OBJECTIVE: To predict the discharge destination of rehabilitation patients using signal detection analysis.
DESIGN: Cross-sectional and follow-up studies.
Subject: The subjects were 324 patients discharged from a hospital in Fukuoka, Japan, between April 2005 and March 2006 and 313 patients discharged from the same hospital between 1 April and 31 October 2006.
METHODS: The discharge destinations of the 324 patients were predicted using signal detection analysis. As a validation study, 7 variables identified in the first analysis were used to categorize 313 patients, organized retrospectively into 8 groups, and to calculate the home discharge rate in each group.
RESULTS: A patient’s activities with respect to daily living, key person preference, dementia, age, route taken to hospitalization, residence before hospitalization, and gender were significant predictors of his or her discharge destination. Signal detection analysis established 8 subgroups, with 17.9–99.1% of the patients returning home after discharge. As a validation study, the actual and expected rates in the 8 subgroups were compared, and no significant difference was observed between the rates in any subgroup.
CONCLUSION: Signal detection analysis is a useful technique for predicting the discharge destination of rehabilitation patients.
Key words: discharge destination, prediction, rehabilitation, signal detection analysis.
J Rehabil Med 2008; 40: 261–268
Correspondence address: Akihito Hagihara, Department of Health Services Management and Policy, Graduate School of Medicine, Kyusyu University, 3-1-1 Maidashi, Higashi-ku, Fukuoka, 812-8582, Japan. E-mail: hagi-ca@hsmp.med.kyushu-u.ac.jp
Submitted May 7, 2007; accepted October 29, 2007
Introduction
The ratio of national medical expenditure to gross national income in Japan is increasing due to an ageing population and the rising cost of medical advances (1). Thus, the control of medical expenses is an important issue currently facing Japan. In the field of rehabilitation medicine it is important to determine patients’ discharge destinations at an early stage of hospital admission in order to use medical resources efficiently. In the clinical setting, it is important to examine the amount of rehabilitation necessary, while considering such factors as the anticipated effectiveness of rehabilitation (i.e. the expected degree of recovery), the patient’s predicted destination after discharge, their preferred destination after discharge, their quality of life (QoL), and activities of daily living (ADL) status at the start of rehabilitation soon after hospital admission. In addition, for patients who lived at home prior to hospitalization and who will be moving into a care facility, it is necessary to apply for nursing insurance in order to receive welfare payments and admission to a care facility. Although there are regional differences in Japan, in many cases there is a waiting period for access to care facilities, and patients are not guaranteed a place immediately after their rehabilitation is complete at the hospital. As a result, some patients remain hospitalized, not for rehabilitation purposes, but for non-medical reasons, such as to wait for an opening at a nursing facility. Such situations represent an inappropriate use of rehabilitation resources. It is thus desirable to be able to predict a patient’s discharge destination at the start of rehabilitation intervention, in order that the rehabilitation programme functions smoothly and effectively. If a change in residence to a nursing facility is predicted, it is necessary to apply for nursing insurance, which requires 28 days for application processing, and to apply for admittance to a care facility as early as possible. If the predicted discharge is to the patient’s home, necessary home modifications can be completed prior to the patient’s discharge from hospital. These types of actions would contribute to a smoother hospital discharge.
To date, many predictive studies have examined 3 objective variables: ADL status (2–8), the number of days hospitalized (5, 7) and the discharge destination (3, 4, 8–11). Those studies that considered a patient's discharge destination indicated that patients are more likely to return home if they are young and have maintained their ADL (3, 6, 9–11). Common statistical methods used in making these kinds of predictions include the χ2 test with stratification and multivariate analysis. These statistical methods are effective when a model includes the interaction of 2 variables. However, when a model includes the interaction of 3 or more variables, there are typically problems with multi-colinearity, and the interpretation of higher-order interactions is very complex. To address this problem, the signal detection analysis (SDA) method of Kraemer (12–14) has been advocated.
To verify the validity of this new method in predicting the discharge destination of patients in rehabilitation units, relevant data were collected from patients who lived at home prior to hospitalization. Various factors affect the discharge destination of patients in rehabilitation units, including personal factors (e.g. age, gender, family structure, residential state, residential region, social support, income) and physical factors (e.g. underlying diseases, ADL status, the presence of dementia (4, 9, 10, 15–18)). In addition, it is probably not a single factor, but a combination of factors that influence a patient’s discharge destination. With 27 potential variables, there are in excess of 134 million possible combinations. Obviously, logistic regression analysis cannot adequately deal with every possible combination. SDA recursively reveals the strongest interaction of factors among groups, based upon the largest χ2 statistic and significance probability (p < 0.05), and in the absence of that factor, the remaining factors are again analysed recursively. Because SDA examines all combinations of variables, it prevents the multi-colinearity problems that often plague multivariate regression analyses when so many explanatory variables are involved (12–14). In addition, because SDA can analyse the joint and interactive effects of factors and identify subgroups with common characteristics, it is easy to interpret the findings, and intervention approaches can thus be tailored to each subgroup’s characteristics (19–21). From the perspective of healthcare policy, the results of this study may contribute to the effective use of rehabilitation programmes.
