Yu Zheng, PhD1,2,3,5, Xiaojun Wu, MPH5, Yini Dang, MD4, Yan Yang, BSc5, Jan D. Reinhardt, PhD2,5,6,7 and Yingjie Dang, MD5
From the 1Department of Rehabilitation Medicine, West China Hospital, Sichuan University, 2Institute for Disaster Management and Reconstruction, Sichuan University-Hong Kong Polytechnic University, Chengdu, Sichuan, 3Interdisciplinary Division of Biomedical Engineering, The Hong Kong Polytechnic University, Hong Kong, 4The First Affiliated Hospital of Nanjing Medical University, Nanjing, 5Department of Rehabilitation Medicine, Wuxi Rehabilitation Hospital, Wuxi, Jiangsu, China, 6Swiss Paraplegic Research, Nottwil and 7Department of Health Sciences and Health Policy, University of Lucerne, Lucerne, Switzerland
OBJECTIVE: To study the prevalence of idiopathic scoliosis in children aged 6–13 years and the corresponding predictors in China, based on a representative sample from Beitang District, Wuxi.
METHODS: A total of 11,024 primary school students aged 6–13 years were enrolled. Students who had an angle of trunk inclination (ATI) ≥ 5° (determined by the forward bending test (FBT) combined with a scoliometer) were referred to our clinic for X-ray examination. Their diagnosis was confirmed by Cobb angles of ≥ 10°. Data reflecting demographics (e.g. age, sex, etc.) and clinical characteristics (i.e. Cobb angle) were collected.
RESULTS: Approximately 2.6% of the children were screened positive (determined by ATI). Four boys and 7 girls had a definite positive diagnosis, suggesting an idiopathic scoliosis prevalence of 0.22%. The mean Cobb angle of those diagnosed was 13.2 ± 5.9°. The highest prevalence was found in girls aged 12–13 years. Predictors identified in weighted and unweighted bivariate logistic regression were being female, use of single-shoulder bags, and more time using a computer.
CONCLUSION: A relatively low overall prevalence of idiopathic scoliosis was found in primary school children of Beitang District, Wuxi, China. In future screening more attention should be paid to females, those who use single-shoulder bags, and those who spend more time using a computer.
Key words: idiopathic scoliosis; screening; Cobb angle; prevalence; predictor.
J Rehabil Med 2016; 48: 00–00
Correspondence address: Yingjie Dang, Department of Rehabilitation Medicine, Wuxi Rehabilitation Hospital, No. 100 Beitang Street, Wuxi, 214043, China. E-mail: dangyingjie6004@163.com; Jan D. Reinhardt, Institute for Disaster Management and Reconstruction, Sichuan University-Hong Kong Polytechnic University, Huanghe Middle Road, Shuangliu County, Chengdu, 610207, China. E-mail: jan.reinhardt@paraplegie.ch
Accepted Mar 18, 2016; Epub ahead of print May 11, 2016
INTRODUCTION
Scoliosis is a “three-dimensional torsional deformity of the spine and trunk” involving lateral curvature in the frontal plane, axial rotation in the horizontal plane, and/or disturbance of the sagittal plane normal curvatures, kyphosis and lordosis (1). A common diagnostic criterion is a Cobb angle of 10° or more. Scoliosis can be classified into different types according to age of onset, aetiology, severity and type of curve. Each type shows different characteristics, such as rate of curve progression, degree and pattern of the three-dimensional deformity. The term idiopathic scoliosis (IS), which is the major group, applies to all patients for whom it is not possible to find a specific disease causing the deformity and who are diagnosed when non-idiopathic scoliosis has been excluded. IS often appears in otherwise healthy children, and can progress in relation to multiple factors during growth (2).
