Melanie Weiss1, Thomas Mettang2, Ulrich Tschulena3, Jutta Passlick-Deetjen4 and Elke Weisshaar1
1Department of Clinical Social Medicine, Occupational and Environmental Dermatology, University Hospital, Ruprecht-Karls University, Heidelberg, 2Department of Nephrology, DKD Helios Clinic, Wiesbaden, 3Fresenius Medical Care Deutschland GmbH, Bad Homburg, and 4Department of Nephrology, University Hospital, Dusseldorf, Germany
Chronic itch is a common symptom in haemodialysis (HD) patients, which is often underestimated. The aim of this cross-sectional study was to investigate the prevalence and factors associated with chronic itch in HD patients. A total of 860 HD patients from a randomly selected cluster-sample of patients attending dialysis units in Germany were included. The patients’ mean ± SD age was 67.2 ± 13.5 years, 57.2% were male. The point prevalence of chronic itch was 25.2% (95% CI 22.4–28.1), 12-month prevalence was 27.2% (95% CI 24.1–30.3) and lifetime prevalence was 35.2% (95% CI 31.9–38.3). Chronic itch was significantly less prevalent in patients with secondary glomerulonephritis as primary renal disease. A history of dry skin, eczema, and age < 70 years were significantly associated with chronic itch. General health status and quality of life were significantly more impaired in subjects with chronic itch. This is the first representative cross-sectional study using a precise definition of chronic itch and using different prevalence estimates of chronic itch in HD patients. It demonstrates that chronic itch is a long-lasting burden significantly impairing patients’ health. Key words: epidemiology; haemodialysis; HRQOL; itch; itch prevalence; uremic pruritus; uremic itch; epidemiology of itch.
Accepted Mar 2, 2015; Epub ahead of print Mar 5, 2015
Acta Derm Venereol 2015; XX: XX–XX.
Melanie Weiss and Elke Weisshaar, Department of Clinical Social Medicine, Occupational and Environmental Dermatology, University Hospital Heidelberg, Thibautstrasse 3, DE-69115 Heidelberg, Germany. E-mail: Elke.Weisshaar@med.uni-heidelberg.de
Chronic itch (CI), defined as itch lasting for more than 6 weeks, is the most frequent skin symptom caused by multiple dermatoses, various systemic diseases, as well as drugs, neurogenic and psychiatric diseases (1, 2). In end-stage renal disease (ESRD) CI may be termed uraemic itch or “chronic kidney disease-associated itch” (3). Many patients with ESRD depending on haemodialysis (HD) treatment are affected by CI (3), with worldwide prevalence ranging from 10% to 85% (1, 4). This wide range can be explained mainly by the use of different measures; whereas some studies measure lifetime prevalence explaining higher numbers, others take current symptoms into account (1). The prevalence and burden of CI in HD patients are often underestimated (5), although studies point to significant reductions in health-related quality of life (HRQOL) and higher mortality risk caused by CI (4, 6–8). Since the number of patients on HD will increase continuously (as a result of the demographic situation and increased survival of ESRD patients), CI in dialysis is a field of particular interest.
The aim of this study was to investigate the prevalence of CI in HD patients, considering different prevalence estimates in a cross-sectional study using a representative, randomly selected cluster-sample representative for all dialysis units in Germany and using standardized measures (1, 9–14). The primary study outcomes were point, 12-month and lifetime prevalence of CI according to a validated study instrument (9, 10, 15). Secondary outcome measures were characteristics of CI (e.g. severity, localization) (9, 10, 13, 15), the aetiology of ESRD, generic HRQOL (SF-12) (12, 16), itch-related QOL (ItchyQol) (11), and anxiety and depression (Hospital and Anxiety Depression Scale (HADS) (14, 17).
