OBJECTIVE: To assess the quality of current evidence as to the effectiveness of robot-assisted gait training in spinal cord injured patients, focusing on walking ability and performance.
METHODS: A search was conducted in MEDLINE, Web of Knowledge, Cochrane Library, Physiotherapy Evidence Database (PEDro) and Digital Academic Repositories (DAREnet) (1990–2009). Key words included “spinal cord injury”, “(robot-assisted) gait rehabilitation” and “driven gait orthosis”. Articles were included when complete and incomplete adult spinal cord injured patients participated in robot-assisted gait training intervention studies. The methodological quality was rated independently by 2 researchers using “van Tulder criteria list” and “evaluation of quality of an intervention study”. Descriptive analyses were performed using the Population Intervention Comparison Outcome (PICO) method.
RESULTS: Two randomized controlled trials (mean quality score: 11.5/19) and 4 pre-experimental trials (mean quality score: 24.25 (standard deviation; SD 0.28)/48) involving 43 patients with incomplete, acute or chronic lesions between C3 and L1 were analysed. Five studies used the Lokomat and one used the LokoHelp. Although some improvements were reported related to body functions and activities, there is insufficient evidence to draw firm conclusions, due to small samples sizes, methodological flaws and heterogeneity of training procedures.
CONCLUSION: There is currently no evidence that robot-assisted gait training improves walking function more than other locomotor training strategies. Well-designed randomized controlled trials are needed.
Key words: spinal cord injury; robot-assisted gait training; locomotion training.
J Rehabil Med 2010; 42: 520–526
Correspondence address: Eva Swinnen, Vrije Universiteit Brussel, Faculty of Physical Education and Physiotherapy, Laarbeeklaan 103, BE-1090 Brussels, Belgium. E-mail: eswinnen@vub.ac.be
Submitted September 9, 2009; accepted January 21, 2010
Introduction
Locomotor ability is frequently affected in people with spinal cord injury (SCI). Decreased mobility is a factor associated with an increased risk of lower life satisfaction and quality of life post-SCI (1, 2). Improving mobility or the ability to get around in an energy-efficient manner may improve health-related quality of life in persons with SCI (3).
Different modalities of gait rehabilitation are used in the domain of neurological rehabilitation, such as manually assisted over-ground training and manually assisted treadmill training, both with or without BWS (4–6). Studies with neurological patients, such as those with stroke (7, 8) or incomplete SCI (4), showed that subjects improved their ability to walk on a treadmill and/or over-ground. However, the techniques are cumbersome and highly demanding in terms of staffing and time (9).
Robot-assisted gait training was introduced in the late 1990s. Nowadays, different systems are commercially available, including the “Lokomat” (10–13) and the “Gait trainer” (14). There are also systems designed specifically for research, such as “LOPES” (15, 16). The Lokomat is a motor-driven gait orthosis secured to a patient’s legs while the patient him/herself is supported by a body weight support (BWS) system over a motorized treadmill (17). The patient’s legs are guided on the treadmill according to a pre-programmed physiological gait pattern (10). A similar system is the lokoHelp, an electromechanical gait device fixed onto the band of a motor-driven treadmill. The device, used in combination with a BWS harness, transmits the treadmill movement to levers that induce the stance and swing phases (18).
Cost-effectiveness is an important factor in daily practice of rehabilitation training. The high cost of robot devices raises the question of efficiency in comparison with other training strategies. There is a huge difference in the cost of equipment used for conventional over-ground training or treadmill training with or without BWS. However, the reduced number of therapists involved in robot-assisted training should reduce the costs.
Thus far, there is no clear evidence for the effectiveness of robot-assisted gait training in terms of the SCI patient’s daily functional outcome, such as body functions (i.e. motor score and spasticity), activities (i.e. walking ability) and participation (i.e. ability to work). The aim of the present study was to reveal and qualitatively assess the current evidence with regard to robot-assisted gait training in persons with SCI, whether or not they were compared with other gait rehabilitation modalities. The focus of the study was on the effects on walking-related functional outcome measurements.