Methods
Subjects
The subjects included 2 groups of patients who underwent rehabilitation at a hospital in Fukuoka City, Japan. The first group of 691 patients underwent rehabilitation between April 2005 and March 2006. The exclusion criteria were: death during hospitalization (n = 38); patients whose rehabilitation was discontinued due to deterioration of their condition (n = 65); those who were admitted to the hospital from care facilities (n = 99); those for whom there were missing data (n = 10); and those aged 85 years or older (n = 155). The remaining 324 patients served as the predictive model group. The aim was to obtain data from those patients receiving rehabilitation at the hospital in order to predict their destinations after discharge. Thus, patients who were admitted to the hospital from nursing facilities were excluded because they were predicted to return to their facilities following discharge. Patients who were very elderly (i.e. 85 years or older) were also excluded. Because the nuclear family is common in Japanese society, it is often difficult for the primary caregiver (i.e. the wife or husband) to take care of her/his elderly partner due to lack of family support at home. A patient’s advanced age may significantly affect the discharge destination. In addition, ≥ 85 years of age is defined as the oldest old (22).
To verify the accuracy of the prediction methodology, a second group of patients was also considered: 313 patients who were discharged from the same hospital between 1 April 2006 and 31 October 2006. Relevant data (i.e. information on the statistically significant variables identified using the first predictive model group) on each of the 313 patients were accumulated during the study period.
The hospital is a mixed-care medical institution with a general ward, a ward for long-term care, and a convalescence/rehabilitation ward. The professional staff includes 25 physical therapists, 21 occupational therapists, 5 speech therapists and 6 social workers.
Study variables
There were 27 independent variables and 1 dependent variable (Table I). The following information was ascertained prior to and during each patient’s hospital stay: personal attributes such as the patient’s gender and age, residential address and physical activity level before hospitalization. The physical therapists (PTs) and occupational therapists (OTs) who were assigned to take care of a patient ascertained each patient’s physical status and functional independence level at the beginning of the rehabilitation programme. Functional independence was assessed using the Functional Independence Measure (FIMTM) (23). The patients were divided into 3 groups based on the following diagnoses: cerebrovascular accidents, orthopaedic disorders, and disuse syndrome. Disuse syndrome is a type of hypo-activity, with musculoskeletal inactivity, resulting in the depletion of body systems, typically associated with mechanical or prescribed immobilization, severe pain, or an altered level of consciousness. The patients were also divided into 3 groups based on the key person or primary caregiver in their lives: children, spouse and other. The presence of dementia was also recorded based on the DSM-IV-TR (24). This determination was performed separately by the rehabilitation staff, including the PTs, OTs and the ward nurses. Patients were diagnosed as having dementia if the therapists and nurses both agreed that the patient showed signs of dementia or if no consensus was reached. Patients were determined not to have dementia if the therapists and nurses both agreed that the patients did not show signs of dementia. Several physical abilities were also assessed, including standing up, sitting, rolling over in bed, maintaining a position, and walking. If a patient’s ability to perform a particular action was unclear, the patient was deemed to be “unable” to perform the movement. To assess these abilities accurately, the assessment was performed twice by different evaluators. Patients were also determined to “have ataxia” if ataxia was observed or suspected from a nose–finger–nose test (25) and from observations of their ability to assume a standing position, maintain a sitting position, and walk. Patients were determined to “have motor paralysis” if they were in Brunnstrom stages I–V (26). Patients were determined to “have a sensory disturbance” if a disturbance involving superficial or deep sensation was observed or suspected (25). Finally, patients were determined “not to have a decline in muscle strength” if their muscle test score, determined according to the method of Daniels & Worthingham (27), was 4 or higher. The FIMTM was used for the ADL evaluation.
Analysis
In the first half of the study, using SDA to focus on the parameters of “sensitivity” and “specificity,” we used 27 variables (Table I) and identified the factors affecting the discharge destination of the study subjects. The overall approach was as follows. First, “sensitivity” was defined as “the percentage of patients who had specific factors among the patients who returned home” (12, 13). “Specificity” was defined as “the percentage of patients who did not have specific factors among patients who did not return home” (i.e. those placed in facilities). Kraemer proposed the following index, which uses the definitions of sensitivity and specificity, and demonstrated that the index follows a χ2 distribution with 1 degree of freedom (12, 13):
χ2 = n*[(SE–Q)/Q’]*[(SP–Q’)/Q] (df=1)
where n = total number, SE = sensitivity, SP = specificity, Q = (patients who returned home)/(total number) (n), Q’ = (1–Q).
This signal detection parameter is equivalent to the χ2 statistic (df = 1), which means that the subjects are categorized into a 2 × 2 table consisting of dependent and independent variables. When the parameter reports the largest χ2 value (df = 1), the variable and its cut-off point in the equation is the best predictor of discharge destination. Because the χ2 value is largest, this variable and its cut-off point divide the population into 2 subgroups that are mutually exclusive.