Juvenile idiopathic scoliosis (JIS) and adolescent idiopathic scoliosis (AIS) develop at the age of 4–10 and 11–18 years, respectively, and account for most cases of IS (2). AIS is the most common type, comprising 90–85% of cases of IS in children. According to a systematic review by Konieczny et al. (2), AIS occurs in the general population in a wide range from 0.47% to 5%, 2–3% is the value most often published (3). In China, a recent study investigating the prevalence of IS in primary and middle school students in Guangzhou, reported a total prevalence of 0.49% in primary school students aged 7–13 years; JIS and AIS ranged from 0.26% to 0.46% and 0.55% to 1.12%, respectively (4). Wang and colleagues screened 57,581 primary and middle school students in Beijing and reported ranges from 0% to 0.28% for JIS and 0.27% to 2.72% for AIS (5). Thus, JIS and AIS are common diseases in children. With an initial school-based general surface measurement, and a subsequent hospital-based X-ray examination, the deformity can be detected early for intervention. Although there has been much debate about the necessity and efficacy of IS screening, this action is to be recommended (6). It is worth conducting standard screening for IS, not only to overcome the limitations of previous studies, but to fill the epidemiological blank in this area.
Scoliosis leads to problems in body structure, body function, activity and participation. If the scoliosis angle at completion of growth exceeds a “critical threshold” (most authors assume this to be between 30° and 50°), there is a higher risk of health problems in adult life, cosmetic deformity, visible disability, pain, progressive functional limitations and decreased quality of life (7). Although we can assume a multifactorial origin of IS, it is important to identify predictors of IS in order to design future screening projects more economically.
The objective of this study was to better understand the prevalence of IS, as well as prevalence according to age and gender in children aged between 6 and 13 years, and the predictors of IS in China, based on a representative sample of primary school children from Beitang District, Wuxi.
METHODS
Design
Observational, cross-sectional study.
Sample
From March to June 2014, 11,024 children were screened for IS when undergoing an annual physical examination issued by the National Health and Family Planning Commission and Ministry of Education of the People’s Republic of China in schools in Beitang District, Wuxi, China. There were 5,908 boys and 5,116 girls in the study, age range 6–13 years. All 11 primary schools in this district were enrolled, with no special consideration for geographical or economic representation. In addition, all screened children were of Chinese origin, with the child and both parents having been born in Wuxi. The study was approved by the Chinese Ethics Committee for Registering Clinical Trials. All participants received detailed information about the aim of the study and were assured of anonymity, following which they gave their informed consent.
Measures
Screening stage. An experienced screening team, comprising 15 orthopaedic surgeons, rehabilitation physicians, therapists and nurses, from Wuxi Rehabilitation Hospital performed the initial school-based screening. Boys and girls were examined separately. The screening examination began with the children standing straight, with their back to the doctor, head up and the arms relaxed at the sides. With the children in this position, factors such as shoulder asymmetries, scapula prominence, unequal waistline or arm distances, and lower limb length inequality were checked. Thereafter, the Adam’s forward bending test (FBT), combined with determination of ATI by scoliometer (Orthopedic Systems Inc., Haywood, CA; USA) was performed, while allowing the upper extremities to hang freely with the palms opposed in a relaxed manner. The screeners viewed students from the front as well as from the side (8, 9). This process took approximately 1 min for each child. The inter-observer agreement regarding ATI degree from the scoliometer was very high, with an intra-class correlation (ICC) of 0.86 (95% confidence interval (95% CI) 0.85–0.88). Children who had an angle of trunk rotation (ATR) of 5° or more on the scoliometer were referred to Wuxi Rehabilitation Hospital for further evaluation with whole-spine antero-posterior and lateral radiograms (10, 11). Data collected at this stage are listed in Table I.
|
Table I. Descriptive data collected at the screening stage |
|
Items |
|
Gender |
|
Age |
|
Body mass index |
|
Family history |
|
Distance from home to school |
|
Schoolbag type |
|
Single-shoulder bag |
|
Backpack |
|
Trolley case |
|
Schoolbag weight |
|
Activity time |
|
Extracurricular sport |
|
Extracurricular activity |
|
Study |
|
Computer use |
|
Sleeping posture |
|
Supine |
|
Prone |
|
Lateral |
|
Calcium and milk supplement |
|
Calcium supplement (infant phase) |
|
Calcium supplement (currently) |
|
Milk supplement |
|
Annual family income |
|
Birth mode |
|
Eutocia |
|
Caesarean |
Diagnostic stage. Each child was first referred for clinical examination by an experienced orthopaedic surgeon, followed by an X-ray of the whole spine when deemed necessary. Final diagnosis of scoliosis, as defined by the Scoliosis Research Society (SRS), was based on Cobb angles of 10° or more measured by 2 different observers (Ning Sun and Tao Wang), and further interventions were advised (3). The inter-observer agreement regarding Cobb angles from the standing AP radiographs was excellent, with an ICC of 0.94 (95% CI 0.92–0.96). For each child, the orthopaedic surgeon completed a form reporting the presence or absence of IS, recording its curve type (single or double), curve level, Cobb angle and Risser scores. Children who were diagnosed with non-idiopathic scoliosis were excluded at this stage.