Research questions
The International Classification of Functioning, Disability and Health (ICF) (19) was used as a framework to define the following research questions:
• With regard to impairments in body functions: Do persons with SCI achieve reduced spasticity and/or a better motor score after robot-assisted gait training compared with before?
• With regard to limitation in daily activities: Do persons with SCI achieve a higher walking speed, walking endurance, greater step length and step symmetry, and better balance after robot-assisted gait training? Do they walk more independently (fewer walking aids, less support from others)? Do they have a higher level of activity of daily living after training? Are persons with SCI who could not walk before robot-assisted gait training able to walk over-ground or on a treadmill after training?
• With regard to restrictions in participation: Do persons with SCI achieve a higher level of social participation and quality of life after robot-assisted gait training than before?
• Finally: Do persons with SCI have a better outcome in the components of the ICF after robot-assisted gait training compared with matched subjects given another type of gait rehabilitation training?
Methods
Search strategy
A computerized search was conducted for English, French, German and Dutch articles published between 1990 and January 2009. The electronic databases PubMed (MEDLINE), Web of Science (ISI Web of Knowledge), Cochrane Library (Cochrane Controlled Trials Register), Physiotherapy Evidence Database (PEDro) and Digital Academic Repositories (DAREnet) were investigated by 1 researcher. Key words and combinations of key words were used to search the electronic databases and were organized following the Population Intervention Comparison Outcome (PICO) model (Table I). In this model the search strategy can be organized based on the topics: population (P), intervention (I), control group (C) and outcome (O). Different combinations of the topics can be made with the use of AND, OR and NOT in order to achieve a specific selection of literature (20). Together with the databases, the reference lists in the articles and narrative reviews were scanned separately for relevant publications. An independent selection in 3 categories (irrelevant, possibly relevant, and relevant) was made by 2 researchers after they had read the full texts.
| Table I. Key words and combinations of key words used in the search. The terms in the columns are allied with “OR”, and “AND” was used between the “P” (population), “I” (intervention) and “O” (outcome) columns |
| P: Population | I: Intervention | C: Comparison | O: Outcome |
| -(Incomplete) Spinal cord injury/injuries -Spinal fractures -Tetraplegia/quadriplegia -Paraplegia | -(Robot-assisted) gait rehabilitation/training -(Robot-assisted) step rehabilitation/training -Motorized rehabilitation/training -Automatic orthoses -Locomotor rehabilitation/training | -Conventional therapies | -Walking/gait speed -Walking/gait endurance -Walking capacity -Step/stride length -Balance -Spasticity -Walking independently -Walking aids -Support -Activity of daily living -Quality of life -Social participation |
Inclusion and exclusion criteria
Included were:
• Studies with adult (18+ years) patients with an acute or chronic SCI (cervical, thoracic and lumbar), incomplete or complete lesions, and an AIS (ASIA Impairment Scale) score of A (complete, no motor or sensory function in S4–S5), B (incomplete, sensory but no motor function below the neurological level and includes S4–S5), C (incomplete, motor function below the neurological level, and more than half of key muscles below this level have a muscle grade < 3) or D (incomplete, motor function below the neurological level, and at least half of key muscles below this level have a muscle grade ≥ 3) (21).
• Effect studies on robot-assisted gait rehabilitation programmes with the primary aim of improving gait function and which encompassed outcome scores on walking speed, walking endurance, walking independence, step length, balance, spasticity, walking aids, support by others, activity of daily living, quality of life and/or social participation.
Excluded were:
• Studies using functional electrical stimulation, surgery or the use of body-weight support systems without robot-assisted gait training;
• Studies with outcome exclusively focused on physical capacity, electromyographic activity and/or cardio-respiratory functioning;
• Animal studies or studies on children.
