Marcus Schley1, Roman Rukwied1, James Blunk1, Christian Menzer1, Christoph Konrad2, Martin Dusch1, Martin Schmelz1 and Justus Benrath1
1Department of Anesthesiology and Operative Intensive Care Mannheim, Medical Faculty Mannheim, University Heidelberg, Heidelberg, Germany, and 2Department of Anesthesiology, Kantonsspital Luzern, Switzerland
The nerve fibres underlying histamine-induced itch have not been fully elucidated. We blocked the lateral femoral cutaneous nerve and mapped the skin area unresponsive to mechanical stimulation, but still sensitive to electrically induced pain. Nerve block induced significantly larger anaesthetic areas to mechanical (100 mN pin-prick, 402 ± 61 cm2; brush, 393 ± 63 cm2) and heat pain stimuli (401 ± 53 cm2) compared with electrical stimulation (352 ± 62 cm2, p < 0.05), whereas the anaesthetic area tested with 260 mN (374 ± 57 cm2) did not differ significantly. Histamine was applied by iontophoresis (7.5 mC) at skin sites in which mechanical sensitivity was blocked, but electrical stimulation was still perceived 30 min after the nerve block (n = 9). In these areas iontophoresis of histamine provoked itching in 8/9 subjects with a mean maximum of 4.6 ± 1 (on an 11-point rating scale). Histamine-induced itch can thus be perceived at skin sites where input from mechano-sensitive polymodal nociceptors is blocked. In conclusion, input from mechano-insensitive nociceptors is sufficient to generate histamine-induced itch. Key words: pruritus; peripheral nerve block; axon reflex erythema; heat pain; mechanical pain.
(Accepted September 18, 2012.)
Acta Derm Venereol 2013; 93: XX–XX.
Martin Schmelz, Department of Anesthesiology, University of Heidelberg, DE-68167 Mannheim, Germany. E-mail: martin.schmelz@medma.uni-heidelberg.de
The neuronal pathways mediating histaminergic itch in humans have been discussed controversially for decades. However, the question appeared to be resolved after a class of mechano-insensitive C-fibres were characterized that responded to histamine application with a time course that matches the accompanying itch sensation (1). The role of mechano-insensitive afferents in histamine-induced itch was supported by experiments in the cat, reporting mechano-insensitive spinothalamic projection neurones that responded to histamine iontophoresis with a similar time course to that of itch sensation in humans (2). Moreover, recordings from a patient with chronic itch, showing spontaneously active histamine-sensitive “itch-fibres” confirmed a specific class of afferent nerve fibres for the sensation of itch (3). However, recent results from histamine stimulation in monkeys have suggested that “polymodal” (heat- and mechano-sensitive) nociceptors may also be involved in histamine-induced itch (4, 5). Mechano-insensitive C-nociceptors (CMi) cannot be activated even by strong mechanical stimuli (750 mN v.Frey) and are characterized by high electrical and thermal thresholds (6). Due to their high activation thresholds, the CMi fibres cannot be selectively stimulated for differential functional analysis. The innervation territories of CMi fibres are approximately twice as large (7) as those of polymodal nociceptors (8). Approximately 10% of the mechano-insensitive nociceptors are histamine-responsive (9). The innervation territories of these histamine-sensitive mechano-insensitive fibres cover large areas (diameters up to 8 cm (1)). In a given cutaneous peripheral nerve, these larger innervation territories of CMi-fibres may extend further than that from mechano-sensitive afferents, and thus may allow for their selective stimulation: following a complete nerve block the sensory capacity in the border zone of anaesthesia will be determined by the unblocked neighbouring skin nerves that partially overlap the innervation territories of the anaesthetized fibres. Obviously, a sensation is still provided in this zone by the unblocked skin nerves. It can be hypothesized that the larger innervation territories of mechano-insensitive nociceptors reach farther into the anaesthetized skin compared with the mechano-sensitive units with smaller innervation territories. Thus, under these conditions, one would predict that after a nerve block there is an area of skin adjacent to the completely anaesthetized skin in which mechano-insensitive fibres from unblocked neighbouring skin nerves can be excited, whereas this skin area does not contain mechano-sensitive nociceptors from the unblocked neighbouring skin nerves. In this particular zone of differential overlap, the excitation of polymodal mechano-sensitive nociceptors by, for example, pin-prick stimulation, would not be perceived, whereas high-intensity electrical stimulation, also recruiting mechano-insensitive units, should still evoke pain.