Materials and Methods
The German Epidemiological Hemodialysis Itch Study (GEHIS) was established in 2012 as a prospective observational prevalence study based on data from patients’ questionnaires and patients’ data provided by the dialysis units. Data collection took place between May and September 2013. Inclusion criteria were: those patients who were diagnosed with ESRD and undergoing HD treatment; minimum age 18 years; sufficient knowledge of the German language; ability to read and write; and no major current cognitive impairment. Ethical approval was obtained from the University of Heidelberg, Germany (no. S-648/2012). The study was established according to the guidelines of good clinical practice and conducted in full accordance with the protocols of the World Medical Association’s Declaration of Helsinki. Patients gave their written informed consent. All results were reported in line with the “Strengthening the Reporting of Observational Studies in Epidemiology (STROBE)” recommendations (18).
Calculation of power and sample size was conducted to enable an epidemiological prevalence study (19). The minimum sample size needed to detect a prevalence of 20% with 95% confidence and 5% precision is 425 patients. To take into consideration the design effect of cluster sampling, this value was multiplied by 2 (19), resulting in a minimum sample size of 850 patients. Further details of the database and the sampling are given in Appendix S11. All selected dialysis units were sent an information letter by post explaining the study’s background/details and asking for permission to serve as a study centre.
Study instruments
Prior to this study the needs of HD patients with CI were identified by conducting qualitative interviews (20). Information relying on these interviews served to select patient-oriented outcome measures. The feasibility and reliability of the study instruments used were proofed within a pre-study investigating 20 HD patients in a unit that was excluded from the GEHIS study’s data pool.
All patients received a questionnaire about sociodemographic data (sex, age, occupational status, education, marital status, ethnic origin). Itching was assessed with the patient questionnaire about current CI, CI within the last 12 months, and CI ever in life (10). Additional items assessed atopic diseases (eczema, rhinitis, asthma), dry skin and quality of sleep by self-report. A norm-based 12-item questionnaire (SF-12 questionnaire (16)) measured perceived HRQOL that describes the degree of general physical health status and mental health distress. Summary scores are obtained for each of the physical and mental domains by summing across all 12 items for each, whilst lower scores indicate lower levels of health. Anxiety and depression, measured by 14 items on a scale ranging from 0 to 3 for the subscales anxiety and depression (HADS questionnaire) (17), were assessed in all HD patients, valuing between 0 and 21 for both subscales, also indicating higher score levels for lower level of health. Dialysis parameters (e.g. duration of HD) were provided by the HD unit for all patients and double-checked by the investigator.
If a patient reported current CI or CI within the past 12 months, a second itch-specific questionnaire assessed duration, frequency, characteristics, localization, course and deterioration of CI (13, 21), as well as suspected causes of CI. To assess mean, current and strongest subjective severity of experienced CI, a visual analogue scale (VAS) ranging from 0 (no itch) to 10 (worst imaginable itch) was used (22, 23). Itch-related QOL was measured using the ItchyQoL questionnaire, assessing 22 items with a Likert scale ranging from 1 (never) to 5 (always) in the subscales symptoms, functionality, emotions and self-efficacy (11).
Procedure
Patients were approached by a research fellow or a dermatologist of the Department of Clinical Social Medicine, University Heidelberg, at the beginning of the dialysis session. A patient’s information and a consent sheet were given to those expressing intention to participate. After signing, the questionnaires were distributed and completed immediately by the patient. In order to include 850 HD patients across a total of 25 dialysis units, patient recruiting ceased in each dialysis unit once a sufficient number (34 patients) had been included.
Statistical analyses
A Microsoft Access 2003 database was used for data entry. Data entry was conducted twice by 2 independent people. All observed random or potentially systematic inconsistencies in resulting data were solved to maximize data quality. Statistical analyses were performed using SPSS (version 20) for Windows. Nominal and ordinal data were analysed by computing absolute (n) and relative frequencies (%), respectively, and 95% confidence intervals were computed. First Fisher’s exact t-test and χ2 statistics were used to identify variables that were significantly associated with CI and severity of CI in univariate analysis. Differences between continuous variables (e.g. age) were analysed using independent t-tests/analysis of variance (ANOVA). Associations between severity and duration of CI, HRQOL and emotional well-being are reported using Pearson’s correlation coefficients. p-values below 0.05 were considered significant.