Methodological quality assessment
The studies were sorted into 3 categories: randomized controlled trials (RCT), quasi-experimental trials (clinical trials without random assignment), and pre-experimental trials (i.e. case reports, uncontrolled clinical trials) (22). The “van Tulder criteria list for the methodological quality assessment” (23, 24) was used to score the RCTs. This scale has a total maximum score of 19 points, subdivided into 11 points for the internal validity criteria, 6 points for the descriptive criteria and 2 points for the statistical criteria. For the quasi-experimental trials and pre-experimental trials, we used the “The evaluation of quality of an intervention study” (25). This rating system scores a maximum of 48 points, and consists of different components such as study question, study design, subjects, intervention, outcomes, analysis, and recommendations. Two researchers scored the studies independently and Cohen’s kappa was used to test inter-rater reliability.
Results
Methodological quality assessment
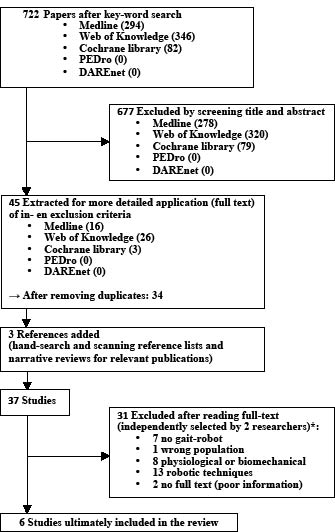
The flowchart (Fig. 1) gives an overview of the search strategy. Ultimately, 6 studies were included in the review (17, 18, 26–29). All the subjects in these studies were persons with an incomplete lesion.
Fig. 1. Search strategy. *The studies were selected independently by 2 researchers. There was initially a consensus between the 2 researchers for 26 of the 37 (70%) of the papers when sorted into 3 categories: irrelevant (excluded), possibly relevant (discussed to arrive at a consensus), and relevant (included). After discussion, there was a 100% final consensus between the 2 researchers. PEDro: Physiotherapy Evidence Database; DAREnet: Digital Academic Repositories.
Initially there was disagreement between the 2 raters about the methodological quality scores in 4 of the 38 items in the true RCTs and in 13 of the 96 items in the quasi-experimental trials and pre-experimental trials, resulting in a Cohen’s kappa of, respectively, 0.81 and 0.79. After using the consensus method, the mean van Tulder score of all the included studies was 11.5 and the mean score on the evaluation of quality of an intervention study was 24.25 (standard deviation(SD) 0.28). Only the study of Hornby et al. (17) showed a score on methodological quality below 50%, mainly due to low scores on the questions about study design, intervention and analysis.
Descriptive analysis
Following the PICO method, Table II presents the main characteristics, and the methodological scores of the included studies.
| Table II. Main characteristics and methodological scores of the included studies |
| Ref. | Study design | Population | Intervention (I) | Comparison groups (C) | Outcome |
| Author, year (ref) | Category/methodological quality | Lesion level | AIS-score (I/C) | Age (years) range (mean) | Time after injury | Sample size | Robot/training frequency (sessions) | Treadmill speed (kmph) | BWS (% body weight) | Patients | Therapy | Outcome measurements | Results |
| Field-Fote et al., 2005 (26) | RCT van Tulder score: 12/19 (IVC: 6/11, DC: 4/6, SC: 2/2) | C4, C5, C6, T2, T7, T10 | (I) | 37–56 (43.2) | 1 year, 1.8 years, 4.4 years, 10 years, 11.4 years, 23.4 years | 6 | Lokomat / 60’, 5×/week for 12 weeks (27–54) | Initially: 2.6 (↑0.16/week) finally goal: 3.2 | ≤ 30% | 3 × 7 SCI | BWS TT+ phys. assist. – BWS TT+ simul. – BWS OT+ stimul. | Walking speed, Step length, Step symmetry | Walking speed: ↑57% (slow walkers), ↓19% (fast walkers) Step length: ↓1% in stronger and 22% in weaker legs Step symmetry: ↑24% |