Data analysis
All statistical analysis was performed using Stata version 13.0. Descriptive statistics in terms of frequencies, means and standard deviations were provided for those screened positive, as well as for students who attended the physical examination in comparison with those who did not.
Overall prevalence, as well as prevalence by gender and age group, were estimated from unweighted and weighted (see below) logistic regression models of diagnostic status based on the inverse logit of coefficients (12). Confidence intervals (CIs) for the estimated prevalence were estimated based on maximum likelihood estimation and the observed information matrix. Furthermore, a linear regression model was performed with the data from those who were screened positive and for whom X-rays were available (n = 128). In this model, Cobb angle degrees were regressed on age, gender and their interaction (ordinary least squares). Bivariate odds ratios and 95% CIs for having a definite diagnosis by risk factors were calculated for those who attended the physical examination.
Unit-non response (non-respondents, i.e. those screened positive, but not attending the physical examination) was adjusted for by inverse probability weights based on propensity score modelling (13). The basic idea of propensity scores is to replace a collection of confounding covariates in any observational study with a function of these covariates. In this study, the application principle is to replace covariates of both respondents and non-respondents with the predicted probabilities of attending the physical examination among those screened positive. Those probabilities (the propensity scores) were estimated from a logistic regression model with backward selection of response status on demographics and predictive factors. Inverse probability weights could then be derived with which weighted analysis was performed. The idea is to give respondents who are more similar to non-respondents a higher weight in the analysis.
RESULTS
Descriptive characteristics of the study population
At the end of the screening, 282 (190 boys and 92 girls) out of 11,024 (2.6%) children were found to be positive as a result of ATI. We were unable to obtain clinical information for 154 of the 282 eligible individuals, representing a response rate of approximately 45%. Descriptive characteristics of respondents and non-respondents are shown in Table II.
|
Table II. Descriptive characteristics of the study population |
||||
|
|
Total (n = 282) |
Non-respondents (n = 154) |
Respondents (n = 128) |
p-value of difference* |
|
Gender, n (%) |
|
|
|
0.085 |
|
Male |
190 (67.38) |
97 (62.99) |
93 (72.66) |
|
|
Female |
92 (32.62) |
57 (37.01) |
35 (27.34) |
|
|
Age, years, mean (SD) |
10.21 (1.89) |
10.71 (1.76) |
9.61 (1.87) |
< 0.001 |
|
School grade, n (%) |
|
|
|
0.053 |
|
1 |
36 (12.77) |
12 (7.79) |
24 (18.75) |
|
|
2 |
38 (13.48) |
20 (12.99) |
18 (14.06) |
