Akihiko Asahina1, Atsuko Niizuma1, Ayaka Ohzono2, Norito Ishii2, Hiroshi Koga2 and Takashi Hashimoto2
Departments of Dermatology, 1National Hospital Organization Sagamihara National Hospital, 18-1 Sakuradai, Minami-ku, Sagamihara, Kanagawa 252-0392, and 2Kurume University School of Medicine, and Kurume University Institute of Cutaneous Cell Biology, Kurume, Japan. E-mail: asahina-tky@umin.ac.jp
Accepted May 20, 2014, Epub ahead of print May 23, 2014
Pemphigoid nodularis is a variant of bullous pemphigoid (BP) with the clinical manifestation of prurigo nodularis, and it may lack clinical blistering (1). We present a case of this entity showing papillary neutrophilic microabscesses histopathologically, together with a diverse set of IgG, IgA, and IgE autoantibodies in the sera.
CASE REPORT
A 47-year-old Japanese woman presented with severely pruritic lesions which had persisted for 7 months. Excoriated and crusted nodules were distributed on the trunk and extremities. Small tense blisters and erosions were also observed, and the upper palate was involved (Fig. 1a,b). Laboratory examinations revealed eosinophilia (11.5%: 1,232/mm3) and increased levels of serum CRP (3.24 mg/dl; normal: 0–0.4 mg/dl) and IgE (15,700 IU/ml; normal: 0–250 IU/ml). A biopsy specimen of a nodule showed hyperkeratosis, acanthosis, and perivascular infiltration with subepidermal cleft formation. The papillary dermis showed accumulation of neutrophils with some eosinophils (Fig. 1c). Another biopsy from a blister confirmed its subepidermal localisation, and inflammatory cells included more eosinophils than neutrophils (Fig. 1d). Direct immunofluorescence (DIF) revealed linear basement membrane zone (BMZ) deposits of IgG, IgA and C3, while neither IgM nor IgE was deposited (Fig. S1a,b1). Indirect immunofluorescence (IIF) showed IgG anti-BMZ antibodies, and the presence of IgA anti-BMZ antibodies was also detected weakly. On IIF of 1M NaCl-split-skin sections, both IgG and IgA antibodies bound to the epidermal side (Fig. S1c,d1), however, IIF failed to detect IgE anti-BMZ antibodies. Enzyme-linked immunosorbent assays (ELISAs) revealed circulating IgG antibodies to the BP180 NC16a domain (index 108.67; normal < 9) and the N- and C-terminal domains of BP230 (index 101.04; normal < 9). ELISAs also revealed IgA antibodies to BP230 (optical density (OD) 0.256; normal < 0.15), but not to BP180 (OD 0.07; normal < 0.15). By novel ELISAs using the same recombinant proteins (Ohzono A et al., unpublished data), IgE antibodies reacted with both BP180 (OD 1.37; normal < 0.141) and BP230 (OD 2.044; normal < 0.021). Immunoblotting of normal human epidermal and dermal extracts revealed IgG antibodies to the 190 kDa periplakin, without other reactivities of IgG, IgA and IgE antibodies. However, immunoblotting detected IgG, but not IgA or IgE, antibodies reactive with the BP180 NC16a domain (Fig. S2a1), with no reactivity to the BP180 C-terminal (BP915) domain (Fig. S2b1). By using concentrated culture supernatant of HaCaT cells, IgG, but not IgA or IgE, antibodies to the 120 kDa LAD-1 were detected (Fig. S2c1).
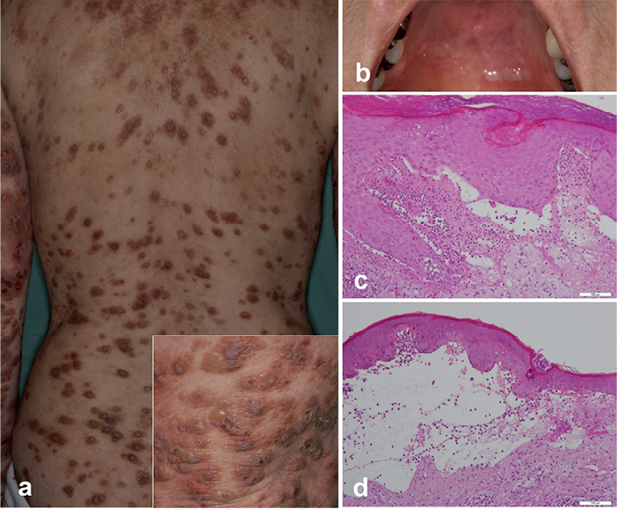
Fig. 1. Clinical manifestations and histopathological findings. Elevated pruritic nodules on the back studded with excoriations and occasional crusts. Insert shows small tense blisters on the right arm (a). Erosion on the upper palate (b). Biopsy specimen of a pruriginous nodule on the right lower arm showing subepidermal cleft formation and papillary neutrophilic microabscesses (c), and an adjacent blister showing its subepidermal localisation with inflammatory cells consisting of more eosinophils than neutrophils (d) (Haematoxylin-eosin stain; original magnifications × 200).
