Yonit Wohl1,3, Julio Wainstein2,3 and Yosefa Bar-Dayan2,3
1Department of Dermatology, Barzilay Medical Center, Ashkelon, 2Diabetes Unit, Wolfson Medical Center, Holon, and 3Sackler School of Medicine, Tel Aviv University, Tel Aviv, Israel
Prevalence of atopic dermatitis (AD) has been studied mainly in infants and young children. Surveys on AD in adolescents and adults are based largely on self-reported questionnaires and cohorts of different ages, precluding meaningful comparisons and conclusions. We aimed to provide data on the prevalence of AD and its relation to demographic factors in adolescents. A large database of youths eligible for military service was searched for subjects one year before service at the age of 18. Parameters included country of origin, age of immigration to Israel, education, and presence and severity of AD. AD rose 3-fold for both genders over the study period 1998–2008 (0.4–1%). The increase was higher in females (0.5–1.2%) than males (0.3–0.9%). Mild and moderate disease was higher in females than males. Severe AD was rarely noted. Russian (OR 1.47), Asian (OR 1.15) and Euro-American (OR 1.26) origins had highest association with AD. Immigrants after age 7 had higher association with AD than Israeli-born subjects and immigrants before age 7 (OR 1.88 vs. 1.33). More years of education was significantly associated with high prevalence of AD. The increased prevalence of AD during the 11-year period underscores the need for awareness of the condition in this population, in which AD affects health and quality of life. Key words: atopic dermatitis; adolescents; immigration; education.
Accepted Feb 4, 2014; Epub ahead of print Feb 17, 2014
Acta Derm Venereol
Dr. Yosefa Bar-Dayan, Diabetes Unit, Wolfson Medical Center, Tel Aviv University, IL-52100 Holon, Israel. E-mail: bardayan@netvision.net.il
Atopic dermatitis (AD) is an intensely itchy, chronic eczematous disorder stemming from complex genetic and environmental factors that contribute to an excessive immune response to allergens coupled with epidermal barrier dysfunction. It is very common worldwide in all ages, and has shown an increasing prevalence over the past decades. Characteristic morphologic appearance and prevalence of the disorder change with the age of the patient. Data on childhood atopic dermatitis show a range of 1-year period prevalence rate from less than 2% in Iran and China, to around 10–12% in the US, and 20% in Western Europe and Australia (1–3).
The disease can persist or even begin in adulthood, and adolescents are a particularly vulnerable population with respect to the course of the disease and its impact on everyday life. The financial cost of AD is great due to both direct medical costs and indirect costs such as time lost from work or school.
Research on AD has focused mainly on infancy and early childhood, with few epidemiology surveys in adolescents carried out mainly in Germany (4), Latin America (5) and Asia (6, 7). In Germany, a recent prospective cohort study on 2,857 adolescents reported an incidence in 16–20-year-olds of 1.7% (4). In Latin America, a cross-sectional, questionnaire-based survey of 165,917 adolescents 13–14-years-old yielded a high prevalence of the skin disorder: mean prevalence in the entire sample was 10.6% (5). The point prevalence of AD in 2,032 Korean adults examined by dermatologists in a single medical centre was 2.6% (6), while the overall 8-year prevalence of AD was 6.7% in a national retrospective cohort study in Taiwan based on almost 100,000 enrollees from the national insurance register between the years 2000–2007 (7).
The available data on AD is based largely on surveys that employed self-reported questionnaires and mixed age groups of participants, raising the possibility of recall, response and diagnosis biases. The medical screening for universal military service in the Israeli Defense Forces described here offers a unique opportunity to study the prevalence of AD in adolescents in a less biased manner in a cohort composed entirely of 17-year-olds – the age when adolescents are screened prior to compulsory military service that begins at the age of 18. The aim was to investigate the relationship between disease prevalence and demographic factors in adolescents over a period of 11 years, in order to identify trends and associated risk factors.