Results
Study sample
Of the 25 dialysis units contacted, 10 declined the invitation to participate (primary response rate 60%). Three dialysis units were hospital-based and were excluded from the sample. Thus, 13 additional dialysis units had to be randomly selected according to the respective geographical quadrants (Fig. S11).
Table I. Sociodemographic and medical parameters of the study population (n = 860)
|
Female |
Male |
|
|
Total, n (%) |
368 (42.8) |
492 (57.2) |
|
Age, years, mean (SD) |
68.2 (13.9) |
66.5 (13.7) |
|
Occupational status, % (n) |
||
|
Working |
5.7 (21) |
12.2 (60) |
|
Retired |
71.0 (260) |
69.0 (339) |
|
Other |
23.3 (85) |
18.7 (92) |
|
Schooling, % (n) |
||
|
Elementary |
91.0 (323) |
79.2 (373) |
|
Advanced |
9.0 (32) |
20.8 (98) |
|
Marital status, % (n) |
||
|
Married/in partnership |
43.6 (161) |
66.2 (323) |
|
Widowed |
36.6 (134) |
10.7 (52) |
|
Divorced |
7.9 (29) |
7.8 (38) |
|
Single |
10.9 (40) |
14.6 (71) |
|
Origin, % (n) |
||
|
Germany |
91.2 (331) |
90.3 (438) |
|
Other |
8.8 (32) |
9.7 (47) |
|
Dialysis characteristics |
||
|
Time since dialysis treatment started, months, mean ± SD (n) |
58.3 ± 54.9 (333) |
57.4 ± 57.2 (450) |
|
Duration of weekly dialysis sessions, h, mean ± SD (n) |
12.8 ± 1.1 (354) |
13.0 ± 1.4 (481) |
|
Efficacy of HD treatment, Kt/V mean ± SD (n) |
1.6 ± 0.4 (317) |
1.4 ± 0.3 (443) |
SD: standard deviation; HD: haemodialysis; Kt/V: urea clearance in relation to urea distribution volume.
A total of 860 HD patients were included in the study (Table I). The patients’ ages ranged from 21 to 99 years (mean age 67.2 years, standard deviation (SD) 13.4 years). Of all HD patients 2.6% (n = 21) reported having atopic eczema (also called eczema), 7.0% (n = 58) reported having allergic asthma, 8.4% (n = 69) reported having hay fever, and 67.1% (n = 552) reported having dry skin.
Prevalence of chronic itch
Of the HD patients 25.2% were currently affected by CI, 27.2% reported having CI within the past 12 months, and 35.2% reported having had CI at least once in their life (Tables II and III). Throughout all prevalence items (point, 12-month and lifetime) men were more likely to have CI, e.g. point prevalence was 23.9% in female vs. 26.2% in men. However, sex was not significantly associated with CI in all prevalence estimates. The mean ± SD age of HD patients with current CI (point prevalence) was 64.3 ± 13.8 years and of non-affected HD patients 68.2 ± 13.2 years. There was a significant association between the prevalence of CI (point prevalence) and age. Patients aged < 70 years were significantly more affected by CI than those ≥ 70 years (Fig. 1). No significant differences in prevalence estimates emerged in relation to ethnic origin, schooling, and patients’ marital status. However, patients in employment were significantly more likely to have experienced CI ever in life, whilst current occupational status did not correlate significantly with the other prevalence estimates. The 12-month prevalence, lifetime prevalence and point prevalence of CI were significantly higher in HD patients with self-reported eczema, hay fever and dry skin. All prevalence items were significantly higher in HD patients with a history of eczema compared with those without eczema.