|
|
3 |
44 (15.60) |
21 (13.64) |
23 (17.97) |
|
|
4 |
36 (12.77) |
23 (14.94) |
13 (10.16) |
|
|
5 |
54 (19.15) |
32 (20.78) |
22 (17.19) |
|
|
6 |
74 (26.24) |
46 (29.87) |
28 (21.88) |
|
|
BMI, mean (SD) |
1.26 (0.19) |
1.27 (0.19) |
1.25 (0.18) |
0.528 |
|
Family history, n (%) |
10 (3.58) |
3 (1.99) |
7 (5.47) |
0.157 |
|
Distance from home to school, mean (SD) |
2.46 (2.25) |
2.70 (2.63) |
2.17 (1.66) |
0.049 |
|
Schoolbag type, n (%) |
|
|
|
|
|
Single-shoulder bag |
12 (4.26) |
1 (0.65) |
11 (8.59) |
0.001 |
|
Backpack |
247 (87.59) |
141 (91.56) |
106 (82.81) |
0.027 |
|
Trolley case |
23 (8.16) |
12 (7.79) |
11 (8.59) |
0.807 |
|
Schoolbag weight, mean (SD) |
6.57 (4.10) |
4.83 (2.54) |
8.67 (4.61) |
< 0.001 |
|
Activity time, mean (SD) |
|
|
|
|
|
Extracurricular sport |
4.60 (3.53) |
4.19 (3.20) |
5.10 (3.85) |
0.030 |
|
Extracurricular activity |
2.57 (3.02) |
1.79 (3.25) |
3.52 (2.42) |
< 0.001 |
|
Study |
10.27 (6.11) |
10.76 (6.57) |
9.67 (5.46) |
0.137 |
|
Computer use |
3.38 (3.42) |
2.18 (2.18) |
4.83 (4.03) |
< 0.001 |
|
Sleeping posture, n (%) |
|
|
|
|
|
Supine |
61 (21.63) |
40 (25.97) |
21 (16.41) |
0.052 |
|
Prone |
41 (14.54) |
8 (5.19) |
33 (25.78) |
<0.001 |
|
Lateral |
180 (63.83) |
106 (68.83) |
74 (57.81) |
0.059 |
|
Calcium and milk supplement, n (%) |
|
|
|
|
|
Calcium supplement (baby) |
157 (55.67) |
92 (59.74) |
65 (50.78) |
0.132 |
|
Calcium supplement (now) |
23 (8.16) |
13 (8.44) |
10 (7.81) |
0.848 |
|
Milk supplement |
185 (65.60) |
114 (74.03) |
71 (55.47) |
0.001 |
|
Annual income/10,000, mean (SD) |
9.02 (7.69) |
7.16 (6.29) |
11.26 (8.61) |
< 0.001 |
|
Eutocia, n (%) |
205 (72.70) |
124 (80.52) |
81 (63.28) |
0.001 |
|
Risser grade, n (%) |
|
|
|
|
|
0 |
115 (89.84) |
|
115 (89.84) |
|
|
1 |
1 (0.78) |
|
1 (0.78) |
|
|
2 |
7 (5.42) |
|
7 (5.42) |
|
|
3 |
2 (1.56) |
|
2 (1.56) |
|
|
4 |
3 (2.34) |
|
3 (2.34) |
|
|
*p < 0.001. BMI: body mass index; SD: standard deviation. |
||||
Respondents reported significantly higher values for schoolbag weight, time spent playing extracurricular sports, computer time and annual family income, more often used a single-shoulder back, and less often a backpack, were younger, lived in a shorter distance from school, and were less often supplied with milk supplement. The majority of respondents had Risser grade zero.
Prevalence
Of the 128 children who were screened positive and attended the physical examination 11 diagnoses (4 boys and 7 girls) were confirmed by Cobb angles greater than 10° (8.6%). Three had thoracic curves (27.3%), 6 had thoraco-lumbar curves (54.6%) and 2 had lumbar curves (18.2%). No double curve was detected. Cobb angles of those diagnosed ranged from 10° to 28° with a mean of 13.2° (standard deviation (SD) 5.9°). Menstruation had not commenced in the 7 diagnosed-positive girls. Descriptive characteristics for the diagnosed-positive and diagnosed-negative subjects are shown in Table III.