We therefore blocked the lateral femoral cutaneous nerve (LFCN) in healthy volunteers and mapped the anaesthetic areas for touch, pin-prick, heat, and electrical stimulation. When areas were identified in which mechanical stimuli were not perceived, but electrical current still evoked pain, histamine was applied in this zone. Activation of histamine-sensitive fibres was assessed subjectively by psychometric itch recording and objectively by measurement of the axon reflex flare.
Methods
Subjects
Ten healthy Caucasian male volunteers were recruited from the university and local community. Participants were 30 ± 6.3 years old (mean ± standard deviation (SD); age range 22–36 years), height 181 ± 1.9 cm, weight 74.6 ± 8.4 kg. All participants provided written informed consent in accordance with the Declaration of Helsinki prior to participation, as approved by the local ethics committee. Participants were informed about health risks of the study and instructed about risks or side-effects of a nerve block of the LFCN. In addition, written information was provided about the study procedures. Participants with atopic disposition and who were on antihistaminic medical treatment, or who had had such treatment in the past, were excluded.
Peripheral nerve block
Ultrasonography (Sonoline G40, Siemens, Germany) was used to identify the anatomical structure of the LFCN. With the ultrasound probe (11.4 MHz) scanning transversely approximately 1 cm inferior to the anterior superior iliac spine (ASIS), the sartorius muscle was identified. The LFCN runs superficially along the sartorius muscle in a tissue plane deep to the fascia lata, but superficially to the fascia iliaca.
A G27-needle was inserted in line with the ultrasound probe in between the 2 fasciae and the location of the LFCN confirmed by the patient experiencing an electric sensation (DS7A Digitimer Ltd, Welwyn Garden City, UK). After identification of the LFCN, a total volume of 1 ml mepivacaine 1% was injected, and 30 min thereafter the sensory capacity areas of the LFCN tested for different stimulus modalities (see below). LFCN blockade was always conducted by the same experimenter.
Sensory testing
Areas of anaesthesia were mapped in the following order: (i) mechanical stimuli, (ii) electrical stimuli and (iii) heat stimuli.
For mechanical stimulation, a soft brush (SenseLab brush05, Somedic, Sweden) was used to test for A-beta function and von Frey hairs (diameter 0.5 mm, Touch Test Sensory Evaluator, North Coast Medical, Morgan Hill, CA, USA) to apply touch (100 mN) and pin-prick (260 mN). The first stimulus was applied to control (not anaesthetized) skin well outside the innervation territory of the LFCN, starting at the anterior spina iliaca superior and at knee level. Stimuli were administered for 1 s and repeated in steps of 0.5 cm in approximately 0.5 Hz intervals towards the innervation territory of the LFCN. Participants kept their eyes closed and were instructed to report each mechanical stimulus (brush, touch or pin-prick) they perceived with “yes”. If no response was recorded, stimuli were administered at this site 3 times and participants inquired 4 times to respond to the stimulus with “yes” (stimulus perceived) or “no” (stimulus not perceived). A 50% false-positive estimate was set as the threshold to indicate the area of anaesthetized skin and spots were marked accordingly with a coloured pen.
Thereafter, transcutaneous electrical stimuli (5 mA, 1 Hz, 0.5 ms pulse width) were generated by a constant current stimulator (DS 7 A Digitimer Ltd, Welwyn Garden City, UK) and administered via a smooth pointed metal electrode (1 mm tip) attached to the skin. An EKG-electrode (5 cm2) served as anode and was connected to the calf of the tested leg. Stimuli were applied in the same pattern as described above for mechanical stimulation. The skin area where the pulses did not evoke a stinging sensation was marked with a pen.