Table II. Prevalence of chronic itch according to the aetiology of end-stage renal disease (ESRD)
|
Aetiology of ESRD |
Frequency of disease within total sample % (n) |
Chronic itch (n=196) |
||
|
Lifetime prevalence % (n) |
12-month prevalence % (n) |
Point prevalence % (n) |
||
|
Primary chronic glomerulonephritis |
13.5 (115) |
45.0 (54)a |
35.0 (42)a |
32.5 (39)a |
|
Pyelonephritis |
3.1 (27) |
30.0 (9) |
16.7 (5) |
16.7 (5) |
|
Hypertensive nephritis |
14.8 (127) |
31.6 (43) |
27.2 (37) |
25.7 (35) |
|
Secondary glomerulonephritis |
26.5 (228) |
27.8 (64)b |
20.9 (48)b |
19.1 (44)b |
|
Hereditary renal disease |
11.7 (101) |
41.2 (42) |
34.3 (35) |
35.3 (36)a |
|
Unknown aetiology |
18.7 (161) |
41.7 (50) |
31.7 (38) |
28.3 (34) |
|
Other |
11.6 (100) |
37.7 (40) |
29.2 (31) |
24.5 (26) |
aSignificantly higher than the mean prevalence (n = 860) at p < 0.05. bSignificantly lower than the mean at p < 0.05.
Table III. Prevalence of chronic itch in the study sample and according to sex (n = 860)
|
Prevalence estimates |
Total |
Female |
Male |
|||||
|
% (n) |
95% CI |
% (n) |
95% CI |
% (n) |
95% CI |
|||
|
Point prevalence |
25.2 (217) |
22.3–28.1 |
23.9 (88) |
19.3–28.5 |
26.2 (129) |
22.6–30.0 |
||
|
12-month prevalence |
27.2 (234) |
24.2–30.5 |
25.8 (95) |
21.5–30.6 |
28.3 (139) |
24.6–32.2 |
||
|
Lifetime prevalence |
35.2 (303) |
32.0–38.1 |
34.5 (127) |
29.4–39.2 |
35.8 (176) |
31.5–40.2 |
||
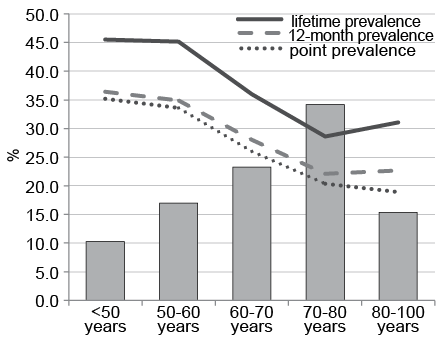
Regarding the aetiology of ESRD, there were no significant differences in prevalence estimates in pyelonephritis, hypertensive nephritis, unknown and other aetiologies (Table II). Patients with primary chronic glomerulonephritis were significantly more likely to have experienced CI (point, 12-month and lifetime prevalences); however, after adjusting for age and time since HD treatment started, the aetiology was no longer significantly associated with CI. Chronic itch was significantly less prevalent (all prevalence estimates) in patients with secondary glomerulonephritis as primary renal disease.
Characteristics of chronic itch
Important characteristics of CI are shown in Table IV and Fig. 2. Of the patients studied, 57.1% suggested that dialysis treatment caused their CI (n = 109). Of these, 40.4% reported that the CI was worst directly after HD (n = 44), 33.9% during HD (n = 37), and 24.8% did not report any relationship (n = 27). Mean ± SD severity of CI ((weakest + worse severity)/2)) of CI of all patients was 4.1 ± 1.7, worst severity within the last 6 weeks was 6.5 ± 2.5. The severity at the time of investigation was 4.2 ± 2.6. In female patients the severity of weakest and mean CI was significantly higher compared with men (weakest CI: 2.1 vs. 1.4; mean CI: 4.6 vs. 3.8). In patients with CI affecting the arm(s), leg(s) and hand(s), CI was significantly more severe when asking about severity of the weakest itch. Localization on the arm(s) and leg(s) were characterized by significantly worse severity of mean CI. The highest CI scores were significantly more frequent in patients with itching of the legs. CI was reported to affect the shunt arm by 27.6% of patients (n = 54). In these, current CI score was significantly lower compared with patients without CI on their shunt arm (4.4 vs. 3.4).
