) corticosteroid treatment within 1 month before testing. Age- and sex-matched healthy controls were recruited from within family and colleagues. All subjects gave their informed consent prior to participation. The study was approved by the ethics committees of Hasselt University and Rehabilitation and MS centre Overpelt.
Descriptive outcome measures
The EDSS score was provided by the treating neurologist (21). The modified Fatigue Impact Scale (MFIS) was used to assess the impact of fatigue on daily life (22, 23). This self-reported fatigue questionnaire assesses fatigue in daily life in the psychosocial, mental and cognitive domain. Twenty-one items are scored between 0 (never) and 4 (almost always), giving a score between 0 and 84. The higher the score on the MFIS, the greater the impact of fatigue experienced during daily life. The cut-off score of 38 to distinguish between non-fatigued and fatigued patients was described by Flachenecker et al. (24).The Motricity Index (MI) was used to assess arm muscle strength. Pinch grip, elbow flexion and shoulder abduction were measured and scored, with a maximum of 100 indicating a normal score (25). As in Lamers et al. (8), the dominant hand was determined with the following questions: “Are you currently left- or right-handed and has this changed due to MS?”.
Experimental design and outcome measures
Maximal voluntary hand grip strength was recorded by use of a digital JAMAR hand grip module (E-link, Biometrics Ltd, Newport, UK) (26). Hand grip strength measurements were performed in a neutral wrist posture (arms resting at sides, elbows flexed at 90°, wrist positioned in the mid-range of pronation and supination, maximal 30° wrist extension) according to the recommendations of the American Society of Hand Therapists (ASHT). The hand grip device installed in the second position (27) After a short period of familiarization with the digital JAMAR hand grip module, all tests were performed bilaterally in random order. Every patient was instructed to perform 15 repeated maximal contractions at their own pace. The maximal hand grip strength is defined as the maximal value from the first 3 dynamic trials. During these repeated hand grip contractions, patients were verbally guided on the number of contractions. The dynamic fatigue index was calculated as previously described by Schwid et al. (14), based on the ratio between the maximal strength of the first 3 and last 3 dynamic hand grip trials, according to the following formula: 100*[ 1–(MVC2/MVC1)] (MVC: maximal voluntary isometic contraction). A schematic representation of the dynamic fatigue index can be found in Fig. 1A.
Following 1 min rest, a 30 s sustained maximal hand grip contraction was performed, without feedback about the elapsed time. The static fatigue index was calculated as described previously by Surakka et al. (12). Firstly, the maximal hand grip strength and the time of this maximal hand grip strength (Tmax) was determined. The maximal hand grip strength (in kg) was multiplied by 30 minus Tmax to calculate a hypothetical area under the curve (HAUC). This HAUC represents the curve that would be obtained if the subject kept the force constant throughout the 30 s. Then, the actual area under the strength curve (AUC) was calculated on the basis of the area under the force-time curve from Tmax to 30 s. This AUC was divided by the HAUC. The fatigue static index was then calculated using the following formula: 100%*[1–(AUC/HAUC)] (12). A schematic representation of the static fatigue index can be found in Fig. 1B. The higher the fatigue indices, the more the maximal hand grip strength decreases over time, and thus the more fatigability occurs.
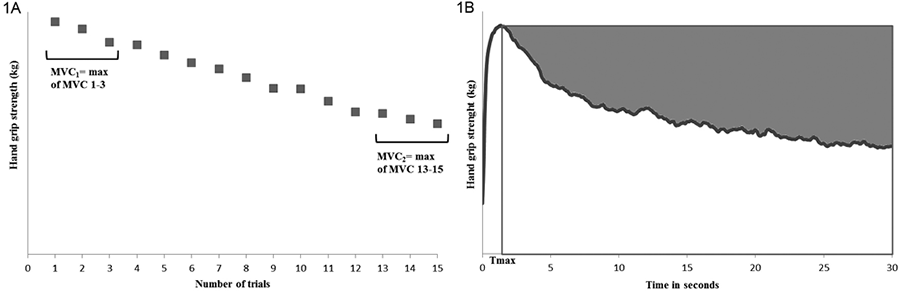
Fig. 1. Calculation methods for the fatigue indices. (A) The dynamic fatigue index is calculated based on the ratio between MVC1 and MVC2 (MVC: maximal voluntary isometic contraction). (B) The static fatigue index is illustrated by the grey shaded area.