|
Table III. Descriptive characteristics of diagnosed-positive and diagnosed-negative population |
|||
|
|
Total (n = 128) |
Diagnosed-positive (n = 11) |
Diagnosed-negative (n = 117) |
|
Male, n (%) |
93 (72.7) |
4 (36.4) |
89 (76.1) |
|
Age, mean (SD) |
9.6 (1.9) |
11.2 (1.0) |
9.5 (1.9) |
|
BMI, mean (SD) |
17.8 (2.6) |
17.8 (2.7) |
17.6 (2.1) |
|
Distance from home to school, mean (SD) |
2.2 (1.7) |
2.7 (2.7) |
2.1 (1.5) |
|
Schoolbag type, n (%) |
|
|
|
|
Single-shoulder bag |
11 (8.6) |
6 (54.5) |
5 (4.3) |
|
Backpack |
106 (82.8) |
5 (45.5) |
101 (86.3) |
|
Trolley case |
11 (8.6) |
1 (9.1) |
10 (8.5) |
|
Schoolbag weight, mean (SD) |
8.7 (4.6) |
10.7 (3.0) |
8.5 (4.7) |
|
Activity time, mean (SD) |
|
|
|
|
Extracurricular sport |
5.1 (3.9) |
4.1 (2.9) |
5.2 (3.9) |
|
Extracurricular activity |
3.5 (2.4) |
3.7 (2.7) |
3.5 (2.4) |
|
Study |
9.7 (5.5) |
9.1 (5.8) |
9.7 (5.5) |
|
Computer use |
4.8 (4.0) |
8.5 (5.0) |
4.5 (3.8) |
|
Sleeping posture, n (%) |
|
|
|
|
Supine |
21 (16.4) |
0 (0) |
21 (17.9) |
|
Prone |
31 (24.2) |
1 (9.1) |
29 (24.8) |
|
Lateral |
76 (59.4) |
10 (90.9) |
66 (56.4) |
|
Calcium and milk supplement, n (%) |
|
|
|
|
Calcium supplement (baby) |
65 (50.8) |
2 (18.2) |
63 (53.8) |
|
Calcium supplement (now) |
10 (7.8) |
1 (9.1) |
9 (7.7) |
|
Milk supplement |
71 (55.5) |
2 (18.2) |
69 (59.0) |
|
Annual income/10,000, mean (SD) |
11.3 (8.6) |
5.5 (3.1) |
11.8 (8.8) |
|
Eutocia, n (%) |
81 (63.3) |
4 (36.4) |
77 (65.8) |
Detailed weighted and unweighted overall prevalence, and prevalence by age and gender are shown in Table IV. Unweighted overall prevalence of IS for primary school students in Wuxi aged between 6 and 13 years was estimated at 0.22% (95% CI 0.13–0.39%). Adjusted for non-response, estimated prevalence was slightly lower, with a larger CI (0.21%, 95% CI 0.08–0.5%).
|
Table IV. Weighted and unweighted overall prevalence and prevalence by age and sex |
|||||
|
Age range, years |
Prevalence in boys (95% CI) |
|
Prevalence in girls (95% CI) |
||
|
Unweighted |
Weighted |
|
Unweighted |
Weighted |
|
|
6–7 |
0 |
0 |
|
0 |
0 |
|
8–9 |
0 |
0 |
|
0 |
0 |
|
10–11 |
0.35 (0.11–0.91) |
0.17 (0.04–0.57) |
|
0.59 (0.23–1.16) |
0.52 (0.1–1.44) |
|
12–13 |
0.29 (0.04–1.56) |
0.08 (0.01–0.68) |
|
1.22 (0.41–2.54) |
0.88 (0.18–2.53) |
|
Total |
0.14 (0.05–0.35) |
0.07 (0.03–0.21) |
|
0.36 (0.18–0.65) |
0.34 (0.1–0.83) |
The unweighted prevalence in girls and boys were 0.36% (95% CI 0.18–0.65%) and 0.14% (95% CI 0.05–0.35%), respectively, whereas the weighted prevalence were slightly lower (0.34% for girls, 95% CI 0.1–0.83%, and 0.07% for boys, 95% CI 0.03–0.21%). We did not find any confirmed cases in children younger than 10 years, therefore estimates can be provided only for children aged 10 years and older. Unweighted prevalence in girls increased from 0.59% (95% CI 0.23–1.16%) at the age of 10–11 years to 1.22% (95% CI 0.41–2.54%) at the age of 12–13 years. For boys, the prevalence was 0.35% (95% CI 0.11–0.91%) at the age of 10–11 years and 0.29% (95% CI 0.04–1.56%) at the age of 12–13 years.
Weighted prevalence increased from 0.52% (95% CI 0.1–1.44%) at the age of 10–11 years to 0.88% (95% CI 0.18–2.53%) at the age of 12–13 years in girls and decreased from 0.17% (95% CI 0.04–0.57%) at the age of 10–11 years to 0.08% (95% CI 0.01–0.68%) at the age of 12–13 years in boys.