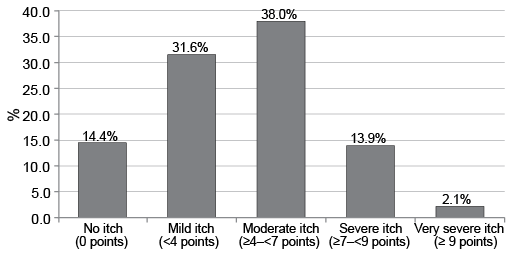
Fig. 2. Distribution of subjective itch severity at the time of investigation according to visual analogue scale (VAS) (according to ref. 22).
Table IV. Clinical characteristics of chronic itch (n = 196)
|
Duration of chronic itch (n = 187) |
Relative frequency (%) (n) |
|
6 weeks–6 months |
11.9 (22) |
|
6–12 months |
15.1 (28) |
|
1–10 years |
61.1 (113) |
|
> 10 years |
11.9 (22) |
|
Occurrence of chronic itch (n = 194) |
Relative frequency (%) (n) |
|
Daily |
63.4 (123) |
|
Weekly |
23.7 (46) |
|
Monthly |
60.4 (13) |
|
Less frequent |
6.2 (12) |
|
Severity of chronic itch (VAS) (n = 158) |
Mean (SD) |
|
Mean severity of itch |
4.1 (1.7) |
|
Worst itch |
6.5 (2.5) |
|
Current itch |
4.2 (2.6) |
|
Most frequent localizations of chronic itch |
Relative frequency (%) (n) |
|
Legs |
54.6 (107) |
|
Back |
52.0 (102) |
|
Scalp |
43.9 (86) |
SD: standard deviation; VAS: visual analogue scale.
Dialysis characteristics
The mean ± SD time on HD was 58 months (4.8 years) ± 56.2 months, min–max: 1–463 months (38.6 years)) (Table I). There was a significant correlation between time on HD and prevalence estimates. In patients with current and/or CI within the last 12 months, mean time on HD was 68.9 ± 69.4 months. Mean time on HD was 50.8 ± 45.5 months in HD patients without CI. The mean duration of weekly dialysis sessions was 12.9 h (SD 1.3). When grouping into 2 categories (< 12 and > 12 h/week), no significant difference was identified; however, lifetime prevalence was significantly associated with the duration of weekly dialysis sessions (31.7% in < 12 h, 37.7% in > 12 h). The mean ± SD dialysis efficacy, measured by Kt/V, was 1.5 ± 0.3 (min 0.8, max 3.1) in patients with CI and 1.5 ± 0.4 (minimum 0.5, maximum 3.1) in patients not affected by CI (Table I).
Emotional well-being, sleep and health-related quality of life
Associations between CI and all QOL measures were analysed focusing on the point prevalence of CI as these instruments investigate the last 1 or 4 week(s) in a patient’s life (Table V). HADS of all HD patients did not show a significant correlation between the occurrence of CI and the HADS in both subscales. However, the mean severity of CI was significantly associated with the HADS’s subscale anxiety, meaning a higher level of impairment concerning anxiety in HD patients with CI. There was no significant association with the HADS’s subscale depression.
Analysis of the SF-12 of all HD patients detected significantly worse values for both the SF-12 physical and mental component summary in HD patients affected by CI in relation to non-affected HD patients (Table V).
The mean ± SD value of ItchyQol in HD patients with CI was 2.0 ± 0.7). There was no significant difference according to sex. CI patients with worse itch showed significantly higher impairment. The strongest associations were found between the ItchyQoL in total and the mean severity of CI (p = 0.50) and worst itching (p = 0.48).