PATIENTS AND METHODS
Subjects
The screening process of all Israeli Jewish citizens for compulsory military service begins at the age of 17. It includes individuals with conditions that will eventually exclude them from service, and does not include Israelis of Arab origin and Ultra-Orthodox Jews, who are exempt from military service. An extensive evaluation including medical and socio-demographic profiling is performed at this time, about one year before actual recruitment at the age of 18.
The retrospective study covered all candidates for military service who appeared at the recruitment office between January 1, 1998 and December 31, 2008. The demographic parameters gathered from the database included country of origin, date and age of immigration to Israel, and years of education. The medical parameters – mainly the presence and severity of AD – were compiled from the database which included physical examination by a general practitioner and, in case of a cutaneous disorder, by a dermatologist.
Demographic parameters
Country of origin was defined as the subject’s father’s country of birth. If this was Israel, it was defined according to the paternal grandfather’s country of birth. The data were then categorised into 6 groups: 1) Euro-American - including Europe, the Americas, Australia and South Africa; 2) Asian – including Asia, Turkey, but not Russia, 3) Africa, 4) Ethiopian, 5) Russian and the former Soviet Union republics, and 6) Israeli – both father and paternal grandfather born in Israel.
The subjects were classified into 2 subgroups: born in Israel or born elsewhere and immigrated to Israel after the age of 7 years.
Level of education was the number of school years: < 10, 10–12, and > 12.
Medical surveillance
General practitioners performed the medical examinations at the recruiting office. Demographic characteristics and medical history were obtained from a comprehensive pre-examination questionnaire, a review of the primary care physician’s record on the subject, and an in-depth interview that included questions about cutaneous symptoms. This was followed by a general physical examination. Conscripts with known or suspected AD were referred to a dermatologist for further evaluation to determine the diagnosis.
Disease categories
The criteria for cutaneous disease categories were established in 1997 primarily to identify the conscript’s suitability for combat duty. AD is diagnosed based on history and clinical features of itchy eczematous lesions, xerosis and lichenification of the skin with characteristic distribution. In the military health system, AD is divided into 5 groups according to the severity of the disease, with grades from 1 to 5 in increasing severity (Table I). Subjects in our study were further divided into 3 groups as follows: mild disease including grades 1 and 2, moderate disease including grade 3, and severe disease including grades 4 and 5.
Table I. Classification of atopic dermatitis by severity, rated from 1 to 5 with increasing severity
|
Grade |
Criteria |
|
1 |
History of atopic dermatitis, disease remission for at least a year |
|
2 |
Rare disease exacerbations, involvement of minimal skin area, sporadic topical treatment with good response |
|
3 |
Frequent disease exacerbations, on-going topical treatment or systemic treatment for long periods |
|
4 |
Severe exacerbation with large skin involvement and severe functional impact, intensive treatment intervention is mandatory |
|
5 |
Extensive skin involvement with severe functional impairment, partial response to on-going or recurrent treatment |
Statistical analysis
Chi-square analysis of AD prevalence was carried out with SPSS software, version 15; p < 0.05 was considered significant. Logistic regression models were used to measure the association between AD and the various parameters in a multivariate analysis.
RESULTS
Of the 845,326 17-year-olds registered in the database, 339,427 (41%) were females and 505,899 (59%) were males. The overall prevalence of AD was 0.5% among males and 0.7% among females (p < 0.01). The prevalence of AD steadily and significantly increased for both genders over the 11-year study period, from 0.3–0.9% for males and from 0.5–1.2% for females (Fig. 1).
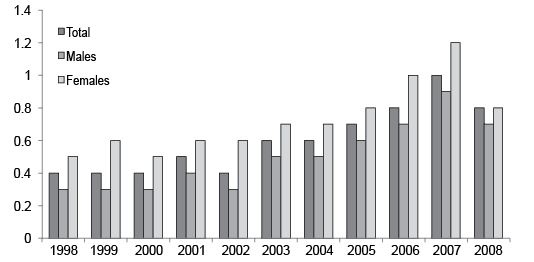
Fig. 1. Percentage of adolescents with atopic dermatitis during each year of the survey (n = 4,891).