Cobb angle by age and sex
Fig. 1 shows marginal predictions from weighted and unweighted regressions of Cobb angle on age, sex, and their interaction. Unadjusted for unit non-response all regression coefficients were significant at p < 0.05, demonstrating an increase in Cobb angles with age for girls but not for boys. Coefficients were no longer significant in the weighted model, indicated by a decrease in slope for girls in the weighted predictions.
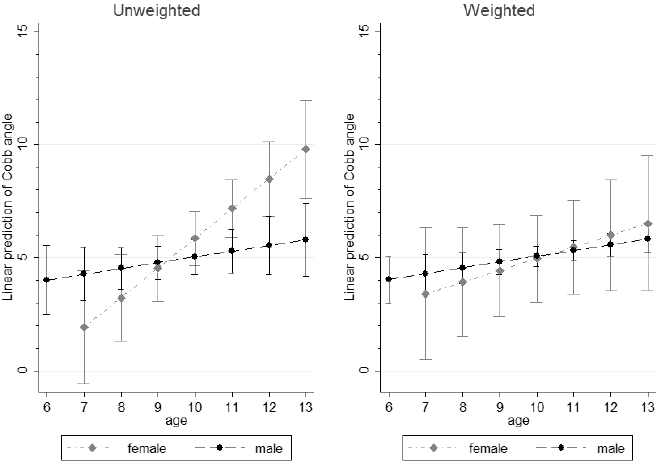
Fig. 1. Cobb angle by age and sex. Estimated means and 95% confidence intervals (95% CIs) from adjusted and unadjusted regression models of Cobb angle by age, gender (p < 0.05), and interaction (p < 0.05).
Predictors from logistic regression model
Bivariate odds ratios from logistic regression models are shown in Table V. In the unweighted models, being male, using a backpack, having received calcium supplementation as an infant, receiving milk supplementation at the time of study, and higher annual family income showed significantly decreased odds of being diagnosed, while age, using a single-shoulder bag, more time spent using a computer, and left lateral sleeping position were associated with increased odds. In the weighted models age, using a backpack, calcium supplementation as an infant, milk supplementation, and left lateral sleeping position were no longer significant. However, in addition to the other previously identified predictors, those with increased time of other extracurricular activities showed significantly increased odds.
|
Table V. Odds rations and 95% confidence intervals for variables from bivariate logistic regression models |
|||||||||
|
|
Unweighted |
|
Weighted |
||||||
|
OR |
CI Lower bound |
CI Upper bound |
p of difference* |
|
OR |
CI Lower bound |
CI Upper bound |
p of difference* |
|
|
Male |
0.18 |
0.05 |
0.66 |
0.010 |
|
0.1 |
0.02 |
0.56 |
0.008 |
|
Age |
1.87 |
1.18 |
2.99 |
0.008 |
|
1.14 |
0.79 |
1.63 |
0.485 |
|
Distance from home to school |
1.2 |
0.88 |
1.64 |
0.257 |
|
0.65 |
0.17 |
2.51 |
0.534 |
|
Schoolbag type |
|
|
|
|
|
|
|
|
|
|
Single-shoulder bag |
15.42 |
3.64 |
65.29 |
< 0.001 |
|
9.04 |
1.53 |
53.44 |
0.015 |
|
Backpack |
0.13 |
0.04 |
0.48 |
0.002 |
|
0.23 |
0.04 |
1.19 |
0.079 |
|
Trolley case |
1.07 |
0.12 |
9.24 |
0.951 |
|
0.94 |
0.09 |
9.57 |
0.956 |
|
Weight of schoolbag |
1.09 |
0.97 |
1.22 |
0.141 |
|
1.1 |
0.95 |
1.26 |
0.201 |
|
Activity times |
|
|
|
|
|
|
|
|
|
|
Extracurricular sports |
0.91 |
0.74 |
1.11 |
0.341 |
|
0.99 |
0.8 |
1.21 |
0.887 |
|
Extracurricular activities |
1.04 |
0.81 |
1.33 |
0.765 |
|
1.48 |
1.01 |
2.17 |
0.047 |
|
Study at home |
0.98 |
0.87 |
1.1 |
0.733 |
|
0.93 |
0.79 |
1.1 |
0.411 |
|
Computer use at home |
1.25 |
1.08 |
1.45 |
0.004 |
|
1.17 |
1.01 |
1.36 |
0.033 |
|
Sleeping posture |
|
|
|
|
|
|
|
|
|
|
Prone |
0.27 |
0.03 |
2.16 |
0.215 |
|
0.26 |
0.03 |
2.5 |
0.242 |
|
Left lateral |
4.65 |
1.27 |
16.95 |
0.020 |
|
0.72 |
0.13 |
4.03 |
0.711 |
|
Right lateral |
1.24 |
0.34 |
4.48 |
0.748 |
|
2.99 |
0.53 |
16.87 |
0.214 |
|
Calcium and milk supplements |
|
|
|
|
|
|
|
|
|
|
Calcium supplement (baby) |
