OBJECTIVE: Recent reports demonstrated that low-frequency repetitive transcranial magnetic stimulation (rTMS) over the unaffected hemisphere improved the affected hand function in chronic stroke patients. We investigated whether 1 Hz rTMS improved the motor learning of the affected hand in patients after stroke.
DESIGN: A double-blind study.
Patients: Twenty patients with chronic subcortical stroke.
METHODS: The patients were randomly assigned to receive either a sub-threshold rTMS over the unaffected hemisphere (1 Hz, 25 minutes) or sham stimulation, and all patients performed a pinching task after stimulation. We evaluated the motor function of the affected hand and the excitatory and inhibitory function of the affected motor cortex by transcranial magnetic stimulation.
RESULTS: Compared with sham stimulation, rTMS induced an increase in the excitability of the affected motor cortex (p < 0.001) and an improvement in acceleration of the affected hand (p = 0.006). Moreover, the effect of motor training on pinch force was enhanced by rTMS (p < 0.001). These improvement in the motor function lasted for one week after rTMS and motor training (p < 0.001).
CONCLUSION: rTMS improved the motor learning of the affected hand in patients after stroke; thus, it can apply as a new rehabilitation strategy for patients after stroke.
Key words: repetitive transcranial magnetic stimulation, neuronal plasticity, motor learning, stroke, rehabilitation.
J Rehabil Med 2008; 40: 298–303
Correspondence address: Naoyuki Takeuchi, Department of Rehabilitation Medicine, Hospital of Hokkaido University, North14 West5 Sapporo JP-060-0814, Japan. E-mail: naoyuki@med.hokudai.ac.jp
Submitted August 16, 2007; accepted January 2, 2008
Introduction
Functional recovery after stroke is related to various plastic processes leading to central nervous system reorganization (1–4). Various concepts are emerging that aim to enhance the beneficial plasticity and thus improve functional recovery (3, 5). However, it is necessary to develop strategies for improving the beneficial effects of neuro-rehabilitative treatments.
Recent reports demonstrated that low-frequency repetitive transcranial magnetic stimulation (rTMS) over the motor cortex of the unaffected hemisphere improved the motor function of the affected hand in patients after chronic stroke (6–8). Inhibition of the excitability of the unaffected hemisphere by rTMS at 1 Hz would decrease the transcallosal inhibition (TCI) from the unaffected to the affected hemisphere and increase the excitability of the affected hemisphere; this ultimately would translate into improved motor function of the affected hand (7). This study was based on the hypothesis that the unaffected hemisphere is disinhibited due to a reduction in the TCI from the affected hemisphere. Consequently, this disinhibited, unaffected hemisphere may increase the TCI to the affected hemisphere and impair the function of the affected hand (4, 9, 10).
It was thought that the application of rTMS at 1 Hz over the unaffected hemisphere may be useful as a new rehabilitation therapy for stroke patients (6–8). However, in a previous study, a continuous improvement in the motor function could not be induced by using only a single rTMS (7). Therefore, for rehabilitation of patients after stroke, it may be important to impart additional motor training or use neuropharmacological intervention while the changes are being generated by rTMS; this would improve the motor function. In particular, motor training after rTMS appears to be an attractive approach for enhancing motor recovery. Modulating the activity of a given neural network by rTMS may render the system more receptive to the motor learning process, thereby enhancing its efficacy (7). However, to our knowledge, no studies have investigated the effects of motor training combined with 1 Hz rTMS over the unaffected hemisphere in patients after stroke. Therefore, we studied whether the combination of rTMS at 1 Hz over the unaffected hemisphere and motor training could improve the function of the affected hand in patients after stroke.