During the entire 11-year study period there was a significantly higher increase in disease rates in females compared to males. The prevalence of mild disease was higher among females than males, while a significant increase in the prevalence of mild disease was noted for both genders: 0.2–0.7% in males and 0.4–0.9% in females, p < 0.01. Moderate disease was more prevalent in women than in men (0.2% versus 0.1%, p < 0.01), but rates remained stable over the years for both genders (Fig. 2). Severe AD was noted very rarely, in 0.006% of the subjects.
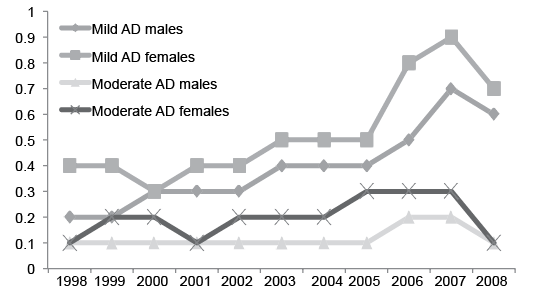
Fig. 2. Percentage of adolescents with atopic dermatitis according to severity and gender during each year of the survey (n=4,891).
Country of origin and immigration influenced disease prevalence in both genders (Table II). Asian and Euro-American origins were associated with a higher prevalence of AD compared with African and Ethiopian origins (OR 1.15 and OR 1.26 versus OR 0.80 and OR 0.27, respectively, p < 0.01). Origin in Russia and the republics of the former Soviet Union demonstrated the highest association: OR 1.47, 95% CI 1.26–1.59, p < 0.01. Immigration to Israel after the age of 7 years was associated with significantly higher prevalence of the disease in comparison to Israeli born subjects and immigrants before the age of 7 years: OR 1.88, 95% CI 1.73–2.045, p < 0.01 vs OR 1.41, 95% CI 1.33–1.49 (p < 0.01), respectively.
Table II. Logistic regression for atopic dermatitis adolescents (n = 4,891)
|
Variable |
OR |
95% CI |
p-value |
|
Russian origin |
1.41 |
1.26–1.59 |
< 0.001 |
|
European or American origin |
1.26 |
1.12–1.41 |
< 0.001 |
|
Asian origin |
1.15 |
1.022–1.29 |
< 0.001 |
|
African origin |
0.80 |
0.71–0.91 |
0.001 |
|
Ethiopian origin |
0.27 |
0.19–0.40 |
< 0.001 |
|
Immigrant after 7 years of age |
1.88 |
1.7–2.04 |
< 0.001 |
|
Israeli born and immigrant before 7 years of age |
1.41 |
1.33–1.49 |
< 0.001 |
|
10–12 years of education |
2.63 |
1.91–3.63 |
< 0.001 |
|
> 12 years of education |
11.76 |
8.31–16.64 |
< 0.001 |
|
Female gender |
1.79 |
1.68–1.90 |
< 0.001 |
More years of education was significantly associated with high prevalence rates of AD: OR 2.63 for 10–12 years of education and OR 11.76 for more than 12 years of education, p < 0.01.
The female part of the cohort exhibited stronger associations between all the above variables and AD than the males: OR 1.79, 95% CI 1.68–1.90, p < 0.01.
DISCUSSION
Reported prevalence rates during adolescence and adulthood are 0.9–4% (1, 3). According to recent data, recurrence rate is around 2.4%, and high persistence rates of disease symptoms are seen among adolescents, especially those with positive personal or parental atopic background, high socioeconomic status, female gender, and high-risk occupations (4).
In our large population-based cohort study, the overall annual point prevalence of AD was 0.5% in males and 0.7% in females. During the decade of the study, there was a 3-fold increase in the prevalence rate of the disease in both genders.