Thus, the dependent variable (discharge destination) and the independent variables in Table I were entered into the model, along with minimal and maximal values and intermediate cut-off points. The variables had 1–107 cut-off points (Table I). For example, “sex” (male, female) and “employment status” (employed, unemployed) had only 1 cut-off point. “Key person” had 3 cut-off points: (children, spouse or other), (spouse, children or other), (other, children or spouse). “Types of disorder” also had 3 cut-off points: (cerebrovascular disease, orthopaedic disorders or disuse syndrome), (orthopaedic disorders, cerebrovascular disease or disuse syndrome), (disuse syndrome, cerebrovascular disease or orthopaedic disorders). “FIMTM score” had 107 cut-off points (18, 19, …., 126).
| Table I. Subject profiles. Values are mean (%) unless otherwise shown |
| Subjects in the model development study (n = 324) |
| Patient’s personal factors |
| 1. Gender |
| Male | 123 (38.0) |
| Female | 201 (62.0) |
| 2. Age, years (mean (SD)) |
| Total | 69.26 (13.54) |
| Male | 65.70 (14.54) |
| Female | 71.44 (12.43) |
| 3. Residential address |
| Urban | 256 (79.0) |
| Suburban/rural | 68 (21.0) |
| 4. Route taken for hospitalization |
| Another hospital | 148 (45.7) |
| Outpatient clinic | 176 (54.3) |
| 5. Residence prior to hospitalization |
| One’s own property | 247 (76.2) |
| Rental housing | 77 (23.8) |
| 6. Employment status |
| Employed | 61 (18.8) |
| Unemployed | 263 (81.2) |
| 7. Key person |
| Children | 145 (44.8) |
| Spouse | 136 (42.0) |
| Other | 43 (13.2) |
| 8. Status of patient and key person |
| Living together | 221 (68.2) |
| Not living together | 103 (31.8) |
| 9. Patient’s preferred residence after hospitalization |
| Home | 256 (79.0) |
| Other than home | 68 (21.0) |
| 10. Key person’s preferred residence after hospitalization |
| Home | 292 (90.1) |
| Other than home | 32 (9.9) |
| Patient’s disease state |
| 11. Types of disorder |
| Cerebrovascular disease | 58 (17.9) |
| Orthopaedic disorders | 153 (47.2) |
| Disuse syndrome | 113 (34.9) |
| 12. Dementia |
| Yes | 49 (15.1) |
| No | 275 (84.9) |
| Physical capacity prior to hospitalization |
| 13. Standing up |
| Yes | 309 (95.4) |
| No | 15 (4.6) |
| 14. Sitting |
| Yes | 320 (98.8) |
| No | 4 (1.2) |
| 15. Rolling over |
| Yes | 316 (97.5) |
| No | 8 (2.5) |
| 16. Maintaining a standing position |
| Yes | 312 (96.3) |
| No | 12 (3.7) |
| 17. Walking |
| Yes | 306 (94.4) |
| No | 18 (5.6) |
| Physical capacity at the start of rehabilitation |
| 18. Standing up |
| Yes | 265 (81.8) |
| No | 59 (18.2) |
| 19. Sitting |
| Yes | 284 (87.7) |
| No | 40 (12.3) |
| 20. Rolling over |
| Yes | 283 (87.3) |
| No | 41 (12.7) |
| 21. Maintaining a standing position |
| Yes | 256 (79.0) |
| No | 68 (21.0) |
| 22. Walking |
| Yes | 177 (54.6) |
| No | 147 (45.4) |
| 23. Ataxia |
| No | 312 (96.2) |
| Yes | 12 (3.7) |
| 24. Paralysis |
| No | 267 (82.4) |
| Yes | 57 (17.6) |
| 25. Sensory disturbance |
| No | 240 (74.1) |
| Yes | 84 (25.9) |
| 26. Muscle strength |
| No | 92 (28.4) |
| Yes | 232 (71.6) |
| 27. FIMTM score (mean) (SD) | 100.7 (29.00) |
| range | 18–126 |
| Dependent variable |
| 28. Discharge destination |
| Home | 267 (82.4) |
| Other than home | 57 (17.6) |
| Subjects in the cross-validation study (n = 313) |
| 1. Discharge destination (home) | 232 (74.1) |
| 2. Age (mean) (SD) years | 70.27 (12.54) |
| 3. FIMTM score (mean) (SD) | 94.89 (27.16) |
| range | 18–126 |
| 4. Gender |
| Male | 108 (34.5) |
| Female | 205 (65.5) |
| 5. Key person’s preferred residence after hospitalization |
| Home | 243 (77.6) |
| Other than home | 70 (22.4) |
| 6. Dementia |
| No | 223 (71.2) |
| Yes | 90 (28.8) |
| 7. Route taken for hospitalization |
| Outpatient | 136 (43.5) |
| Another hospital | 177 (56.5) |
| 8. Age (years) |
| ≥ 80 | 74 (23.6) |
| ≤ 79 | 239 (76.4) |
| 9. FIM |
| ≤ 51 | 32 (10.2) |
| ≥ 52 | 281 (89.8) |
| 10. Residence prior to hospitalization |
| One’s own property | 195 (62.3) |
| Rental housing | 118 (37.7) |
| SD: standard deviation. |
With respect to the probability of returning home, based on the largest χ2 measure, the algorithm for the signal detection model checked every variable in Table I and the possible cut-off points to determine the optimally efficient variable and its cut-off point. After the signal detection algorithm selects the first optimally efficient variable along with its cut-off point, which is the largest χ2 value, the programme then begins to look for the next optimally efficient variable and its cut-off point, separately, in each of the newly divided subgroups. This procedure is repeated separately for all of the remaining variables until one of the following occurs: (i) no further predictors occur in a newly formed subgroup; (i) no more significant variables are detected at a level of p < 0.05; or (iii) the number of subjects in the newly divided group becomes too small (n ≤ 10) (14). In summary, based on the probability of returning home, we used this method and relevant variables listed in Table I to divide the study population into subgroups that were maximally discriminated from each other and mutually exclusive.