| Hornby et al., 2005 (27) | RCT van Tulder score: 11/19 (IVC: 6/11, DC: 4/6, SC: 1/2) | Above T10 | B/C/D (I) | | 14–180 days | 10 | Lokomat / 30’, 3×/week for 8 weeks | 2 | Initially: 25–78 finally: 0–78 | 2 × 10 SCI | BWS TT + phys. assist. – BWS OT | FIM-L, WISCI, 10MWT, 6MWT, LEMS, Ashworth Scale, SCATS, Balance, Forward reach, Timed up and go test | FIM-L, WISCI and LEMS: sign ↑ Ambulate without manual assistance: ± 50% |
| Wirz et al., 2005 (29) | Pre-experimental clinical trial (uncontrolled CT) Quality score: 28/48 | 2 × C3, 5 × C5, 2 × C6, 2 × C7, 1 × T1, 2 × T8, 1 × T9, 2 × T10, 3 × L1 | C/D (I) | 22–64 (40,5) | 5 × 2 years, 5 × 3 years, 3 × 4 years, 2 × 6 years, 1 × 8 years, 1 × 10 years, 1 × 13 years, 1 × 16 years, 1 × 17y | 20 | Lokomat / 45’, 3–5×/week for 8 weeks (24–37) | 1.5–2.4 | 0–50 (mean 37 ± 17) | | | WISCI-II, 10MWT, 6MWT, Timed up and go test, LEMS†, Ashworth scale†, SCATS† | WISCI II: ↑ 10MWT: ↑(mean 0.11 ± 10 m/s, 56% ± 60%) 6MWT: ↑(mean 32.3 ± 37.5 m, 53% ± 50%) Timed up and go test: ↑(mean 25 ± 30 s, 32% ± 19%) LEMS: ↑(mean 2.5 – initially: 32, finally: 35) Ashworth scale: no changes SCATS: ↓extensor spasm score |
| Freivogel et al., 2008 (18) | Pre-experimental clinical trial (case study) Quality score: 25/48 | T12 | C (I) | 37 | 16 months | 1 | LokoHelp / 30’, 3–5×/week for 6 weeks (20) | 0.3–1.8 | Initially: 10–30 | | | FAC, 10MWT, Motricity Index, Berg Balance Scale, Modified Ashworth scale, Rivermead Mobility Index | FAC: initially: 0, finally: 3 10MWT: initially: unable, finally: 0.2 Motricity Index: initially: 67, finally: 73 Berg Balance Scale: initially: 13, finally: 13 Modified Ashworth scale: initially: 0, finally: 0 Rivermead mobility index: initially: 5, finally: 7 |
| Winchester et al., 2005 (28) | Pre-experimental clinical trial (case study) Quality score: 24/48 | 2 × C5, 2 × C6 | C/D (I) | 20–49 | 14 weeks, 6 months, 1 years, 4 years | 4 | Lokomat / 60’ (≥ 20 minutes as + OT), 3×/week for 12 weeks | Initially: 2.0 finally: 2.3–3.2 | Initially: 35–59 finally: 0–44 | | | WISCI-II, Over-ground speed, Assistive device | WISCI II: ↑ Gait speed: ↑ Assistive device: P1: stand with assistance → ambulating + single-tip cane P2: walker + AFO + manual assistance → single tip cane + AFO P3: unable to stand → ambulate + forearm walker + manual assistance P4: unable to stand → unable to stand |
| Table I. Contd. |
| Ref. | Study design | Population | Intervention (I) | Comparison groups (C) | Outcome |
| Author, year (ref) | Category/methodological quality | Lesion level | AIS-score (I/C) | Age (years) range (mean) | Time after injury | Sample size | Robot/training frequency (sessions) | Treadmill speed (kmph) | BWS (% body weight) | Patients | Therapy | Outcome measurements | Result |
| Hornby et al., 2005) (17) | Pre-experimental clinical trial (case study) Quality score: 20/48 | C6, T2 | C (I) | 40–43 | | 2 | Lokomat / 60’, 1–3×/week for 7–9 weeks (20–27)* | 2.0–2.5 | Initially: 46–59 finally: 12–37 | | | LEMS, FIM, WISCI II, 10MWT, 6MWT, Timed up and go test, Functional reach test, Modified Sitting Functional Reach Test | LEMS: initially: 19–31, finally: 46–30 FIM: initially: 0–5, finally: 5–6 WISCI II: initially: 0–13, finally: 13–13 Gait speed: initially: 0.11m/s (p2), finally: 0.36–0.14 m/s Gait endurance: initially: 100 feet (p2), finally: 460–179 feet TUG: finally: 30.6 s (p1) Functional Reach Test: initially: > 10 (p2), finally: > 10–> 10 Modified Sitting Functional Reach Test: initially: 10 (p2), finally: 6–7 |