These trends are in line with the current, constantly changing map of the epidemiology of the disease around the world. In a recent comprehensive multi-centre study aimed at determining worldwide trends in AD in a cohort of around 4,900,000 children and adolescents, Williams and coworkers (8) showed mixed patterns: a decrease in prevalence rates from some previously high-prevalence countries in the developed world including the UK and New Zealand, and a continuous increase in Russia and the developing countries of South America, Kenya, Southeast Asia and the Far East (8, 9). The data on our adolescent population show certain similarities to reported prevalence rates, which are high in parts of the western world and rising on the Asian and African continents.
Explaining these global trends requires addressing the complex interplay between genetic and environmental factors in the development of the disease. For example, the most studied pathogenic factor in recent years is filaggrin – a key protein involved in maintaining skin barrier function and hydration. Mutations in the filaggrin gene (FLG) cause ichthyosis vulgaris and are a major predisposing factor for AD in individuals of European and Asian descent, but less commonly in the Ethiopian population (10–12). Nevertheless, demographic changes, especially in urban areas, and environmental risk factors for AD such as improved personal hygiene and exposure to various cosmetic and industrial antigens seem to account in large part for the changing epidemiology of AD.
The population we studied is unique, being composed of diverse ethnic-genetic backgrounds from several waves of immigration over the past several decades. The population also reflects the phenomena of a rapidly developing country undergoing urbanisation and rapid economic changes.
We found AD in adolescents to be independently and highly associated with the country of origin, immigration age, and level of education – findings in line with the known increase in the frequency of the disease among people who immigrate to developed countries from underdeveloped countries (13). The Israeli population experienced huge waves of immigration from different parts of the world, the largest from Russia, and, indeed, these immigrants were found to have the highest risk for AD, although similar levels were seen for those immigrating from the US, South America and Ethiopia.
In addition to atopic genetic divergence, education levels were associated with AD in our adolescents. Change in family size, personal hygiene, and increasing use of cosmetics together with the absence of chronic exposure to microbial antigens in early life and the increasing exposure to occupational and industrial allergens are only some of the other possible contributory factors.
Interestingly, we found consistent and significant gender differences in the prevalence ratios – in line with our previous findings of female predominance in most subtypes of inflammatory and autoimmune diseases (14). This may be the consequence of the immune response in women, which renders them more prone to inflammatory and autoimmune diseases then men, and the use of cosmetic products (15, 16).
Strengths and limitations of this study
Population-based epidemiological studies of AD are usually performed on representative samples, often leading to inaccuracies. The retrospective nature of our study also constitutes a limitation. The strength of the study lies in its being based on national data that include valid measurements for almost the entire population of a certain age, conducted by physicians according to specific definitions of clinical diseases and their symptoms and diagnoses. Additional strengths are the magnitude of the population and its demographic and ethnic diversity, which add an extra dimension to the study.
Heath policy implications
The health status of teenagers not only reflects the cumulative incidence of diseases throughout childhood, but also helps predict the health status of adults and estimate its impact on health services. This is especially important for the military where adolescents are the majority.
AD has a chronic relapsing course and can greatly influence the quality of life of those affected. Moderate and severe AD, which constitutes about 20% of patients according to the literature and reflected in our study, may lead to loss of self-esteem, depression and poor job or school performance, as well as interference with family and social life. Because the morbidity of dermatologic disease often results in an inability to work or an adverse effect on social function, these disease patterns among adolescents have relevance for the planning of manpower and services in numerous settings, be it the military, educational institutions, the work place, or general health services. It is also noteworthy that atopy is a well-established risk factor for allergic contact dermatitis (17).
Whether due to changes in demography or lifestyle habits, the marked rise in AD in the adolescent and young adult populations reflected in our study requires public health planning and allocation of resources for the prevention, management and further research of the disease.
The authors declare no conflict of interest.
REFERENCES