Methods
Subjects
The study population comprised 20 patients after stroke (mean age 62.3 (standard deviation (SD) 8.4) years (Table I)). The inclusion criteria were as follows: (i) first-time ischaemic stroke of more than 6 months duration; (ii) with subcortical infarction only, confirmed by magnetic resonance imaging (MRI); (iii) improved motor deficits of the unilateral upper limb to the extent that patients could perform a pinching task; and (iv) normal Mini-Mental State Examination score. The exclusion criteria were as follows: (i) severe internal carotid artery stenosis; (ii) seizure; and (iii) an intracranial metallic implant. The patients were randomly assigned to 2 groups: the rTMS group (10 patients) and the sham group (10 patients). The former received real rTMS, while the latter received sham stimulation.
All subjects gave their written informed consent, and the study protocol was approved by the local ethics committee of the Hokkaido University Graduate School of Medicine.
| Table I. Clinical characteristics of patients after stroke |
| Patient no. | Age (years)/sex | Duration after stroke (months) | Paretic side | FMS | Lesion site |
| Total (%) | Hand (%) |
| Real-rTMS group | | | | | |
| 1 | 43/M | 7 | L | 86 | 83 | Corona radiata, internal capsule |
| 2 | 56/M | 8 | R | 79 | 96 | Putamen |
| 3 | 71/M | 21 | R | 47 | 54 | Thalamus |
| 4 | 55/M | 60 | R | 77 | 88 | Putamen, corna radiata |
| 5 | 61/M | 10 | R | 33 | 29 | Basal ganglia, internal capsule |
| 6 | 70/M | 41 | R | 86 | 96 | Thalamus |
| 7 | 72/F | 18 | R | 77 | 83 | Corona radiata |
| 8 | 54/F | 21 | L | 58 | 54 | Corona radiata, internal capsule |
| 9 | 59/M | 60 | R | 67 | 58 | Corona radiata |
| 10 | 71/M | 8 | L | 47 | 38 | Internal capsule |
| Mean (SD) | 61.2 (9.7) | 25.4 (20.8) | | 65.7 (18.5) | 67.9 (24.3) | |
| Sham-rTMS group | | | | | |
| 11 | 64/M | 8 | R | 91 | 96 | Thalamus |
| 12 | 72/M | 24 | R | 58 | 63 | Putamen, corna radiata |
| 13 | 68/M | 21 | L | 44 | 50 | Internal capsule |
| 14 | 55/F | 9 | R | 67 | 63 | Basal ganglia, internal capsule |
| 15 | 60/M | 34 | L | 92 | 96 | Putamen, corna radiata |
| 16 | 70/M | 16 | L | 85 | 83 | Internal capsule |
| 17 | 52/M | 15 | R | 89 | 88 | Corona radiata, internal capsule |
| 18 | 67/F | 121 | R | 45 | 25 | Internal capsule |
| 19 | 55/M | 8 | L | 82 | 96 | Basal ganglia, internal capsule |
| 20 | 71/M | 88 | R | 39 | 46 | Thalamus |
| Mean (SD) | 63.4 (7.4) | 34.4 (38.6) | | 69.2 (21.2) | 70.6 (25.0) | |
| FMS: Fugl-Meyer scale (16) (percentages of maximum points in the upper limb (66 points) and in hand (24 points)); SD: standard deviation. |
Experimental design
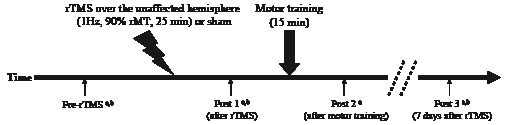
The measurements for assessing the motor function (acceleration and pinch force) were performed at pre-rTMS (Pre-rTMS) and post-rTMS (Post 1, immediately after the rTMS; Post 2, after motor training; and Post 3, 7 days after rTMS). The parameters of transcranial magnetic stimulation (TMS) (i.e. resting motor threshold (rMT), amplitude of motor evoked potentials (MEPs), and intracortical inhibition (ICI)) were evaluated at Pre-rTMS, Post 1 and Post 3. We did not evaluate the rMT, MEPs and ICI values immediately after motor training (Post 2) because the motor performance modulates the excitability of the motor cortex and ICI (11). It took 3 min to assess the motor function and 10 min to measure TMS parameters. Fig. 1 shows the time course of the experiment.