Statistical analysis
All statistical analyses were performed using SPSS (IBM SPSS Statistics 20, ©IBM, Armonk, NY, US). Significance was set at p < 0.05.To study the differences between the dominant and non-dominant arms, a paired samples t-test was used. In addition, in a subgroup of the persons with MS, who presented with an asymmetrical impairment of hand grip strength (defined as a hand grip strength ratio between left and right hands > 10%), the more and less affected sides were compared using a paired samples t-test. Furthermore, Pearson’s correlation coefficient was used to determine the correlations between the fatigability of the dominant and non-dominant hand as well as between the more and less affected hands.
Independent samples t-tests were applied for comparison between healthy controls and persons with MS. For the analysis of subgroups according to disability level, the total group of persons with MS was subdivided according to their EDSS score. Subgroups of persons with MS with mild to moderate (EDSS < 6) MS and severe (EDSS ≥ 6) MS (5) were compared with healthy controls using one-way analysis of variance (ANOVA) with post-hoc Tukey’s tests in case of significant group differences. Correlations between disability level, muscle strength and fatigue indices were examined using Pearson’s correlation coefficients.
Results
Subject characteristics
Subject characteristics of the 30 persons with MS and 16 healthy controls are described in Table I. Twenty-five of the persons with MS were right-handed. Two persons with MS changed hand dominance, and were now mostly using the left instead of the right hand. Most of the persons with MS had clinically detectable muscle weakness, as shown by the score on the MI, but 8 persons with MS had no muscle weakness in their upper limbs, as indicated by a maximal score of 100 on the MI. The median EDSS score of the total group of persons with MS was 4.0 (range 1.5–8.5). According to the MFIS cut-off score of 38 (23, 24), none of the healthy controls were fatigued, while 14 of the persons with MS experienced abnormal fatigue during daily life. Persons with MS reported significantly more fatigue on the MFIS than the control subjects (p < 0.001) (Table I).
|
Table I. Subject characteristics |
||||
|
Healthy controls (n = 16) |
Persons with MS |
|||
|
Total group (n = 30) |
EDSS < 6 (n = 17) |
EDSS ≥ 6 (n = 13) |
||
|
Age, years, mean (SD) |
50 (11) |
52 (12) |
49 (10) |
56 (12) |
|
Hand dominance, R/L, n |
14/2 |
25/5 |
15/2 |
10/3 |
|
Type of MS, RR/SP/PP, n |
/ |
13/12/5 |
11/5/1 |
2/7/4 |
|
Disease duration, years, mean (SD) |
/ |
16 (10 |
14 (9) |
17 (11) |
|
MFIS (0–84), mean (SD) |
11.6 (9.9) |
35.6 (21.0) |
36.8 (20.6) |
34.0 (22.3) |
|
Motricity index D (0–100), mean (SD) |
/ |
91 (13) |
95 (9) |
85 (15) |
|
Motricity index ND (0–100), mean (SD) |
/ |
86 (13) |
91 (8) |
79 (15) |
|
R: right; L: left; D: dominant hand; ND: non-dominant hand; MS: multiple sclerosis; EDSS: Expanded Disability Status Scale; SD: standard deviation; RR: relapsing – remitting; SP: secondary progressive; PP primary progressive. |
||||
Hand grip fatigability according to hand dominance and hand grip impairment
Table II provides an overview of the results. In healthy controls, there was a statistically significant difference in maximal hand grip strength between the dominant and non-dominant arms (p < 0.05). However, no significant difference between arms was detected for the dynamic and static fatigue indices (p = 0.078 and p = 0.1, respectively). There was a high and significant correlation between the static fatigue index of the dominant and non-dominant hands (r = 0.945, p < 0.001), but not for the dynamic fatigue index (r = 0.433, p = 0.093).