In the second half of the study, to verify the accuracy of the prediction method based on the SDA, 313 patients were categorized into the several subgroups on the basis of the significant variables identified in the first analysis. The actual and predicted rates of those who returned home after unit discharge were then compared in each subgroup (Tables I and II).
The subgroups created by this SDA were compared in terms of patients’ in-patient personal attributes and physical status. The distribution was tested using χ2 analysis or the Kruskal-Wallis test. Calculation software created by Kraemer (Stanford University School of Medicine, Stanford Preventative Medicine Laboratory) was used for the SDA, and the SPSS statistical package (ver. 11) was used for the other analyses.
Ethical review of the study
The study was approved by the ethics committee at the hospital on 8 March 2005.
Results
A summary of the information for the various subgroups is given in Table I. As indicated, more women (62.0%) than men were in the study group, and the mean subject age was 69.3 years. Concerning the route to hospitalization, over 40% were transferred from another hospital, whereas approximately 50% came from an outpatient clinic. The percentage of patients living at home prior to hospitalization was high, in excess of 70%. Most of the patients’ key people were their children (44.8%), followed by their spouse (42.0%), then others (13.2%). Slightly less than 70% of the patients lived with their key person. Approximately 80% of the patients stated a preference for their own home as their destination after discharge. Similarly, a large majority (90.1%) of the key people preferred that the patient be discharged to their own home. Regarding disease type, cerebrovascular accidents accounted for slightly less than 20% of the causative events, whereas orthopaedic disease accounted for approximately 50%, and other conditions, such as Parkinson’s disease, were present in approximately 35% of the patients. The mean FIMTM value, which represents the level of independence in daily activities, was 100.7. Finally, after discharge, more than 80% of the patients returned to their own homes, whereas slightly less than 20% were transferred to care facilities.
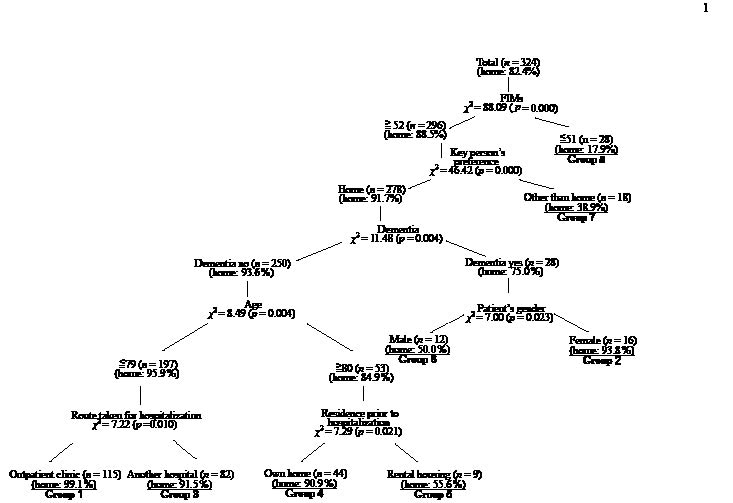
Fig. 1 shows those factors that were related to a patient’s discharge destination based on the SDA. The FIMTM scores at the start of rehabilitation intervention was most strongly related to a patient’s discharge destination (χ2 = 88.09, p < 0.001), and the patients were divided into the < 51 group (Group 8) or the ≥ 52 group based on their scores. Within the < 51 group (Group 8), 17.9% of the patients returned to their homes, whereas approximately 80% of the patients were discharged to care facilities. No further splits were found for this group when applying the 3 stopping rules specified above. In comparison, for the ≥ 52 group, the key person’s preferred discharge destination had the highest association with a patient’s actual discharge destination (χ2 = 46.42, p < 0.001), and the patients were divided into 2 groups according to the key person’s preference: the “home” group and the “other-than-home” group (Group 7).
Fig. 1. Factors associated with the discharge destination of rehabilitation patients (n = 324).