More than half of the HD patients with CI reported difficulties with falling asleep more than once a week. Of the patients studied, 32.7% complained about the quality of sleep, in those with CI this was 54.5%. Impaired quality of sleep was significantly associated with CI, but not with its severity (Table V).
Table V. Emotional well-being, health-related quality of life (HRQOL) and sleep characteristics in haemodialysis (HD) patients (n=860) with and without chronic itch (CI)
|
Point prevalence of CI |
|||
|
Yes |
No |
Total |
|
|
HADS (subscales ranging 0–21), mean (SD) |
|||
|
Subscale anxiety |
8.7 (2.3)* |
8.4 (2.4) |
8.5 (2.4) |
|
Subscale depression |
9.7 (2.3) |
9.5 (1.9) |
9.6 (2.0) |
|
SF-12 (subscales ranging 0–100), mean (SD) |
|||
|
Physical component summary |
34.6 (10.4)* |
37.1 (10.8) |
36.4 (10.8) |
|
Mental component summary |
52.0 (10.4)* |
54.3 (9.2) |
53.7 (9.6) |
|
Quality of sleep |
|||
|
Duration of sleep (h), mean (SD) |
6.1 (1.8)* |
6.5 (1.8) |
6.4 (1.8) |
|
Difficulties falling asleepa, % (n) |
54.5 (114)* |
38.5 (232) |
42.8 (335) |
|
Impaired quality of sleep, % (n) |
48.3 (102)* |
26.8 (164) |
32.7 (276) |
a At least once a week.
*Significantly different at p < 0.05.
HADS: Hospital Anxiety and Depression Scale; SD: standard deviation.
Discussion
The occurrence of itch as a bothersome symptom in HD patients has been reported for decades without identifying the causes or any treatments that substantially relieve the symptom (3, 24). A wide range of estimates of itch-prevalence has been found in several studies of HD patients (1, 4, 7, 8). Most studies of patients with CI do not include the occurrence of CI within the whole group of HD patients (1, 4, 7, 8). Previous studies have not differentiated between acute and chronic itch (> 6 weeks) (2). The ItchyQol, a previously validated measure of itch-related QOL was used for the first time in HD patients (11).
The major strength of our study is that, for the first time, different prevalence estimates were obtained in HD patients with CI. GEHIS demonstrates that approximately two-thirds of those affected by CI once in their life have CI continuously. This is in contrast to chronic skin diseases characterized by CI, e.g. atopic dermatitis (AD, also called eczema) (25). Our results show that CI in HD patients is chronic and lasts for many years, which may be explained by the lack of sufficient therapies (3, 24) and underestimation of the condition by nephrologists (5). Compared with prevalence estimates obtained in the general population (point prevalence 13.5%, lifetime prevalence 22%) (9), CI occurs considerably more frequently in HD patients. There was a trend for men to be more affected than women, although this was not significant. Other studies in the general or patient population have always showed women to be more affected by CI (1, 9, 10, 15, 26, 27, 28). Our findings may be explained by the fact that more men are on HD treatment (4). Recent reports have even concluded that men have a higher odds of moderate to extreme itch (4, 8). In contrast to previous studies (8, 10, 15, 26, 27) the current study found that younger HD patients were more frequently affected by CI.
For the first time, our study considered a more detailed analysis of aetiologies of ESRD (29). Those with secondary chronic glomerulonephritis were significantly less affected by CI. A previous investigation suggested increasing the dialysis dose to improve uraemic itch (30). We identified an association between the time since dialysis treatment started and the occurrence of CI, confirming the results of previous research (4, 31). The current study also showed an association of the duration of weekly dialysis sessions and lifetime prevalence of CI, which has not been investigated previously.