Fig. 1. Time course of the experiment. Repetitive transcranial magnetic stimulation (rTMS) was applied over the motor cortex of the unaffected hemisphere at a frequency of 1 Hz and a stimulus intensity of 90% resting motor threshold (rMT) for 25 min (or sham stimulation). After rTMS, the patients performed pinching task for 15 min as motor training. aThe measurements for assessing the motor function (acceleration and pinch force) were performed at pre-rTMS, Post 1, Post 2 and Post 3. bThe measurement of the transcranial magnetic stimulation parameters (rMT, amplitude of the motor evoked potentials (MEPs), and intracortical inhibition (ICI)) were performed at Pre-rTMS, Post 1 and Post 3.
TMS parameters
Single pulse TMS was performed using a 70-mm figure-8 coil and Magstim 200 (Magstim Co., Dyfed, UK), and rTMS was applied using the same coil and a Magstim Rapid stimulator (Magstim Co.). The coil was placed tangentially over the motor cortex at an optimal site for the first dorsal interosseous (FDI) muscle. The optimal site was defined as the location where stimulation at a slightly suprathreshold intensity elicited the largest MEPs in the FDI. This position was marked on the scalp and used throughout the experiment. Electromyographic (EMG) activity was recorded using silver–silver chloride (Ag-AgCl) electrodes positioned in a belly-tendon montage on the skin overlying the FDI. The signal was amplified, filtered (50–2000 Hz), and digitized at a sampling rate of 5000 Hz for off-line analysis (Neuropack; Nihon Koden, Tokyo, Japan). The rMT was determined separately for each stimulator and defined as the lowest stimulator output that could produce MEPs with a peak-to-peak amplitude greater than 50 µV in at least half of the 10 trials. The peak-to-peak amplitude of 10 averaged FDI responses obtained at 120% intensity of the rMT was also determined using Magstim 200.
Paired-pulse stimulation was performed to investigate the ICI in the affected motor cortex (12). To apply paired pulses, a figure-8 coil was connected to a Bistim device (Magstim Co.) that triggered 2 magnetic stimulators. The stimulus intensity of the first conditioning shock was 80% of the rMT and that of the second pulse was 120% of the rMT. We performed the tests at inter-stimulus intervals (ISIs) of 2 and 3 ms. Ten trials were recorded for each ISI, and unconditioned trials (controls) were recorded during complete relaxation. The paired stimulation with each ISI was randomly mixed with the control stimulation. The MEP amplitudes obtained by paired-pulse stimulation were expressed as a percentage of the mean control MEP amplitude, and the ICI was then calculated by averaging these values. We obtained ipsilesional TMS data from 11 patients (6 patients, rTMS group; 5 patients, sham group). We excluded other patients who did not display an MEP of the affected hemisphere from the ipsilesional TMS study section, i.e. patients in whom an MEP was not induced even at 100% stimulator output (4 patients, rTMS group; 5 patients, sham group).
rTMS and motor training
rTMS was applied over the motor cortex of the unaffected hemisphere at a frequency of 1 Hz and a stimulus intensity of 90% rMT measured with Magstim Rapid for 25 minutes (1500 pulses). These rTMS protocols used in the present study were in accordance with the safety guidelines for rTMS application to the motor cortex (13). Sham stimulation was applied over the unaffected hemisphere by positioning the coil perpendicular to the scalp (14) and at the same frequency and intensity used for real rTMS. After rTMS, the patients performed a pinching task for 15 min as motor training, as described in a previous report (15). During the pinching task, the patients were asked to perform a metronome-paced pinch of their index finger and thumb of the affected hand as fast as possible (frequency individualized between 0.3 and 0.5 Hz).