Using the 8 significant predictors identified by the SDA (Fig. 1), the 313 patients were categorized into 8 groups, and the expected rate of those who went to their home in each subgroup was compared with the actual rate. The actual and expected rates in the 8 subgroups were very close, and using the χ2 test no significant difference was observed between these 2 types of rates in any subgroup, implying that the prediction of the outcome of unit discharge by subgroup is very reliable (Table II).
| Table II. Actual and expected numbers of patients discharged to their homes during 1 April to 31 October 2006 (n = 313) |
| | Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 |
| Number | 67 | 26 | 86 | 23 | 16 | 15 | 48 | 32 |
| Age (mean (SD)) (years) | 63.22 (13.33) | 74.88 (10.80) | 63.84 (12.95) | 82.13 (1.14) | 81.88 (1.26) | 70.73 (10.22) | 75.46 (6.73) | 76.19 (6.77) |
| Gender (male) (%) | 29.9 | 0 | 47.7 | 34.8 | 12.5 | 100.0 | 25.0 | 31.3 |
| AD (home) (%) | 98.5 | 92.3 | 91.9 | 95.7 | 56.3 | 60.0 | 37.5 | 15.6 |
| ED (home) (%) | 99.1 | 93.8 | 91.5 | 90.9 | 55.6 | 50.0 | 38.9 | 17.9 |
| p | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
| AD: actual destination; ED: expected destination; SD: standard deviation. The rates of those who went to their home in AD and ED groups were compared using the χ2 test. n.s.: not significant. |
Table III summarizes the characteristics of the SDA-based subgroups. In terms of the “patient’s preference of discharge destination,” the groups with higher percentages of patients discharged to facilities (Groups 6 and 8) had more patients who preferred to be discharged to their own homes than the groups with lower percentages of patients discharged to care facilities (Groups 1, 3, and 4). In terms of disease, the groups with higher percentages of patients discharged to facilities (Groups 6 and 8) had more patients with cerebrovascular accidents than the groups with lower percentages of patients discharged to facilities (Groups 1–4).
| Table III. Profiles of subgroups divided by signal detection analysis. All values are given as percent whereas not otherwise is stated. |
| | Group 1 | Group 2 | Group 3 | Group 4 | Group 5 | Group 6 | Group 7 | Group 8 | p |
| (n = 115) | (n = 16) | (n = 82) | (n = 44) | (n = 9) | (n = 12) | (n = 18) | (n = 28) |
| Patient’s personal factors | | | | | | | | | |
| 1. Gender (male) | 40.9 | 0.0 | 40.2 | 31.8 | 0.0 | 100.0 | 27.8 | 42.9 | 0.000 |
| 2. Mean age (years) | 63.1 | 74.7 | 62.7 | 81.9 | 82.0 | 70.8 | 76.3 | 75.9 | 0.000 |
| 3. Residential address (urban) | 77.4 | 87.5 | 78.0 | 75.0 | 66.7 | 66.7 | 100.0 | 85.7 | n.s |
| 4. Route taken for hospitalization (another hospital) | 0.0 | 43.8 | 100.0 | 43.2 | 44.4 | 50.0 | 55.6 | 71.4 | 0.000 |
| 5. Residence prior to hospitalization (own home) | 81.7 | 87.5 | 70.7 | 100.0 | 0.0 | 75.0 | 38.9 | 75.0 | 0.000 |
| 6. Employment status (employed) | 28.7 | 6.3 | 26.8 | 4.5 | 0.0 | 16.7 | 0.0 | 3.6 | 0.000 |
| 7. Key person (children) | 29.6 | 62.5 | 86.6 | 36.6 | 72.7 | 1.7 | 77.8 | 50.0 | 0.000 |
| 8. Status of patient and key person (living together) | 75.7 | 56.3 | 72.0 | 72.7 | 44.4 | 50.0 | 27.8 | 67.9 | 0.002 |
| 9. Patient’s preferred residence after hospitalization (home) | 94.8 | 12.5 | 93.9 | 97.9 | 88.9 | 16.7 | 55.6 | 17.9 | 0.000 |
| 10. Key person’s preferred residence after hospitalization (home) | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 0.0 | 50.0 | 0.000 |
| Patients’ diseases | | | | | | | | | |
| 11. Type of disease (cerebrovascular disease) | 6.4 | 18.8 | 17.1 | 11.4 | 0.0 | 50.0 | 11.1 | 71.4 | 0.000 |
| 12. Dementia (yes) | 0.0 | 100.0 | 0.0 | 0.0 | 0.0 | 100.0 | 0.0 | 75.0 | 0.000 |
| Physical activity prior to hospitalization | | | | | | | | | |
| 13. Standing up (yes) | 96.5 | 100.0 | 100.0 | 97.7 | 100.0 | 100.0 | 94.4 | 67.9 | 0.000 |
| 14. Sitting (yes) | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 85.7 | 0.000 |
| 15. Rolling over (yes) | 99.1 | 100.0 | 98.8 | 95.5 | 100.0 | 100.0 | 100.0 | 85.7 | 0.005 |
| 16. Maintaining a standing position (yes) | 97.4 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 94.4 | 71.4 | 0.000 |
| 17. Walking (yes) | 97.4 | 100.0 | 98.8 | 95.5 | 100.0 | 91.7 | 94.4 | 64.3 | 0.000 |
| Physical activity at the start of rehabilitation | | | | | | | | | |
| 18. Standing up (yes) | 87.8 | 93.8 | 95.1 | 84.1 | 88.9 | 83.3 | 72.2 | 10.7 | 0.000 |
| 19. Sitting (yes) | 94.8 | 100.0 | 95.1 | 90.9 | 88.9 | 91.7 | 83.3 | 25.0 | 0.000 |
| 20. Rolling over (yes) | 94.8 | 100.0 | 96.3 | 88.6 | 88.9 | 91.7 | 83.3 | 21.4 | 0.000 |
| 21. Maintaining a standing position (yes) | 87.8 | 81.3 | 92.7 | 77.3 | 88.9 | 83.3 | 66.7 | 7.1 | 0.000 |