| IVC: internal validity criteria; DC: descriptive criteria; SC: statistical criteria; RCT: randomized clinical trial; CT: clinical trial; TT: treadmill training; OT: over-ground training; FIM-L: Functional Independence Measure – Locomotor subscale; LEMS: lower extremity motor score; 10MWT: 10-metre walk test; 6MWT: 6-minute walk test; SCATS: spinal cord assessment tool for spasticity; WISCI: walking index for spinal cord injury; FAC: functional ambulation categories; AIS: Asia Impairment Scale; BWS: body weight support; SCI: spinal cord injury. *Only this study described co-interventions (physical and occupational therapy); †n = 10 in 1 of the 4 centres. |
Comparison groups
The RCT by Field-Fote et al. (26) compared robot-assisted treadmill training with treadmill training with manual assistance, treadmill training with stimulation and over-ground training with stimulation. The RCT by Hornby et al. (27) compared robot-assisted BWS treadmill training with therapist-assisted BWS treadmill training and over-ground ambulation with a mobile system.
Training effects
Research question 1. Regarding body function, the RCT from Hornby et al. (27) reported only a significant improvement in Lower Extremity Motor Score after training. The RCT from Field-Fote et al. (26) did not use outcome measurements on this level. The pre-experimental trials reported no changes measured with the (modified) Ashworth scale (18, 29), but a substantial decrease in extensor spasm score after 8 weeks of training as evaluated with the Spinal Cord Assessment Tool for Spasticity (29). Furthermore, the pre-experimental trials suggested an improvement in lower limb strength as measured with the Motricity Index leg score (18). The outcome scores for the Lower Extremity Motor Score are discrepant: no changes (17) vs improvements (17, 29). Significant improvements were reported between 4 and 8 weeks training without specificity to any individual muscle group (29).
Research questions 2. With respect to activity, the RCT from Field-Fote et al. reported that the mean walking speed increased by 57% in a slower walking group, but decreased by 19% in a faster walking group. They also reported on step length, and noted a decrease of 1% in the stronger legs and 22% in the weaker legs. The step symmetry, the ratio of step length between the stronger and weaker leg, however, increased by 24% (26). The RCT of Hornby et al. described significant improvements after training in Functional Independence Measure Locomotor Subscale and Walking Index for Spinal Cord Injury-II scores (27). The pre-experimental trials reported improvements in walking speed (17, 18, 28, 29) and endurance (17, 29). The study of Wirz et al. (29) found a significant increase after training in mean over-ground gait speed of 56% (± 60%) in nearly all subjects and a relative increase of 53% (± 50%) in walking distance (29). Furthermore, in the pre-experimental trials, no changes were found in the Berg Balance Scale (18), whereas a decrease (in inches) was measured with the Functional Reach Test (17). Improvements were presented on the Rivermead Mobility Index (26) and the Functional Independence Measure Locomotor Subscale (17). Discrepant findings of no changes vs improvements (17, 28–29) were presented on the Walking Index for Spinal Cord Injury-II. Improvements were reported for the Functional Ambulation Categories (18), the devices the patients needed to walk (28) and the Timed Up and Go test in all but 2 of the ambulatory subjects, with a mean decrease in time to perform the test of 32% (± 19%) (29).
Research questions 3. No studies were found in the literature regarding outcome measures of participation after robot-assisted gait training in patients with SCI.