Evaluation of motor function
For assessing the motor function, we determined the pinch force and acceleration. The maximum pinch force of the affected hand was determined using a pinch gauge (Pinch Meter SPR-641; Sakai Medical, Tokyo, Japan). The subjects were instructed to use only their thumb and index finger during the pinch force measurements. Ten pinch forces were averaged in each session. Movement acceleration was measured using an accelerometer (model MP110-10-101; Medisens, Sayama, Japan) that was firmly attached to the dorsal side of the proximal phalanx of the thumb. The signal was amplified with a power signal conditioner (model MP110-10-301; Medisens) and digitized at 2000 Hz with a personal computer using a CB-68LPR board (National Instruments, Austin, TX) and LabView software (National Instruments). Fifteen peak accelerations were averaged in each session. The patients were allowed to familiarize themselves with this motor evaluation method on the day before the rTMS experiment.
Data analyses
Data analysis was performed by an investigator blinded to the stimulation type. The data (age, duration after stroke, Fugl-Meyer scale (16), and rMT) were compared between the rTMS and sham groups by using Student’s t-test. The effects of motor training or rTMS were evaluated using an analysis of variance (ANOVA) for repeated measures with TIME as a within-subjects factor and CONDITION (rTMS and sham) as a between-subjects factor. A post-hoc analysis was performed with Bonferroni correction. Any possible correlation between the changes in various parameters was determined by Pearson’s correlation coefficient test as an exploratory analysis. All data were normalized by conversion to percentage change from the mean values of Pre-rTMS.
Results
The subjects did not report any adverse effects during the course of the study. No difference was observed between the rTMS and sham groups with regard to the rMT (unaffected hemisphere: mean 46.9 (SD 9.0) vs 49.4 (SD 11.4) %; affected hemisphere: mean 62.2 (SD 12.3) vs 65.6 (SD 16.3) %), age, the duration after stroke, or Fugl-Meyer scale (Table I).
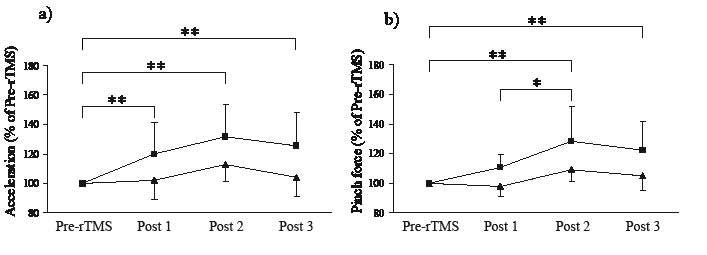
Fig. 2 shows the motor function after rTMS and motor training. A repeated measures ANOVA showed a significant interaction between TIME and CONDITION with respect to acceleration (F3,54 = 3.126, p = 0.033) and pinch force (F3,54 = 3.940, p = 0.013). It also showed a significant effect of TIME on both acceleration (F3,54 = 11.023, p < 0.001) and pinch force (F3,54 = 15.152, p < 0.001). The post-hoc test revealed an improvement in acceleration immediately after rTMS (Pre-rTMS vs Post 1: p = 0.006). This improvement in acceleration lasted for one week after rTMS (Pre-TMS vs Post 3: p < 0.001). The acceleration tended to increase after motor training; however, the effect of motor training on acceleration was not significant (Post 1 vs Post 2: p = 0.085). The post-hoc test did not show any significant improvement in pinch force immediately after rTMS (Pre-rTMS vs Post 1: p = 0.061). However, the motor training after rTMS improved the pinch force (Pre-rTMS vs Post 2: p < 0.001; Post 1 vs Post 2: p = 0.020). This improvement in pinch force also lasted for one week after rTMS (Pre-rTMS vs Post 3: p < 0.001). In the sham group, the motor function increased after motor training; however, the effect was not significant (Pre-rTMS vs Post 2: acceleration, p = 0.067; pinch force, p = 0.107).