|
Table II. Maximal strength and muscle fatigue indices for both dominant hand and non-dominant hand in persons with multiple sclerosis (MS) and healthy controls |
|||||||||
|
Variable |
Healthy controls |
Persons with MS |
|||||||
|
D Mean (SD) |
ND Mean (SD) |
p |
r |
|
D Mean (SD) |
ND Mean (SD) |
p |
r |
|
|
Maximal hand grip strength, kg |
37.9 (9.3) |
35.6 (9.9) |
0.009 |
0.95* |
28.8 (12.2) |
26.5 (10.9) |
ns |
0.83* |
|
|
Dynamic fatigue index, % |
18.0 (8.4) |
21.8 (7.7) |
ns |
0.43 |
20.8 (12.2) |
24.4 (12.3) |
ns |
0.52* |
|
|
Static fatigue index, % |
29.3 (10.9) |
31.1 (11.3) |
ns |
0.95* |
41.5 (12.1) |
42.5 (10.9) |
ns |
0.60* |
|
|
*Significant with p < 0.05. D: dominant hand; ND: non-dominant hand; p: p-value of the paired-samples t-test; ns: non-significant; r: Pearson’s correlation coefficient; SD: standard deviation. |
|||||||||
In persons with MS, there was a trend towards significant difference (p = 0.065) between the maximal hand grip strength of the dominant and the non-dominant hands. In accordance with the healthy persons, no differences were found between the dominant and non-dominant hands for the different grip fatigue indices. Nineteen persons with MS had an asymmetrical hand grip strength impairment, with a hand grip strength difference of more than 10% between the dominant and non-dominant sides. In this subgroup, there was no significant difference between the more and less affected hands, either for the static index (p = 0.249) or the dynamic fatigue index (p = 0.683). There were moderate and significant correlations between the fatigue indices of the dominant hand and the non-dominant hand (r = 0.603 and 0.518 for static and dynamic fatigue indices) and between the more and less affected hand (r = 0.674 and r = 0.479 for the static and dynamic fatigue indices).
|
Table III. Muscle strength and fatigue indices for the dominant arm of healthy controls and persons with multiple sclerosis (MS) with low/high Expanded Disability Status Scale (EDSS) scores |
||||
|
Variable |
Healthy controls (n = 16) Mean (SD) |
Persons with MS |
ANOVA p-value |
|
|
EDSS < 6 (n = 17) Mean (SD) |
EDSS ≥ 6 (n = 13) Mean (SD) |
|||
|
Maximal hand grip strength, kg |
37.9 (9.3) |
32.9 (10.1) |
23.5 (13.1) |
0.003* |
|
Dynamic fatigue index, % |
18.0 (8.4) |
18.1 (10.4) |
24.3 (13.9) |
ns |
|
Static fatigue index, % |
29.3 (10.1) |
37.2 (7.9) |
47.1 (14.5) |
<0.001† * |
|
†p < 0.05 for EDSS < 6 vs EDSS ≥ 6; *p < 0.05 for healthy controls vs EDSS ≥ 6. ns: not significant; ANOVA: analysis of variance; SD: standard deviation. |
||||
Hand grip fatigability in persons with MS and healthy controls for the dominant hand
Remaining analyses were performed for the dominant arm only, in view of the above-described results with hand dominance not significantly affecting the fatigue indices. Compared with healthy controls, the persons with MS have, on average, a significantly lower hand grip strength (p = 0.013). The static fatigue index was significantly higher in persons with MS, with 41% compared with healthy persons with 29% (p = 0.002), which means that persons with MS are not able to sustain their maximal strength as long as healthy controls. A graph of the 30 s isometric hand grip task is shown in Fig. 2. Fourteen out of 30 persons with MS scored higher for their static fatigue index than the maximal value for the static fatigue index of the healthy controls. These 14 persons with MS did not differ significantly from the other persons with MS for maximal muscle strength, score on the MFIS, or disease duration, but they did differ significantly for the mean EDSS score (with a mean difference of 2 points, p = 0.017). In contrast, the dynamic fatigue index, based on repeated maximal contractions, was not significantly different between persons with MS and healthy persons (p = 0.4).
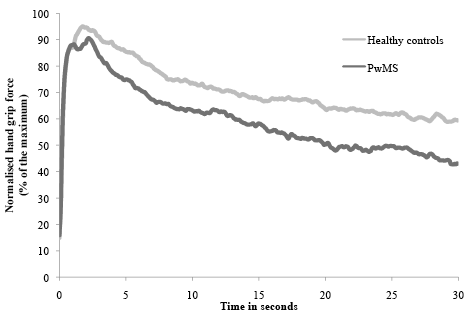
Fig. 2. Mean strength of persons with multiple sclerosis (PwMS) and controls during a 30 s sustained contraction, normalized to the maximal hand grip strength.
Hand grip fatigability according to disease stage
Table III displays the results of maximal muscle strength and muscle fatigue indices for groups of healthy subjects and persons with mild-to-moderate (EDSS < 6) and severe MS (EDSS ≥ 6). Analyses of variance indicated on differences between groups for the maximal hand grip strength (F(2.43) = 6.57; p = 0.003) and the static fatigue index (F (2.43) = 9.65; p < 0.001), but not for the dynamic fatigue index. Post-hoc test showed significant differences between persons with MS subgroups, and between healthy controls and the high EDSS subgroup, for the maximal strength and static fatigue index (Fig. 3). There were no differences between the healthy controls and the low EDSS subgroup for the static fatigue index.