Heat sensitivity, as an additional test for polymodal nociceptors, was mapped by stimulating the skin sites with a contact surface thermode (9 × 9 mm) adjusted to produce a temperature of 45ºC (MSA SenseLab, Somedic, Sweden). Initial thermal stimuli were applied well outside the anaesthetized skin sites and tested for 5 s. Subjects were instructed to report whether they perceived a heat sensation at the sites of stimulation. Stimuli were repeated in 2-cm steps towards the centre of the anaesthetized area of the LFCN. The skin area in which heat was no longer perceived was mapped thoroughly in 1 cm steps, from the non-anaesthetized towards the anaesthetized area. If heat sensation was absent, the edge of the contact surface thermode closest to the non-anaesthetized skin was marked as a border. A time interval of approximately 3 min was maintained between the tests for mechanical, electrical and heat sensation. The entire sensory testing lasted approximately 20 min. The mapping procedure was repeated 180 and 300 min after the injection.
Histamine iontophoresis
Histamine (histamine dihydrochloride, 1% in aqua dest.) was applied by iontophoresis (0.5 mA, 30 s) (WPI A360 Stimulus Isolator, World Precision Instruments, New Haven, CT, USA) via a plastic applicator (diameter 5 mm, volume 50 µl) equipped with a silver chloride electrode connected to the anode of the stimulator. A surface electrode (10 cm2) was attached distally on the subject’s leg as reference cathode.
The stimulation was applied after the complete mapping procedure, approximately 50 min after the injection of 1 ml mepivacaine 1%. The exact site of histamine administration was chosen according to the mapped mechano-sensitive and electro-receptive fields. The maximum distance between the larger area insensitive for mechanical stimuli, and the smaller area insensitive for electrical stimuli, was located. Within this field, the iontophoresis applicator was placed inside the mechano-negative, but electro-positive, area. In particular, it was carefully located at the border of electrical sensitivity and towards the completely anaesthetized zone, in order to minimize the stimulation of neighbouring mechano-sensitive fibres. After the final itch rating, the mechano-insensitivity of the application site was tested with a 260 mN v.Frey filament.
Activation of histamine-responsive mechano-insensitive fibres was assessed by measuring the area of the axon reflex by laser Doppler imaging (Moor LDI, Axminster, UK). Two images were registered as baseline followed by histamine iontophoresis (0.5 mA, 30 s), and immediately afterwards 2 laser Doppler imagin scans of blood flow were captured to assess the histamine-induced axon reflex. Axon reflex areas were defined as pixels that increased their flux values > 2 SD in the baseline scan (10). Subjects were asked to rate the intensity of the itch sensation on an 11-point numerical rating scale (NRS), from 0 (“no itch”) to 10 (“maximum itch imaginable”) and maximum itch-ratings were recorded.
Evaluation of the mapped areas
Skin markings were transferred to a transparency and scanned at 300 dpi. In addition, a photograph was taken with a digital camera for documentation. Areas were assessed off-line by a Digital Imaging and Communications in Medicine computer program (OsiriX, Apple).
Statistics
All data were analysed using the software package Statistica 6.0 (StatSoft, Tulsa, OK, USA). Two-way analysis of variance (ANOVA) and repeated measures with “anaesthesia area” as categorical factor were used. Differences between areas anaesthetic to electrical stimulation and those anaesthetic to the other stimuli were calculated by Fisher’s least significant difference test. Correlations between itch ratings and flare area were analysed by Spearman’s rank correlation, with values of p < 0.05 considered significant.