Fig. 2. The effects of rTMS and motor training. (a) Acceleration; (b) pinch force. Repetitive transcranial magnetic stimulation (rTMS) improved the acceleration of the affected hand (Pre-TMS vs Post 1; p = 0.006) and this improvement in acceleration lasted for one week after rTMS and motor training (Pre-TMS vs Post 3; p < 0.001). The motor training after rTMS improved the pinch force of the affected hand (Pre-rTMS vs Post 2; p < 0.001; Post 1 vs Post 2; p = 0.020). This improvement in pinch force also lasted for one week after rTMS and motor training (Pre-rTMS vs Post 3; p < 0.001). *p < 0.05; **p < 0.01; Error bar, standard deviation; square, the rTMS group; triangle, the sham group.
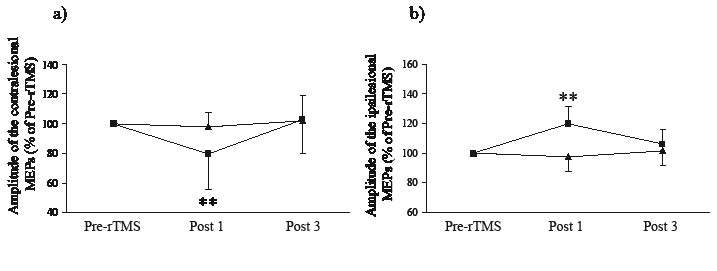
Fig. 3 shows the corticospinal excitability after rTMS. A repeated measures ANOVA for contralesional and ipsilesional MEPs showed a significant interaction between TIME and CONDITION (contralesional: F2,36 = 3.396, p = 0.047; ipsilesional: F2,18 = 5.867, p = 0.011) and a significant effect of TIME on both contralesional and ipsilesional MEPs (contralesional: F2,36 = 6.106, p = 0.005; ipsilesional: F2,18 = 3.946, p = 0.038). The post-hoc test revealed that a decreased contralesional MEP and an increased ipsilesional MEP were produced immediately by rTMS (Pre-rTMS vs Post 1; contralesional: p = 0.005; ipsilesional: p < 0.001) but not by sham stimulation (contralesional: p = 0.770; ipsilesional: p = 0.629). However, these changes induced by rTMS diminished at 7 days after rTMS (Pre-rTMS vs Post 2; contralesional: p = 0.652; ipsilesional: p = 0.225).
Fig. 3. The change in the corticospinal excitability after repititive transcranical magnetic stimulation (rTMS). (a) Amplitude of the contralesional motor evoked potentials (MEPs); (b) amplitude of the ipsilesional MEPs in the affected hemisphere. A decreased contralesional MEP and an increased ipsilesional MEP were produced immediately by rTMS (Pre-rTMS vs Post 1; contralesional: p = 0.005; ipsilesional: p < 0.001). However, these changes induced by rTMS had diminished at 7 days after rTMS. **p < 0.01. Error bar, standard deviation; square, rTMS group; triangle, sham group.
A repeated measures ANOVA for the rMT of the unaffected hemisphere (rTMS group: 102.8 (SD 9.0) % at Post 1 (% of Pre-rTMS) and 102.6 (SD 10.1) % at Post 3; sham group: 102.4 (SD 5.2) % at Post 1 and 102.0 (SD 6.8) % at Post 3) did not show a significant interaction between TIME and CONDITION (F2,36 = 0.019, p = 0.981); furthermore, there was no significant effect of CONDITION (F1,18 = 0.025, p = 0.877) or TIME (F2,36 = 1.550, p = 0.226) on the rMT of the unaffected hemisphere. A repeated measures ANOVA for the rMT of the affected hemisphere (rTMS group: 98.5 (SD 6.6) % at Post 1 (% of Pre-rTMS) and 99.7 (SD 5.4) % at Post 3; sham group: 99.8 (SD 4.6) % at Post 1 and 101.2 (SD 5.2) % at Post 3) did not show a significant interaction between TIME and CONDITION (F2,18 = 0.120, p = 0.888), and there was no significant effect of CONDITION (F1,9 = 0.234, p = 0.640) or TIME (F2,18 = 0.326, p = 0.726) on the rMT of the affected hemisphere. A repeated measures ANOVA for the ICI of the affected hemisphere (rTMS group: 96.4 (SD 29.9) % at Post 1 (% of Pre-rTMS) and 102.4 (SD 36.6) % at Post 3; sham group: 110.9 (SD 25.4) % at Post 1 and 104.3 (SD 30.1) % at Post 3) did not show a significant interaction between TIME and CONDITION (F2,18 = 0.333, p = 0.721), and there was no significant effect of CONDITION (F1,9 = 0.267, p = 0.618) or TIME (F2,18 = 0.088, p = 0.917) on the ICI of the affected hemisphere.