The results on the severity of CI are new and important, demonstrating that HD patients experience constant and moderate itch. This is in contrast to, for example, CI in patients with AD who usually rate very high scores. This may partially explain why CI is underestimated in HD patients. Patients may omit to mention this symptom regarding its occurrence as part of dialysis treatment. Legs, back and scalp were the body areas most frequently affected by CI, which partly confirms previous research (13, 29).
A history of eczema and self-reported dry skin were significantly associated with all CI-prevalence items, demonstrating that HD patients with these characteristics may be prone to develop CI. This may also partially explain why CI occurred more frequently in younger HD patients. Dry skin and eczema were also identified as determinants of prevalent CI in the general population (15).
Previous research showed that the impairment caused by CI is comparable to that of pain (32) and that CI substantially impairs HRQOL in patients depending on HD treatment (7, 21, 31), but no study has investigated different measures of HRQOL including general health and sleep, different prevalence estimates and various clinical characteristics of CI within a single cohort at once. All measures of QOL and general health used in the GEHIS study showed significant impairment of HD patients with CI, but there was no association with the severity of CI.
As we also identified higher anxiety scores in HD patients with CI, we could confirm findings of CI in the general population (15). This suggests that the psychosocial impairment is caused by CI and not by HD treatment. ItchyQol showed a significant impairment in HD patients affected by CI. An association of reduced QOL and itch severity was also identified. The Dialysis Outcomes and Practice Patterns Study (DOPPS) (33) reported CI to markedly affect a HD patient’s life, as CI was strongly associated with impaired QOL, sleep disturbances, depressive symptoms and higher mortality (4). However, results from the DOPPS study additionally showed poor QOL and a high prevalence of depression in HD patients in general (34). These symptoms are often underdiagnosed, undertreated and associated with adverse clinical outcomes (35) and coping skills (36). Data for all, itching and non-itching HD patients of GEHIS, also showed borderline results regarding mental status (HADS>8). Special attention should be paid to sleep quality, as DOPPS showed the itch/mortality relationship to be substantially attributed to poor sleep quality (4). Our results show that the quality of sleep is substantially associated with the prevalence of CI but not with its severity. The results of GEHIS clearly demonstrate that those with CI are significantly more impaired in their HRQOL and their general health status compared with HD patients without CI.
Some limitations of the study need to be discussed. In all observational surveys, systematic biases are possible, but using cluster sampling according to the principle of random sampling and respecting the geographical distribution of German HD units minimizes the likelihood of selection bias. The impact of selective, voluntary participation may influence the findings of the present study; for example, individuals affected by the symptom may have been more likely to participate, whilst patients with health or educational disadvantages as well as tired/depressive/anxious patients may be under-represented. Due to the cross-sectional study design, one has to be cautious about the relationships of CI with medical/lifestyle/psychosocial factors, as we were not able to disentangle potential directions of causality. One may argue that all findings presented in this paper are only representative for Germany. However, due to the comparability concerning standards in HD quality in developed countries our results can be transferred to Western countries.
In conclusion, CI in HD patients must therefore be taken seriously, not only because of its frequency and its resulting burden, but also due to the 23% higher mortality risk among HD patients with CI (8).
AcknowledgEments
We thank all the participating dialysis units and especially all HD patients for participating in this study. We thank Kinan Hayani for his help in obtaining and entering the data.
Conflicts of interest. The study was financially supported by Fresenius Medical Care Germany GmbH. The sponsor, represented by UT, had no influence on the design, conduct, collection and interpretation of data of the study. JP-D and TM, nephrologists, serve as advisors for Fresenius Medical Care Germany GmbH and received payments for consultations and lectures from Fresenius Medical Care Germany GmbH. MW, research associate at the University Hospital Heidelberg, was financed by Fresenius Medical Care Germany GmbH. EW declares no conflict of interest. There was no personal involvement that poses a potential duality of interest by conducting the study and analysing its data.

1http://www.medicaljournals.se/acta/content/?doi=10.2340/00015555-2087
References