Results
Differential local anaesthetic effect
The injection of local anaesthetic provoked an anaesthetic area in the thigh in each of the 10 subjects. In 1 out of 10 subjects, however, no areas of differential innervation territories could be identified. It was notable that in this subject the anaesthetized zones were relatively small, 82 ± 24 cm2 (mean ± standard error of the mean (SEM)), thus indicating failure of a successful nerve block, and thus this subject was omitted from the analysis. The areas anaesthetic (n = 9) to electrical stimulation were the smallest (352 ± 62 cm2), whereas those anaesthetic to brush (393 ± 63 cm2), punctate mechanical stimuli of 100 mN (402 ± 61 cm2) and heat (401 ± 53 cm2) were the largest (p < 0.05). The area anaesthetic to pin-prick stimulation of 260 mN (374 ± 57 cm2) was not significantly larger than the one anaesthetic to electrical stimulation. It is noteworthy that heat stimulation was negative in 4 of the subjects and only weak heat pain was evoked when the 45ºC heat stimulus was kept on the skin for more than 10 s. Fig. 1 shows a specimen of the anaesthetic zones mapped with mechanical, heat and electrical stimuli. The largest distance between the electrically sensitive field and the pin-prick negative mechano-receptive field was 5.5 cm. The minimum distance between positive transcutaneous electrical stimulation and negative pin-prick sensation was 2 cm (mean maximum distance 4.8 ± 1.2 cm). The mechano-insensitivity of the histamine application site was maintained in all 9 subjects after the last itch rating. At 180 min (approximately 100 min after the histamine protocol), the anaesthetic areas had declined by 31–54% (100 mN –40%, 260 nM –44%, brush –45%, electrical stimuli –54%, heat –31%). Sensory loss had subsided after approximately 300 min.
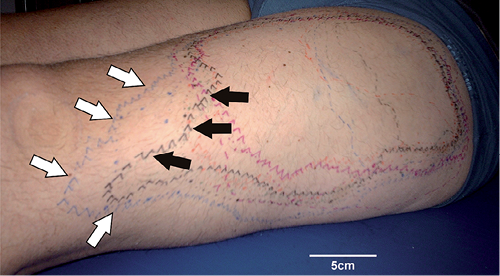
Fig. 1. Specimen of mapped anaesthetic areas 30 min after block of the lateral femoral cutaneous nerve. The borders of mechanical, heat, and electrical sensitivity were mapped and marked on the skin. In 9 out of 10 subjects a particular area was mapped in which mechanical sensitivity was absent (white arrows), but electrical stimuli still perceived (black arrows).
Histamine iontophoresis and itch intensity
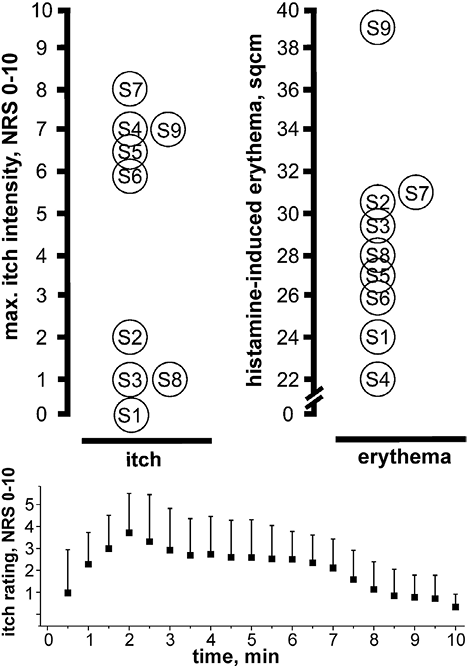
Histamine administered iontophoretically at the border of differential sensitivity to mechanical and electrical stimuli (Figs 1 and 2) evoked a large axon reflex erythema of mean 28.9 ± 1.6 cm2 in 9 subjects. In 8 out of the 9 tested subjects, an itch sensation was perceived, which gradually increased and reached peak levels after 1–2 min (Fig. 3). A mean maximum itch intensity of 4.6 ± 1.2 (NRS) was recorded (Fig. 3). The subject who did not perceive any itch (S1 in Fig. 3), however, responded with a moderate histamine-evoked erythema. No correlation between the area of axon reflex erythema and maximum itch ratings was found (Spearman’s rank correlation coefficient 0.22, n.s.).