0.19 |
0.04 |
0.92 |
0.039 |
|
1.49 |
0.19 |
11.56 |
0.704 |
|
Calcium supplement (now) |
1.2 |
0.14 |
10.46 |
0.869 |
|
0.65 |
0.06 |
6.94 |
0.722 |
|
Milk supplement |
0.15 |
0.03 |
0.75 |
0.020 |
|
0.62 |
0.1 |
3.71 |
0.604 |
|
Annual income/1000 |
0.97 |
0.95 |
0.99 |
0.009 |
|
0.98 |
0.96 |
0.99 |
0.009 |
|
Eutocia |
0.3 |
0.08 |
1.07 |
0.064 |
|
0.34 |
0.05 |
2.16 |
0.252 |
|
BMI |
0.66 |
0.02 |
21.18 |
0.814 |
|
0.03 |
0 |
2.54 |
0.123 |
|
*p < 0.001. 95% CI: 95% confidence interval; OR: odds ratio; BMI: body mass index. |
|||||||||
DISCUSSION
With an estimation of approximately 0.22%, this study found a relatively low prevalence of IS in primary school children in Beitang District, Wuxi, China. Thoraco-lumbar curves were most common. Prevalence was higher in girls than boys and there was some evidence of an increased prevalence in older girls, which was, however, not confirmed when adjustment for unit-non response was made. Apart from being female, wearing a single-shoulder bag was identified as a risk factor for diagnosis. Other potential risk factors and their interplay remain largely unclear.
The overall prevalence of IS around the world varies from 0.35% to 13% (14–19). Previous studies from China showed prevalence ranging from 0.1% in Beijing (57,581 children of 5–20 years of age) (5) to 1.77% in Heilongjiang (17,525 children of 7–15 years) (20). Similar to our study Zhang and colleagues (21) found a prevalence of idiopathic and non-idiopathic scoliosis of 0.21 in 8198 school children aged between 7 and 16 years of Hainan Province. Chen et al. (22) reported a prevalence of IS of 0.31% in 12,257 children from Jinzhou aged between 7 and 16 years. In agreement with the current study, previous Chinese studies also reported a higher prevalence in girls compared with boys (4, 5, 23) and a trend for an increasing prevalence with age (4). A factor that contributed to this finding may be the targeted age range (6–13-year-olds). In addition, Grivas et al. concluded that the prevalence of IS decreases at latitudes approaching the equator (0°) (24). Considering the relatively low latitude of Wuxi City (31.6°), it may also contribute to the low prevalence of IS detected in this study.
For the consensus in this field regarding the higher prevalence of IS in girls, the overall predicted sex ratio of girls to boys based on weighted estimates for all age groups in the current study was 5:1 (95% CI 2:1–8:1). This figure was comparable to that of previous reports (14–17), and was also comparable to a study performed by Ohtsuka et al. in Tokyo, which had an overall ratio of 7:1 (25). Wang et al. also found a prevalence ratio female to male of 7:1 in Beijing. These data may indicate a slightly higher overall prevalence ratio of female to male in Asia compared with the common finding of 2:1. Furthermore, we also found a ratio of girls to boys of 3:1 at the age of 10–11 years, but this increased to 11:1 at the age of 12–13 years. The increase in the ratio of girls to boys at the age of 12–13 years may be due to earlier onset of skeletal development for girls (growth spurt).