Research question 4. The RCT by Field-Fote et al. (26) reported that all modalities of locomotor training were associated with improved walking speed and that there were no obvious differences between the groups. The improvements appeared to be most drastic in subjects with the greatest initial impairment in walking function. Detailed statistical analyses suggested a trend for a greater improvement in the groups that were trained using electrical stimulation as an assistance for stepping. The authors described that subjects with better walking function had lower effectiveness with robot-assisted treadmill training (26). Results of the RCT by Hornby et al. (27) showed that mean changes in all treatment groups improved significantly during the training regimen, with significant changes in Functional Independence Measure Locomotor Subscale, Walking Index for Spinal Cord Injury scores and Lower Extremity Motor Score. There were no significant differences in the extent of motor or functional recovery between the treatment groups.
Discussion
The limited number of studies that could be included in this review, in combination with the small number of patients described in the studies and the fairly low methodological scores, demonstrate the low level of evidence currently available with regard to the effectiveness of robot-assisted gait training in persons with SCI. Some improvements were reported related to body function (i.e. motor function) and limitations in activities (i.e. walking speed). The outcome regarding participation was absent and the number of studies using comparison groups was low (n = 2). The number of intervention studies on robot-assisted gait training in persons with SCI, specifically high-quality RCTs, was limited. Several factors can explain this, such as the cost and availability of the robots, the complexity of the pathology (30), the difficulty of creating homogenous test groups, the complexity of gait rehabilitation (5) and the ethical difficulty of constraining co-interventions. The RCT of Hornby et al. (27) is not currently published in its full version, thus we could rely only on the report of the preliminary results.
The populations included in the 2 true RCTs were very different regarding time after injury (chronic lesions (26) vs sub-acute lesions (27)). The largest group of subjects in this review had a low cervical lesion level (C5–C6). This is in accordance with the epidemiological literature (31, 32). Still, there is a great deal of variation of lesion level within and between the studies in our review. The 2 RCTs in this review had a wide range of lesion levels, but all were above T10 (26, 27).
There is a great variability in the outcome measures used for assessing walking performance and ability, i.e. over-ground walking speed is measured in all the studies. However, different measuring modalities were employed to assess locomotor ability: the Walking Index for Spinal Cord Injury-II, the Functional Ambulation Categories and/or the Functional Independence Measure Locomotor Subscale.
The variability in population, training methods, and outcome measurements of the found studies makes it very difficult and not relevant to perform a meta-analysis.
We have to bear in mind that only one study, with only one subject made use of the LokoHelp instead of the Lokomat (18). Furthermore, no intervention studies were published assessing training protocols in patients with SCI by means of other gait training robots.
Mehrholz et al. (33) reviewed RCTs about the effect of locomotor training on the improvement in walking after traumatic SCI. The results covered by this review indicated that, for most of the participants included in the trials, the different types of locomotor training did not differ with respect to safety and acceptability. The authors reported insufficient evidence from RCTs to favour a specific locomotor training strategy for the improvement of walking function in patients with SCI (33).
There is some evidence about the effects of robot-assisted gait training on walking-related outcome measurements in persons with other neurological diseases, such as stroke (34) and multiple sclerosis (35, 36). Stroke patients who received electromechanically assisted gait training in combination with physiotherapy were more likely to achieve independent walking, but did not significantly increase walking velocity compared with patients receiving gait training without these devices (34). The evidence is more limited when we look at the multiple sclerosis population. Based on the 2 published articles currently available (35, 36), robot-assisted locomotor training may improve gait function in patients with multiple sclerosis, but the effect does not seem to be superior to standard training methods.
In conclusion, there is insufficient evidence to draw conclusions about the effectiveness of robot-assisted gait training in persons with SCI because of small samples sizes, methodological flaws and heterogeneous training procedures. Some improvements are reported after robot-assisted gait training in persons with SCI. However, when results are compared with other walking rehabilitation therapies, assessing walking over-ground or on a treadmill with or without BWS, improvements are not greater with robot-assisted gait training than with other training modalities (26, 27). There is a need for well-designed randomized multicentre clinical trials with large, but strictly selected, samples (i.e. with restrictions in AIS impairment score and lesion level) and relevant control groups who received more conventional gait training. There is a lack of clinical trials with extended follow-up periods. There is also a need for use of outcome measurements on the level of participation, including quality of life and social participation.
References