| 22. Walking (yes) | 67.0 | 43.8 | 64.6 | 47.7 | 55.6 | 58.3 | 38.9 | 0.0 | 0.000 |
| 23. Ataxia (yes) | 1.7 | 0.0 | 7.3 | 4.5 | 0.0 | 8.3 | 5.6 | 0.0 | n.s |
| 24. Paralysis (yes) | 10.4 | 12.5 | 12.2 | 11.4 | 0.0 | 33.3 | 16.7 | 75.0 | 0.000 |
| 25. Sensory (yes) | 20.9 | 18.8 | 24.4 | 27.3 | 22.2 | 33.3 | 27.8 | 50.0 | n.s |
| 26. Muscle strength (yes) | 66.1 | 68.8 | 67.1 | 81.8 | 55.6 | 66.7 | 77.8 | 96.4 | 0.000 |
| 27. FIMTM (mean) | 112.9 | 97.3 | 113.7 | 101.6 | 101.2 | 92.8 | 90.7 | 28.4 | 0.000 |
| Dependent variables | | | | | | | | | |
| 28. Discharge destination (home) | 99.1 | 93.8 | 91.5 | 90.9 | 55.6 | 50.0 | 38.9 | 17.9 | 0.000 |
| The Kruskal-Wallis test was used for the continuous variables (numbers 2 and 27), and the χ2 test was used for the remaining categorical variables. n.s.: not significant. |
Discussion
For elderly patients, many factors, including personal factors (e.g. gender, sex, family structure, type of residence, residential address, social support, and income) and physical factors (e.g. underlying disease, state of ADL, presence of dementia), which are mutually related and confounded, affect the discharge destination (4, 9, 10, 15–18). For example, a patient’s discharge destination is not determined by his or her ADL status alone. Instead, factors such as the patient’s gender, family structure, and area of residence are thought to combine to affect the discharge destination. We examined multiple factors using SDA to identify those factors associated with a patient’s discharge destination. Initially, SDA was used to identify the factors associated with discharge to one’s own home. The patients were then divided into groups according to those factors, and the home discharge rate was quantified for each group. Our results provide useful information regarding the selection of a patient’s discharge destination. For the subgroups for which returning patients to their own homes was difficult, several contributing variables were identified: “poor ADL (FIMTM < 51)” (Group 8) (17.9% home discharge rate); “good ADL (FIMTM ≥ 52) and key person’s preferred discharge destination is other than home” (Group 7) (38.9% home discharge rate); “good ADL (FIMTM ≥ 52), key person’s preferred discharge destination is home, and patient has dementia and is male” (Group 6) (50.0% home discharge rate); and “good ADL (FIMTM ≥ 52), key person’s preferred discharge destination is home, and the patient has no dementia, is ≥ 80 years old, and the residence prior to hospitalization was not his/her own home” (Group 5) (55.6% home discharge rate; Fig. 1). Given a poor ADL (FIMTM < 51) or if the key person’s preferred discharge destination was not the patient’s own home, 60–80% of the patients were transferred to a nursing facility after their discharge from hospital. These data, together with the fact that securing nursing insurance and a guaranteed place in a nursing facility is a lengthy process (from application to decision), indicate that planning a patient’s discharge destination should begin as soon as he or she is admitted to the hospital. By doing so, medical resources can be conserved by eliminating hospitalization of patients waiting for a decision regarding their discharge destination. In addition, based on our results, predictions regarding a patient’s discharge destination can be made during the initial stage of hospitalization that will allow staff to create better-tailored rehabilitation programmes. Consequently, it may be possible to decrease the number of patients for whom the effect of intervention is small relative to the amount of rehabilitation given. For example, patients with poor ADL ratings, like those in Group 8, are likely to be discharged to facilities; thus, rehabilitation for those patients should be conducted on the premise that their discharge destination will be a nursing facility. Nursing facilities offer certain advantages for rehabilitation, such as being barrier-free relative to the average home; thus, it is less likely that a patient would be required to have advanced ADL abilities, such as the ability to go up and down stairs. In general, the amount of rehabilitation is an important factor in patient’s ADL improvement (28, 29). If a patient’s discharge destination can be predicted before rehabilitation begins, a decision can be made regarding whether training for a low level of independence in ADL is sufficient (e.g. bed and bathtub transfer), or whether training for a high level of independence in ADL is necessary with the premise that the patient will be living in his or her own home (e.g. going up and down the stairs). Making a timely decision regarding a patient’s discharge destination will therefore lead to the efficient use of healthcare resources.