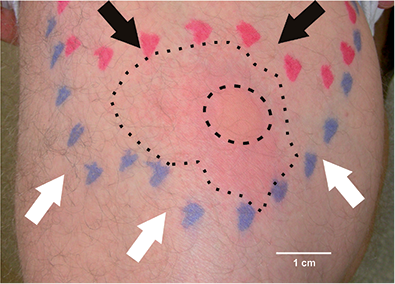
Fig. 2. Histamine iontophoresis was performed within a skin area anaesthetic to pin-prick (white arrows), but sensitive to electrical stimulation (black arrows). Iontophoresis caused a wheal (inner dashed circle) and erythema (outer dotted circle).
Fig. 3. Histamine iontophoresis within a skin area anaesthetic to pin-prick, but sensitive to electrical stimulation induced clear itch sensation in 8 of 9 subjects (left panel) and an axon reflex erythema in all of them (right panel). Time course of itch ratings following histamine iontophoresis is shown in the lower panel. NRS: numerical rating scale (mean + sem, n = 8).
Discussion
Local regional block of the LFCN provoked a large anaesthetic skin area at the lateral thigh, the borders of which were determined by the innervation territories of the neighbouring non-blocked skin nerves. The areas anaesthetic to mechanical stimuli were larger than those anaesthetic to strong electrical stimulation. Thus, the outer part of the anaesthetic skin area was anaesthetic to mechanical stimulation, but electrical stimuli were still perceived as painful. This suggests that mechano-insensitive fibres from neighbouring, non-anaesthetized skin nerves were reaching farther into the innervation territory of the blocked LFCN compared with mechano-sensitive nociceptors. Hence, after the LFCN block, we identified skin areas in which input from mechanically sensitive nociceptors was blocked, whereas input from electrically stimulated mechano-insensitive nociceptors was still perceived. Histamine application in these mechano-insensitive but electrically sensitive areas provoked an itch sensation in 8 out of 9 subjects, indicating that activation of mechano-insensitive nerve fibres was sufficient to provoke histamine-induced itch.
Pruriceptors vs. polymodal nociceptors
Previous studies in cat (2) and man (1, 9) suggested that histamine-induced itch is mediated by a specific subtype of mechano-insensitive C-fibres. Recently, activation of polymodal nociceptors has been described in monkeys after the injection of histamine (4, 5). It is possible that the different mode of histamine application, i.e. injection compared with iontophoresis, leads to higher peak concentrations, and thereby contributes to these differences. Our results do not rule out that activation of polymodal nociceptors by histamine injection plays a role in the ensuing itch sensation. Thus, we can conclude that activation of mechano-insensitive nociceptors is sufficient to provoke histamine-induced itch. However, our study cannot prove that the activation of mechano-insensitive nociceptors is necessary to provoke histamine-induced itch.
A similar time course of itch and pain was generated by applying cowhage spicules, native or inactivated, covered with either histamine or capsaicin (11). As no qualitative difference in the itch reaction of these mediators was found, a common neurophysiological pathway of itch was proposed (11). This common pathway may not include mechano-insensitive fibres, as their activation is functionally linked to the generation of an axon reflex flare erythema (12), and this was not observed following cowhage application (13). Thus, as the results in monkeys show (4, 5), polymodal nociceptors could also be candidates to mediate histamine-induced itch.
Overlapping innervation territories of nociceptors
Our results demonstrate that a peripheral nerve block induced very similar anaesthetic areas for touch and pin-prick. The borders of the anaesthetic areas are determined by the non-blocked skin nerves adjacent to the LFCN. The receptive field sizes of human polymodal nociceptors are approximately 1–2 cm2 in the lower leg (8), with a mean maximal diameter of approximately 20 mm. This extent matches that measured for mechano-sensitive A fibres in the human arm (14). Thus, similar anaesthetic areas for touch and pin-prick are in line with the similar size of innervation territories of the primary afferents. In contrast, mechano-insensitive C nociceptors have larger receptive fields with median diameters of 46 mm (7). Thus, it might be predicted that mechano-insensitive units of neighbouring (not anaesthetized) skin nerves would extend further into the skin innervated by the blocked LFCN according to the larger spatial extent of the innervation territory of mechano-insensitive units. In addition, there is evidence that very strong mechanical stimulation (260 mN) can produce some activation of C-nociceptors normally classified as mechano-insensitive (15, 16); this might explain the rather small anaesthetic areas found for stimulation with a 260 mN filament.