The patient had rheumatoid arthritis (anti-CCP antibody: 12.3; normal: 0–4.4 U/ml), but it was under good control with auranofin and etodolac, and there was no history of taking penicillamine or bucillamine that might induce autoimmune blistering diseases. Under the diagnosis of pemphigoid nodularis, systemic prednisolone 40 mg (1 mg/kg)/day started, together with minocycline and nicotinic acid. Although she responded well to therapy, some blisters recurred afterwards. Subsequently, with prednisolone 7.5 mg/day and cyclosporine 50 mg/day, pruritic nodules were still persistent, but bullous lesions were no longer observed.
DISCUSSION
Considering that DIF and IIF showed IgA anti-BMZ antibodies, our patient can be diagnosed with linear IgA/IgG bullous dermatosis (LAGBD), or a variant of linear IgA bullous dermatosis (LABD) (2). Interestingly, immunoblotting analysis, IgG, but not IgA, antibodies was shown to react with LAD-1, the shedded ectodomain of BP180. While it is possible that IgG antibodies simply recognised the overlapping portion of the NC16a domain and the LAD-1 molecule (3), one can consider the possibility of development of IgG antibodies to multiple epitopes. In BP, intra- and intermolecular epitope spreading occurs relatively frequently, probably as a result of the inflammatory cascade and tissue damage caused by autoimmunity to an initial epitope (3). Intense scratching of pruritic nodules may have released and exposed BP180 and its shedded form to immunocompetent cells, and brought about this particular epitope spreading. The contribution of IgG antibodies against LAD-1 remains speculative, but in mucous membrane pemphigoid, patients with the dual IgG reactivity to full-length BP180 and LAD-1 exhibited most severity (4).
As for IgA antibodies, ELISA showed their reactivity with BP230, but not BP180. This is in agreement with a previous ELISA study revealing the presence of IgA reactivity with BP180 and/or BP230 in BP sera (5). Repeated scratching apparently facilitated autoimmunity against intracellular BP230 and class-switching from IgG to IgA. Our patient had an oral mucosal lesion, and this finding may be supported by circumstantial evidence for a relationship between IgA autoantibodies and the presence of mucosal inflammation (4). In addition, there are cases of LABD showing the clinical picture of prurigo nodularis, suggesting that IgA antibodies themselves may cause pruriginous lesions. However, IgA typically targeted LAD-1 in those cases (6, 7), and IgA anti-BP230 antibodies could be non-pathogenic.
The presence of neutrophilic microabscesses herein suggests that IgA antibodies may recruit neutrophils by cross-linking IgA FcR (FcαRI). Indeed, serum IgA autoantibodies from patients with LABD recruited and activated human granulocytes, mostly neutrophils, at the dermo-epidermal junction in cryosections of human skin (8). There is a report of an atypical case of LABD with IgA anti-BP230 antibodies, who showed non-bullous, subacute prurigo-like eruptions with neutrophilic microabscesses without dermo-epidermal separation (9).
Finally, our patient had IgE antibodies against BP180 NC16a domain and BP230. These antibodies may be present in BP sera, and IgE anti-BP180 antibodies are related to the activity of BP (10). The lack of IgE staining at the BMZ by DIF and IIF was also documented previously (11). Aside from some technical issues, IgE anti-BP180 may preferentially act by binding to high affinity FcεRI; IgE autoantibodies cause activation and degranulation of mast cells, basophils and eosinophils via FcεRI, leading to release of histamine, proteases and other inflammatory mediators. This possibility was strengthened in a model using human skin grafted onto nude mice injected intradermally with BP IgE (12). Intense pruritus in our patient may be a result of such mechanism, forming a vicious itch-scratch cycle.
In conclusion, pemphigoid nodularis may present with numerous autoantibodies, speculatively because of intense scratching of pruritic nodules followed by the release and exposure of multiple antigens. The comorbidity of our patient, i.e. rheumatoid arthritis, also implies predisposition to autoimmunity. Neutrophil infiltration may be linked to the presence of IgA autoantibodies. To address these hypotheses, immunological profiles of pemphigoid nodularis warrant further investigation, as well as the roles of IgA and IgE antibodies.
The authors declare no conflict of interest.

1http://www.medicaljournals.se/acta/content/?doi=10.2340/00015555-1900
REFERENCES