Furthermore, in the total MS group, there was a moderate significant correlation between EDSS score and maximal voluntary hand grip strength (r = –0.43, p = 0.017), as well as with the static fatigue index (r = 0.43, p = 0.019). Maximal hand grip strength and the static fatigue index were, however, not significantly correlated in persons with MS.
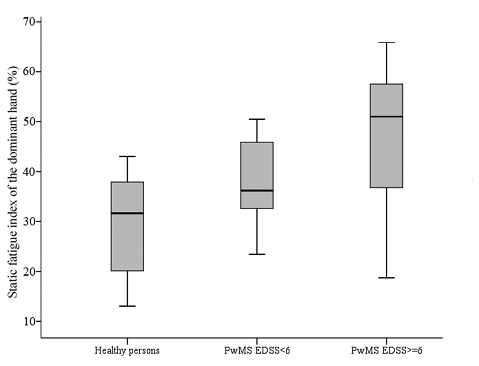
Fig. 3. Box-plots of the static fatigue index in healthy controls, persons with multiple sclerosis (PwMS) with low to moderate Expanded Disability Status Scale (EDSS) score and persons with MS with high EDSS score.
Discussion
This study investigated the effect of hand dominance, hand grip impairment and disability level on maximal hand grip strength and fatigability in healthy controls and persons with MS. Despite differences in hand grip strength between the arms, no impact of hand dominance or strength impairment on hand grip fatigability was found. The static fatigue index, based on an isometric contraction of 30 s discriminated between persons with MS and healthy controls, while the dynamic fatigue index, based on 15 repeated maximal contractions, did not. Fatigability in the upper limb was greater in persons with severe MS compared with persons with mild-to-moderate MS and healthy controls.
Maximal hand grip strength differed significantly between the dominant and non-dominant hands in healthy persons, as documented previously (28). However, no significant difference between hands was found for persons with MS, possibly linked to disease-related muscle weakness (8). Similarly, fatigability was not different between dominant and non-dominant hand, either in healthy controls or in persons with MS. These results are consistent with those of Gordon et al., who reported an equal fatigability of the upper limb in a sample of healthy persons during a submaximal contraction for the dominant and non-dominant arms (20). There were moderate correlations between fatigue indices of the dominant and non-dominant hands and between the more and less affected hands, possibly indicating on a central origin of muscle fatigue, as suggested in previous research (17).
Persons with MS show more fatigability than controls during a sustained maximal hand grip contraction. We found a static fatigue index of 41% for the persons with MS and 29% in the healthy controls. This is in line with Schwid et al. (14), who found a mean static fatigue index of 49.2% for persons with MS vs 30.4% for controls (14). However, when comparing the results of the subgroup of persons with MS with mild to moderate disability with the results of Schwid et al., who included only persons with MS with mild to moderate disability, our subgroup scored lower for the static fatigue index (37.2 ± 7.9%). The static muscle fatigue index has demonstrated validity and reliability for persons with MS, as described by Surakka et al. (12) and Schwid et al. (14). The consistency of findings across studies, despite small differences in calculation method of the static fatigue indices, indicates that this parameter has discriminative power to detect differences in fatigability. For the dynamic fatigue index no group differences were found. This lack of difference was not expected, since, in clinical practice, persons with MS often report a sense of fatigue during repetitive actions. Furthermore, previously, the ratios between 10 or 11 successive maximal hand grip contractions were already able to discriminate between healthy controls and persons with MS with mild to moderate disability (15, 16). A possible explanation is the limited reliability of the dynamic fatigue index (14). Moreover, a higher decline in force is expected during sustained contractions in contrast to repeated contractions, which can explain the different findings of the static and dynamic fatigue indices (29). Furthermore, methodological choices, such as whether to impose a rhythm during repeated contractions or the number of contractions, may influence the discriminative ability of the repeated contractions. In our study, the pace of contractions was not set, because we preferred that all persons with MS would reach their maximal force level, which was unlikely when contraction frequency was pre-determined and speeded up. We allowed a comfortable pace, as this is regarded as more functional and related to daily life. This might explain why our results are different from those reported by Schwid et al., who did apply a fixed rhythm and found a significant difference between healthy controls and persons with MS (14). Following this, it can also be hypothesized that the dynamic fatigue indices should be determined based on a larger number of repetitions (than the 15 used in the current study).