In the rTMS group, the improvement in the motor function after rTMS (Post 1) or motor training (Post 2) showed no significant correlation with the age of the subject, duration after stroke, the Fugl-Meyer scale, or the changes in ipsilesional MEPs and ICI.
Discussion
This study reports that non-invasive cortical stimulation using rTMS over the unaffected hemisphere can improve the motor learning of the affected hand in patients after stroke. These results demonstrate that priming by rTMS enhances the motor training effect of improving the affected hand function in patients after stroke.
We found that rTMS at 1 Hz over the unaffected hemisphere reduced the corticospinal excitability of this region; this result is in agreement with a previous report (17). Moreover, rTMS increased the corticospinal excitability of the affected hemisphere. This result is also consistent with that of a recent study (8). A previous study demonstrated that rTMS at 1 Hz over the unaffected hemisphere induced a decrease in the TCI from the unaffected hemisphere to the affected hemisphere (7). Therefore, rTMS at 1 Hz can facilitate the excitability of the affected hemisphere by reducing the TCI from the unaffected hemisphere. The enhancement of excitability in the motor cortex appears to be a necessity for motor learning (18). The motor cortex in humans is particularly engaged during the early stage of motor consolidation (19). Moreover, several studies have demonstrated that motor cortical plasticity depends on the motor cortex activation history (20–22). Based on these findings, the increased excitability of the affected motor cortex immediately after rTMS may contribute to a more suitable environment for the reorganization of the affected motor cortex by motor learning. Although the excitability of the affected hemisphere returned to baseline levels, the improvement in motor function continued for one week. Many studies have also reported that the acute effect of rTMS lasted for tens of minutes, as much as the stimulation period (23–25). Therefore, for improvement in motor function, it might be important that motor learning induced reorganization while the excitability of the affected motor cortex increased after rTMS. By another mechanism, rTMS over the unaffected hemisphere might reduce the disinhibition of the affected hemisphere, which was induced by the disruption of the TCI. A decrease in the inhibition unmasks the pre-existing, functionally latent neural networks around the lesion, thereby contributing to cortical reorganization (1). Kobayashi et al. (26) have reported that rTMS over the motor cortex induced disinhibition of the contralateral motor cortex. The disinhibition of the affected motor cortex may partly contribute to the functional improvement in the affected hand by unmasking the latent networks. However, we could not detect a change in the ICI of the affected motor cortex after rTMS. This hypothesis needs to be investigated using a larger number of stroke patients.