Skin areas anaesthetic to heat could not be tested with the same spatial resolution as for mechanical stimuli. We would expect them to match those observed for pin-prick stimulation, based on the role of polymodal nociceptors for mechanically and heat-induced pain (17). However, there is also some heat responsiveness in mechano-insensitive nociceptors, albeit their activation thresholds are higher (42ºC to > 50ºC) (7, 9, 18). Given that our test stimulus for heat was only 45ºC, it may have activated just a few of the mechano-insensitive nociceptors, which can explain the delayed response and the lower intensity of heat pain in some of the investigated subjects.
Histamine-induced itch in skin sites anaesthetic for mechanical stimulation
Histamine iontophoresis in skin areas that were anaesthetic for mechanical stimulation provoked itch in 8 of the 9 subjects. This observation suggests that the activation of mechano-insensitive C-fibres is sufficient to cause histamine-induced itch. As pin-prick stimulation was not perceived at the histamine application site, it can be assumed that polymodal nociceptors were either completely blocked or that non-blocked polymodal fibres of neighbouring nerves were not sufficient to convey the pin-prick sensation. As activation of mechano-sensitive fibres at the suprathreshold level for 1 s did not provoke an overt sensation in the partially anaesthetized skin, we would conclude that these mechano-sensitive fibres did not crucially contribute to the itch sensation elicited from the same skin site. However, we cannot completely exclude that tonic discharge of polymodal nociceptors might still contribute to the histamine-induced itch. Although we cannot exclude a role of mechano-sensitive nociceptors, we conclude that histamine can induce itch without overt input from mechano-sensitive nociceptors. Our results therefore support the idea that activation of specific mechano-insensitive histamine-sensitive primary afferents is sufficient to provoke histamine-induced itch.
Itch intensity is encoded by the discharge frequency of the action potentials. Interestingly, the axon reflex flare, mediated by mechano-insensitive nociceptors (12), is already maximum at frequencies of approximately 2–5 Hz (19). Bursts of action potentials at higher frequencies are therefore expected to cause intense itching, but not necessarily a further increase in the axon reflex erythema. Even though higher discharge frequencies increase the release of vasodilatory neuropeptides, such as calcitonin gene-related peptide (CGRP) (20), due to the very steep dose-response curve for CGRP-induced vasodilation (21), CGRP levels might readily become supra-maximum, which explains the homogenous level of vasodilation inside the axon reflex flare (22).
The present study does not exclude a role for polymodal nociceptors in histamine-induced itch, as the activation of polymodal nociceptors by a histamine injection might contribute to the ensuing itch sensation. Moreover, activity of polymodal nociceptors stimulated by cowhage (23) exemplifies the potential of this nociceptor class for the induction of itch. However, previous work suggested that mechanically induced pain accompanying a histamine injection would reduce rather than increase the histamine-induced itch (24). Regardless of a possible contribution of polymodal nociceptors to the histamine-induced itch it should be pointed out that the existence of a histamine-specific pathway of primary afferents and spinal cord neurones for itch does not provide evidence against other, non-specific, pathways for itch (25) that may be activated by cowhage (4, 5) or even capsaicin (11). On the other hand, clinically, histamine-independent pathways of itch are considered crucial in the pathogenesis of chronic itch (26).
In summary, this study demonstrates that blocking the LFCN results in differential areas of anaesthesia to mechanical and electrical stimuli. In skin sites negative to mechanical stimulation, but positive to electrical stimulation, histamine application evokes a typical itch and flare response. We conclude that mechano-insensitive nociceptors are sufficient to provoke histamine-induced itch.
Acknowledgements
This study was supported by the “Kompetenzzentrum Schmerz” Baden-Wuerttemberg, Germany.
The authors declare no conflicts of interest.
References