Since the establishment of nursing insurance in 2000, diverse residence types have become part of the social insurance system in Japan. At present, elderly patients whose post-discharge residence is other than their own home have several options, including nursing facilities that offer long-term care, healthcare facilities that offer long-term care, designated sanatorium-type medical care facilities for those requiring care, nursing care centres and private senior citizens’ homes that offer care services for designated facility residents, and group homes for those with dementia that offer daily life care in a communal living environment. Each facility has its own characteristics; thus, the needs of the patient should be considered when making a selection. This study will be a useful resource for those considering which type of facility (i.e. home or nursing facility) is more suitable for specific patients after discharge because it takes into account several patient characteristics. For example, ADL abilities were high in Group 6, but dementia was observed. Therefore, the first choice for these patients would be a group home, where a small number of elderly patients with dementia could live together while receiving professional nursing care in a setting near their families. Similarly, Group 7 patients had high ADL abilities, but their key person was not amenable to the idea of discharging the patient to his or her own home. Although valid concerns exist regarding the daily lives of such patients, if they receive some support, they should be able to live independently. Therefore, nursing care centres and private senior citizens’ homes with care services for designated facility residents would be a viable choice for these patients. Other options exist, including remodelling residences and facilities so that they can receive patients, healthcare facilities for elderly patients requiring long-term care, designated sanatorium-type medical care facilities for elderly patients requiring long-term care, and designated facilities offering care services. The patients in Group 8 had decreased ADL abilities, so the presumed burden on caregivers is large. Therefore, designated welfare facilities for elderly patients requiring long-term care would be preferred, with the understanding that the patients would require assistance for such ADLs such as bathing, using the toilet and eating.
In a clinical setting, explanations that are easy to understand are important, not only for rehabilitation professionals, but also for patients and their families. Our findings should therefore prove useful in obtaining informed consent with respect to discharge destination from patients. For example, because patients with a specific set of attributes can be categorized into one of the isolated groups and the home return probability of each group is known, patients can be presented with their predicted discharge destination using the format “home return probability is __%”, based on our SDA results. Notably, our results do not differ greatly from those presented in previous reports and may therefore be used intuitively in a clinical setting.
This study offers several important findings. First, a previous study showed an association between a rehabilitation patient’s discharge destination and the key person’s destination preference (30); however, the association with other factors, specifically the manner of association, was unknown in the selection of the discharge destination. Our study, however, showed that for patients with good ADL abilities (FIMTM ≥ 52), the most important factor is the key person’s preference for the discharge destination (Fig. 1). Conversely, for patients with poor ADL, the key person’s preference is unrelated to the discharge destination. Secondly, the patient’s residence type prior to hospitalization (i.e. rental housing or one’s own home) was associated with the discharge destination for patients with certain conditions (Fig. 1). Specifically, the home return rate was high for patients who were 80 years or older with good ADL abilities (FIMTM ≥ 52) and no dementia and whose key person’s preferred discharge destination was the home. This finding may be due to patients who own their own homes having greater financial leeway, or more resources for remodelling than those in rental housing. This finding, however, may reflect residential circumstances specific to Japan; thus, further studies are necessary. Thirdly, for patients with specific conditions, associations were found between discharge destination and dementia and between discharge destination and sex. That is, for those patients with good ADL abilities (FIMTM ≥ 52) and dementia whose key person’s preference for discharge was the home, females had a higher home return rate than males (Fig. 1). Possible explanations for this result include the fact that the male group (Group 6) had a higher mean age, a larger body build, needed more nursing care, and tended to be more violent than the female group (Group 2).
This study has certain limitations. First, although relevant factors were used in the analysis, other important factors, such as those pertaining to financial status and the layout of a patient’s residence, were missing. Secondly, the data were collected at a single hospital, not at multiple hospitals. Because factors other than those used as explanatory variables in this study might potentially influence the determination of the discharge destination (e.g. family structure or income), additional studies are necessary to introduce further improvements to the predictive system.
In conclusion, we identified factors associated with the discharge destination of inpatients using SDA. Multiple factors, confounded in an overlapping manner, influenced the discharge destination chosen. SDA is a suitable method for this type of situation. Furthermore, we obtained meaningful findings from a practical viewpoint. Identifying those factors associated with the selection of a discharge destination in advance of the actual discharge is important for the smooth delivery of rehabilitation services.
References
1. Statistics and Information Department, Minister’s Secretariat, Ministry of Health, Labour and Welfare. Statistics abstracts on health and welfare in Japan, 2002. Tokyo: Health and Welfare Statistics Association; 2003.
2. Chae J, Johnston M, Kim H, Zorowitz R. Admission motor impairment as a predictor of physical disability after stroke rehabilitation. Am J Phys Med Rehabil 1995; 74: 218–223.