In patients after chronic stroke, the unaffected motor cortex might inhibit the motor performance of the affected hand via an abnormal TCI from the unaffected motor cortex to the affected motor cortex (7, 9). Several studies have suggested that the downregulation of the unaffected motor cortex results in an improvement in the motor function of the affected hand in patients after chronic stroke (6–8, 27). However, in patients after acute stroke, it is speculated that increased inhibitory input from the unaffected to the affected hemisphere might control the perilesional activity and reduce oxygen and glucose demands in the stroke penumbra in order to limit the extension of the lesion (2). Therefore, rTMS at 1 Hz over the unaffected hemisphere in acute stroke patients might lead to a poor prognosis due to the induction of an increase in neuronal death. Moreover, rTMS at 1 Hz over the unaffected hemisphere might induce the activation of the compensatory neural pathways, and ultimately, this activation might never result in a complete recovery. Further investigations are required to determine whether a low-frequency rTMS can promote recovery in acute stroke. Another concern of this study needs to be addressed. In this study, we selected patients with a better hand function who could perform pinching tasks and motor training. Moreover, the patients’ lesion was subcortical infarction only. Therefore, this study might have a decreased external validity due to the homogeneous nature of the experimental population. This fact encourages future studies to investigate other stroke populations with different stroke types and clinical characteristics.
A previous study reported that rTMS without motor training improved acceleration for not more than 30 min and that it did not modulate the pinch force (7). However, the combination of a previously reported rTMS protocol and motor training prolonged the improvement in acceleration for 7 days. In addition, motor training after rTMS at 1 Hz induced an increase in the pinch force that was not improved by rTMS alone. Fregni et al. (8) demonstrated that the effects of rTMS in patients after stroke were cumulative and lasted for at least 2 weeks. Therefore, rTMS may be important in rehabilitation of patients after stroke – to impart additional motor training while the changes are being generated by rTMS at 1 Hz and to conduct rTMS cumulatively; this would sustain the effect of rTMS and improve the function of the affected hand.
In conclusion, our results demonstrated that the combination of rTMS over the unaffected hemisphere and motor training could lead to an improvement in the motor function of the affected hand of patients after chronic stroke. These findings will probably be pertinent to the design and optimization of neurorehabilitation strategies for patients after stroke.
Acknowledgments
We thank Mami Onodera for technical support. This work was supported by research project grant-in-aid for scientific research No. 17300179 from the Japan Society for the Promotion of Science.
References
1. Chen R, Cohen LG, Hallet M. Nervous system reorganization following injury. Neuroscience 2002; 111: 761–773.
2. Pascual-Leone A, Amedi A, Fregni F, Merabet LB. The plastic human brain cortex. Annu Rev Neurosci 2005; 28: 377–401.
3. Hummel FC, Cohen LG. Drivers of brain plasticity. Curr Opin Neurol 2005; 18: 667–674.
4. Rijntjes M. Mechanisms of recovery in stroke patients with hemiparesis or aphasia: new insights, old questions and the meaning of therapies. Curr Opin Neurol 2006; 19: 76–83.
5. Ward NS, Cohen LG. Mechanisms underlying recovery of motor function after stroke. Arch Neurol 2004; 61: 1844–1848.
6. Mansur CG, Fregni F, Boggio PS, Riberto M, Gallucci-Neto J, Santos CM, et al. A sham stimulation-controlled trial of rTMS of the unaffected hemisphere in stroke patients. Neurology 2005; 64: 1802–1804.
7. Takeuchi N, Chuma T, Matsuo Y, Watanabe I, Ikoma K. Repetitive transcranial magnetic stimulation of contralesional primary motor cortex improves hand function after stroke. Stroke 2005; 36: 2681–2686.
8. Fregni F, Boggio PS, Valle AC, Rocha RR, Duarte J, Ferreira MJ, et al. A sham-controlled trial of a 5-day course of repetitive transcranial magnetic stimulation of the unaffected hemisphere in stroke patients. Stroke 2006; 37: 2115–2122.
9. Murase N, Duque J, Mazzocchio R, Cohen LG. Influence of interhemispheric interactions on motor function in chronic stroke. Ann Neurol 2004; 55: 400–409.
10. Lefaucheur JP. Stroke recovery can be enhanced by using repetitive transcranial magnetic stimulation (rTMS). Neurophysiol Clin 2006; 36: 105–115.