Prevalence does not seem to be the only characteristic of scoliosis that is influenced by gender and age. Several studies have reported higher Cobb angles in girls than in boys, indicating that scoliosis in girls progresses to a higher grade of severity (26, 27). On the other hand, IS may develop at any time during childhood and adolescence. It is most common in periods of growth spurt between the ages of 6 and 24 months, 5 and 8 years, and 11 and 14 years (28). The rate of development of spinal curvature changes most rapidly at the beginning of puberty (14, 29). In the current study, we regressed Cobb angle degrees on age, gender and their interaction and found a larger tendency of curves increase in girls than in boys with increasing age. Before 10 years of age, boys had larger curves than girls in each age group; however, the slope in girls was greater than in boys. The findings are in agreement with previous studies (2). Considering the periods of growth spurt reported by Negrini et al., boys at the age of 6–8 years and girls at the age of 11–13 years should be under careful observation. Furthermore, according to the 2011 SOSORT guidelines (3), children and adolescents with Cobb angles between 1° and 10° should be observed at least every 8 months. Thus, to avoid unnecessary radiographic exposure, it is essential to target high-risk individuals and determine optimal age groups for school screening, which could be at the age of 11–13 years for girls in whom the curves show a marked increase at growth spurt. Our findings suggest focusing mainly on children aged 10–13 years, while the screening of the 6–9-year-olds could be postponed, as this age group did not justify screening in the current study.
Finally, several predictors were found to be significantly associated with a definite diagnosis of IS from bivariate logistic regression models. For gender, the common finding that diagnosis of IS is much more frequent in females was confirmed in the current study (3). Furthermore, we found a trend of a higher possibility to be diagnosed as IS patient in individuals with increased age from the unweighted bivariate logistic regression model. This may be due to the progression of deformity curves in those who were suspicious. It was also consistent with the tendency of Cobb angle by age in the current study. Poor schoolbag design and relatively longer computer use may also influence the normal biomechanics of the spine. A biomechanical study was performed to investigate axial rotational stability of the spine under dorsally and ventrally directed shear loads (30). The results of that study showed that, at the mid and lower thoracic spine, more axial vertebral rotation occurred under dorsally directed shear loads than under ventrally directed shear loads. The authors postulated that these dorsally directed shear loads can (e.g. during growth) act as an enhancer of slight pre-existent vertebral rotation, whereas ventrally directed loads counteract rotation. This rotation-enhancing force working on the segments of the growing spine with backward inclination could result in a progressive deformation of individual vertebrae because of the Hueter-Volkmann law.
A major limitation of this study is the lack of radiographic data for a sample of screened negative children so that false-negative rates could not be estimated. Compared with previous studies (10, 11, 31), we also found a higher false-positive rate using the scoliometer. The relatively high workload may be one of the reasons that restricted the time for examination and screening specificity. This may be addressed by introducing multiple screening instruments and increasing the number of professional staff at each screening time-point in the future. Furthermore, only 11 children eventually received definite diagnosis. The results regarding predictors generated from bivariate logistic regression model should be treated with caution due to this small number. The application of multiple regression with adequate diagnostic cases is needed in future projects.
A major strength and innovation of the current study was the use of propensity scores to adjust for unit non-response. While this did not influence prevalence estimates much, some risk factors differed between weighted and unweighted analyses.
In conclusion, a relatively low overall prevalence of IS was found in primary school children in Wuxi, Beitang District. The highest prevalence was found in girls aged between 12 and 13 years. Future screening should pay more attention to females, those who use single-shoulder bags, and those who spend more time using a computer.
ACKNOWLEDGEMENTS
Funding for this study was provided by the Wuxi Science and Technology Program (WSTP), China (Grant number: ZD201408).The WSTP had no further role in study design, collection, analysis and interpretation of data, writing of the report, or the decision to submit the paper for publication.
The authors would like to express their gratitude to several people who participated in this study or supported them. First, we acknowledge Fen Qu for her invaluable support, as the screening group leader; she organized and coordinated the data collection process. Guoping Shen and Yangguang Wang are acknowledged for their professional rehabilitation and radiological insights regarding screening and diagnosis of IS. Deep appreciation is also extended to Lijie Zhang and Yingying You who participated in the data collection and processing.
REFERENCES