3. Ween JE, Alexander MP, D'Esposito M, Roberts M. Factors Predictive of stroke outcome in a rehabilitation setting. Neurology 1996; 47: 388–392.
4. Falconer JA, Naughton BJ, Dunlop DD, Roth EJ, Strasser DC, Sinacore JM. Predicting stroke inpatient rehabilitation outcome using a classification tree approach. Arch Phys Med Rehabil 1994; 75: 619–625.
5. Stineman MG, Granger CV. Outcome, efficiency, and time-trend pattern analyses for stroke rehabilitation. Am J Phys Med Rehabil 1998; 77: 193–201.
6. Giaquinto S, Buzzelli S, Di Francesco L, et al. On the prognosis of outcome after stroke. Acta Neurol Scand 1999; 100: 202–208.
7. Brosseau L, Philippe P, Potvin L, Boulanger YL. Post-stroke inpatient rehabilitation. I. Predicting length of stay. Am J Phys Med Rehabil 1996; 75: 422–430.
8. Sonoda S, Chino N, Domen K, Saitoh E. Changes in impairment and disability from the third to the sixth month after stroke and its relationship evaluated by an artificial neural network. Am J Phys Med Rehabil 1997; 76: 395–400.
9. Alexander MP. Stroke rehabilitation outcome: a potential use of predictive variables to establish level of care. Stroke 1994; 25: 128–134.
10. Granger CV, Hamilton BB, Fiedler RC. Discharge outcome after stroke rehabilitation. Stroke 1992; 23: 978–982.
11. Dejong G, Laurence GB. Predicting the stroke patient’s ability to live independently. Stroke 1982; 13: 648–655.
12. Kraemer HC. Assessment of 2 × 2 associations: generalization of signal detection methodology. Am Stat 1998; 42: 37–49.
13. Kraemer, HC, editor. Evaluating medical tests: objective and quantitative guidelines. Thousand Oaks, CA: Sage; 1992.
14. Winklby MA, Flora JA, Kraemer HA. A community-based heart disease intervention: predictors of change. Am J Publ Health 1994; 84: 767–772.
15. Lofgren B, Gustafson Y, Nyberg L. Cross-validation of a model predicting discharge home after stroke rehabilitation. Validating stroke discharge. Cerebrovascular Dis 2000; 10: 118–125.
16. Friedman PJ. Stroke outcome in elderly people living alone. Disability Rehabil 1995; 17: 90–93.
17. Jorgensen HS, Nakayama H, Raaschou HO, Olsen TS. Acute stroke: prognosis and predictors of the effect of medical treatment on outcome and health care utilization. The Copenhagen Stroke Study. Neurology 1997; 49: 1335–1342.
18. Wee JYM, Bagg SD, Palepu A. The Berg balance scale as a predictor of length of stay and discharge destination in a stroke rehabilitation setting. Arch Phys Med Rehabil 1999; 80: 448–452.
19. Hagihara A, Tarumi K, Miller AS, Nabeshima F, Nobutomo K. Work stress and alcohol consumption among white-collar workers: a signal detection analysis. J Stud Alcohol 2000; 61: 462–465.
20. Hagihara A, Tarumi K, Nobutomo K. Grouping participants for tailored worksite weight-loss programs: a signal detection approach. Am J Ind Med 2002; 41: 269–278.
21. Hagihara A, Tarumi K, Nobutomo K. Positive and negative effects of social support on the relationship between work stress and alcohol consumption. J Stud Alcohol 2003; 64: 874–883.
22. Nippon Ronen Igakkai, editor. Textbook of geriatric medicine. Japan: Medical View Company; 2003.
23. Data management service: guide for use of uniform data set for medical rehabilitation and the center for functional assesment research. Guide for use of the uniform data set for medical rehabilitation. Version 3.0. State University of New York at Buffalo, Buffalo; 1991.
24. American Psychiatric Association. Diagnostic and statistical manual of mental disorders: DSM-IV-TR. Washington, DC: American Psychiatric Association; 2000.
25. Tazaki Y, Saito Y, editors. Physical examination of the nervous system. Tokyo: Nanzando Co.; 1997.
26. Brunnstrom S, editor. Movement therapy in hemiplegia. New York: Harper & Row; 1970.
27. Hislop HJ, Montgomery J, editors. Daniels and Worthingham’s muscle testing: techniques of manual examination, 7th edn. Philadelphia: WB Saunders Co.; 2002.
28. Ottenbacher KJ, Janells S. The results of clinical trial in stroke rehabilitation research. Arch Neurol 1993; 50: 37–44.
29. Chen CC, Heinemann AW, Granger CV, Linn RT. Functional gains and therapy intensity during subacute rehabilitation: a study of 20 facilities. Arch Phys Med Rehabil 2002; 83: 1514–1523.
30. Toshimitsu W, Maya N, Akira M, Miki O, Yasufumi H, Motomi T, Shusuke K. Discharge placements of the totally dependent inpatient due to cerebrovascular diseases in the aged and their determining factors. Jpn J Rehab Med 1989; 26: 115–121.