11. Liepert J, Weiss T, Meissner W, Steinrucke K, Weiller C. Exercise-induced changes of motor excitability with and without sensory block. Brain Res 2004; 1003: 68–76.
12. Kujirai T, Caramia MD, Rothwell JC, Day BL, Thompson PD, Ferbert A, et al. Corticocortical inhibition in human motor cortex. J Physiol (Lond) 1993; 471: 501–519.
13. Wassermann EM. Risk and safety of repetitive transcranial magnetic stimulation: report and suggested guidelines from the International Workshop on the Safety of Repetitive Transcranial Magnetic Stimulation, June 5–7, 1996. Electroencephalogr Clin Neurophysiol 1998; 108: 1–16.
14. Lisanby SH, Gutman D, Luber B, Schroeder C, Sackeim HA. Sham TMS: intracerebral measurement of the induced electrical field and the induction of motor-evoked potentials. Biol Psychiatry 2001; 49: 460–463.
15. Muellbacher W, Richards C, Ziemann U, Wittenberg G, Weltz D, Boroojerdi B, et al. Improving hand function in chronic stroke. Arch Neurol 2002; 59: 1278–1282.
16. Fugl-Meyer AR, Jaasko L, Leyman I, Olsson S, Steglind S. The post-stroke hemiplegic patient. 1. A method for evaluation of physical performance. Scand J Rehabil Med 1975; 7: 13–31.
17. Maeda F, Keenan JP, Tormos JM, Topka H, Pascual-Leone A. Modulation of corticospinal excitability by repetitive transcranial magnetic stimulation. Clin Neurophysiol 2000; 111: 800–805.
18. Pascual-Leone A, Tarazona F, Keenan J, Tormos JM, Hamilton R, Catala MD. Transcranial magnetic stimulation and neuroplasticity. Neuropsychologia 1999; 37: 207–217.
19. Muellbacher W, Ziemann U, Wissel J, Dang N, Kofler M, Facchini S, et al. Early consolidation in human primary motor cortex. Nature 2002; 415: 640–644.
20. Iyer MB, Schleper N, Wassermann EM. Priming stimulation enhances the depressant effect of low-frequency repetitive transcranial magnetic stimulation. J Neurosci 2003; 23: 10867–10872.
21. Siebner HR, Lang N, Rizzo V, Nitsche MA, Paulus W, Lemon RN, et al. Preconditioning of low-frequency repetitive transcranial magnetic stimulation with transcranial direct current stimulation: evidence for homeostatic plasticity in the human motor cortex. J Neurosci 2004; 4: 3379–3385.
22. Ziemann U, Iliac TV, Pauli C, Meintzschel F, Ruge D. Learning modifies subsequent induction of long-term potentiation-like and long-term depression-like plasticity in human motor cortex. J Neurosci 2004; 24: 1666–1672.
23. Chen R, Classen J, Gerloff C, Celnik P, Wassermann EM, Hallett M, et al. Depression of motor cortex excitability by low-frequency transcranial magnetic stimulation. Neurology 1997; 48: 1398–1403.
24. Muellbacher W, Ziemann U, Boroojerdi B, Hallett M. Effects of low-frequency transcranial magnetic stimulation on motor excitability and basic motor behavior. Clin Neurophysiol 2000; 111: 1002–1007.
25. Heide G, Witte OW, Ziemann U. Physiology of modulation of motor cortex excitability by low-frequency suprathreshold repetitive transcranial magnetic stimulation. Exp Brain Res 2006; 171: 26–34.
26. Kobayashi M, Hutchinson S, Theoret H, Schlaug G, Pascual-Leone A. Repetitive TMS of the motor cortex improves ipsilateral sequential simple finger movements. Neurology 2004; 62: 91–98.
27. Fregni F, Boggio PS, Mansur CG, Wagner T, Ferreira MJ, Lima MC, et al. Transcranial direct current stimulation of the unaffected hemisphere in stroke patients. Neuroreport 2005; 16: 1551–1555.