The present study expands previous research on muscle fatigue indices that was mostly performed either on samples with mild MS, or without any differentiation according to EDSS level (15, 16). Schwid et al. also included more disabled persons with MS (mean EDSS score 5.5), but without reporting on the correlation between EDSS score and fatigability (14). In the present study, the majority of the included persons with MS showed clinical detectable muscle weakness in the upper limbs. Our results revealed that persons with MS with EDSS scores ≥ 6 showed significantly more fatigability during sustained hand grip tasks compared with persons with MS with lower EDSS (< 6). This could be related to the disease process itself, with decreasing neuronal reserve, or to other factors, such as disuse of the arms because of diminished movement efficiency due to muscle weakness (8). This is further supported by our finding of a significant association between the static fatigue index and the EDSS score. One may relate this association to the presence of muscle weakness in persons with MS with higher EDSS levels. However, even in a subgroup of persons with MS who presented with asymmetrical impairment of the hand grip strength, there was no difference between the more and less affected hands. In addition to this, there were no significant correlations between the maximal hand grip strength and the muscle fatigue indices. These findings are in line with Schwid et al. (14), but contrast with the findings of previous research in samples without major clinical detectable muscle weakness (16, 30), where correlations between fatigability measures and baseline strength were reported. Inconsistent findings across studies may relate to different samples of persons with MS, with the present study probably including a larger proportion of patients with arm-hand dysfunction.
Some limitations of this study need to be addressed when interpreting the results. The conclusions of this report were based on a total sample of 30 persons with MS and 16 healthy controls. This limited sample size cautions against generalization of the study results. However, we have tried to take a representative sample of persons with MS, as indicated by the range of EDSS scores.
Furthermore, one may argue that the fact that the dynamic and static fatigue indices were assessed during a single test session could have influenced our results, where only the second measure was able to differentiate between persons with MS and healthy controls. However, there was a short rest period of 1 min between the repeated hand grip contractions and the sustained hand grip contractions. It has been shown that recovery after a sustained hand-grip contraction until exhaustion is quick (within 2 min) (16). Furthermore, none of the tested subjects indicated feeling fatigued after the short rest period between the measurements. In future research, a randomization of assessments is warranted in order to avoid a possible impact of the first measurement on the following assessments. One could consider other assessment methods, with possibly an easier interpretation, such as endurance time. However, the maximal duration of a sustained maximal or submaximal hand grip force may be of limited use, since it was already shown to be unreliable in healthy subjects (10). Quantifying maximal hand grip strength before and after a standardized exercise protocol, as previously suggested by Dobkin, could be investigated in future research in an attempt to quantify exercise-induced motor fatigue similar to that occurring after activities of daily living (4). Further research is needed to determine the clinical relevance of the static fatigue index for daily life, such as for example, the capacity for uninterrupted wheelchair driving and eating, and the potential impact of exercise on muscle fatigue. In an elderly population, hand grip endurance has been demonstrated to be related to functional capacity (31), but to our knowledge, no such correlation was shown in persons with MS.
Furthermore, in the present study, hand grip muscles were tested, but questions remain as to whether a similar degree of fatigability is present in other upper limb muscles, that are equally necessary in activities of daily living. Similarly, it is unclear whether muscle fatigue is a general feature, i.e. whether measurements in the lower limbs would show the same results as for the upper limb. So far, only Schwid et al. (14) have tested multiple muscles in persons with MS. They suggest that hand grip and ambulation indices, as well as fatigability indices between different muscle groups, are correlated. Others authors, however, suggest that fatigability varies between muscle groups in the lower and upper limbs (32).
Persons with MS with abnormal fatigability needs to be discriminated from persons with MS with fatigability levels in the normal range, in order to eventually select the patients who can benefit from endurance training. However, classifying fatigability of individual persons with MS as abnormal remains difficult, since normative data for fatigue indices are lacking. A powered study with a large number of participants could provide reference norms for these fatigue indices, according to age and sex. In the present study, 14 out of 30 persons with MS showed values greater than the maximal value of the static fatigue index found in our healthy controls, and thus could possibly be classified as having abnormal fatigability. In contrast, Iriarte et al. found that only 7 of the 50 persons with MS had a fatigability value above the 99th percentile of the control groups, based on repeated maximal hand grip contractions, instead of a sustained contraction (16). This might be explained by the fact that our study sample included more persons with MS with higher disability levels. Furthermore, it appears that the classification of fatigability is not only individually defined, but is also dependent on the task, as expected (33).
Acknowledgements
The authors would like to thank Veronik Truyens (Head of Paramedical Services, Rehabilitation and MS Center Overpelt, Belgium) for facilitating patient recruitment and testing.
References
