Marcos Antonio Eleutério-Silva, MSc1,7,8*, Lucas José Sá da Fonseca, MD1,7*, Elizabeth P. P. Velloso, PhD2,7, Glaucevane da Silva Guedes, MSc1,3,7, Walkyria O. Sampaio, PhD2,7, Weriton Ferreira da Silva, MSc1,7, Marco Antônio Mota-Gomes, MD4, Lucy V. da Silva Lima, MD, PhD5, Robson Augusto S. Santos, MD, PhD2,7 and Luíza A. Rabelo, PhD1,6,7
From the 1Laboratório de Reatividade Cardiovascular, Setor de Fisiologia e Farmacologia, Instituto de Ciências Biológicas e da Saúde, Universidade Federal de Alagoas, Maceió, Alagoas, 2Laboratório de Hipertensão, Departamento de Fisiologia e Biofísica, Universidade Federal de Minas Gerais, Belo Horizonte, Minas Gerais, 3Faculdade de Nutrição (FANUT), Universidade Federal de Alagoas, 4Faculdade de Medicina, Universidade Estadual de Ciências da Saúde de Alagoas (UNCISAL), 5Faculdade de Medicina (FAMED), Universidade Federal de Alagoas, Maceió, Alagoas, Brazil, 6Max-Delbrück-Center for Molecular Medicine, Berlin, Germany, 7Instituto Nacional de Ciência e Tecnologia em NanoBiofarmacêutica (N-BIOFAR), Belo Horizonte, Brazil and 8Laboratório de Fisiologia, Imunologia e Farmacologia, Unidade Acadêmica de Ciências da Vida, Universidade Federal de Campina Grande, Cajazeiras, Paraíba. *MAES and LJSF contributed equally to this study.
OBJECTIVE: To evaluate the impact of a short-term cardiovascular physical programme on the metabolic, anthropometric and oxidative stress parameters of women with metabolic syndrome.
METHODS: Thirty sedentary female patients, age range 30–60 years, were invited to participate in a 6-week cardiovascular physical programme. The training consisted of 60-min sessions of aerobic and strength exercises performed 3 times a week; a total of 18 sessions. Anthropometric data, functional exercise capacity, general biochemical profile, serum lipid peroxidation, superoxide dismutase and catalase activity in erythrocytes were evaluated according to standardized protocols. Peripheral vascular function was assessed using applanation tonometry. All assessments were performed before and after the training programme.
RESULTS: The physical programme proved effective in improving the distance covered in the 6-min walk test and in reducing arterial pressure levels, pulse pressure and the Augmentation Index, without modifying heart rate. The plasma thiobarbituric acid reactive substances levels, indicators of oxidative stress, were significantly decreased after the programme. Superoxide dismutase activity was increased in erythrocyte lysates, with no significant change in catalase activity. Waist circumference was significantly decreased compared with baseline.
CONCLUSION: These findings indicate that short-term combined aerobic and strength training may represent an important non-pharmacological approach for treating individuals with metabolic syndrome.
Key words: cardiometabolic diseases; physical training; vascular function; oxidative stress; functional exercise capacity.
J Rehabil Med 2013; 45: 572–579
Guarantor’s address: Luiza Rabelo, Pça Afrânio Jorge, S/N. Bairro Prado, Maceió 57010-020, Alagoas, Brazil. E-mail: luizaa.rabelo@gmail.com; luiza.rabelo@icbs.ufal.br
Accepted Jan 14, 2013; Epub ahead of print May 24, 2013
Introduction
Metabolic syndrome (MetS) is characterized as the coexistence of multiple cardiovascular risk factors, such as hypertension, insulin resistance, type 2 diabetes, central obesity and dyslipidaemia (1, 2), which mutually reinforce each other, resulting in endothelial dysfunction mainly through reduced nitric oxide (•NO) bioavailability and an imbalance between reactive oxygen/nitrogen species (RONS) and antioxidant capacity (3–6). New evidence suggests that oxidative stress is elevated in MetS, contributing to the aforesaid dysfunction (4–6). Furthermore, it is noteworthy that the balance between environmental levels of superoxide and released •NO plays a critical role in the maintenance of normal endothelial function (7, 8).
In this scenario, an important feature in patients with MetS is endothelial dysfunction, which progressively augments arterial stiffness (9, 10). The resulting haemodynamic abnormalities overload the cardiovascular system, reducing exercise capacity and augmenting morbimortality (9–12). With regard to the evaluation of vascular function, it has been demonstrated that arterial tonometry is a reliable method to evaluate arterial stiffness and estimate vascular functionality, since the aortic pulse wave velocity (PWV) and augmentation index (AI) are independently and inversely correlated with global vascular function (9–13).
In addition to traditional pharmacological treatments, changes in lifestyle are expected to prevent cardiovascular complications by augmenting endothelial function. In human studies, moderate physical training enhanced endothelium-dependent vasodilation in hypertensive and chronic heart failure patients (14), and in healthy individuals (15, 16).
Randomized intervention studies have provided strong evidence that physical training is effective as a non-pharmacological therapy for patients with chronic heart diseases (14), diabetes (17), and MetS-related symptoms (18, 19). The majority of these studies used training protocols lasting from 20 weeks to 12 months (15–18). Nevertheless, it is still unclear whether a short-term cardiovascular physical programme (CPP) can improve cardiovascular parameters and oxidative stress status in MetS. The purpose of the present study was to evaluate the impact of a short-term physical rehabilitation programme on metabolic and anthropometric parameters, and on markers related to oxidative stress in women with MetS. In addition, we evaluated, for the first time, the response of subjects with MetS to the 6-min walk test (6MWT), a well-known instrument for assessing the functional exercise capacity of patients affected by different chronic diseases (19, 20).
Methods
Subjects
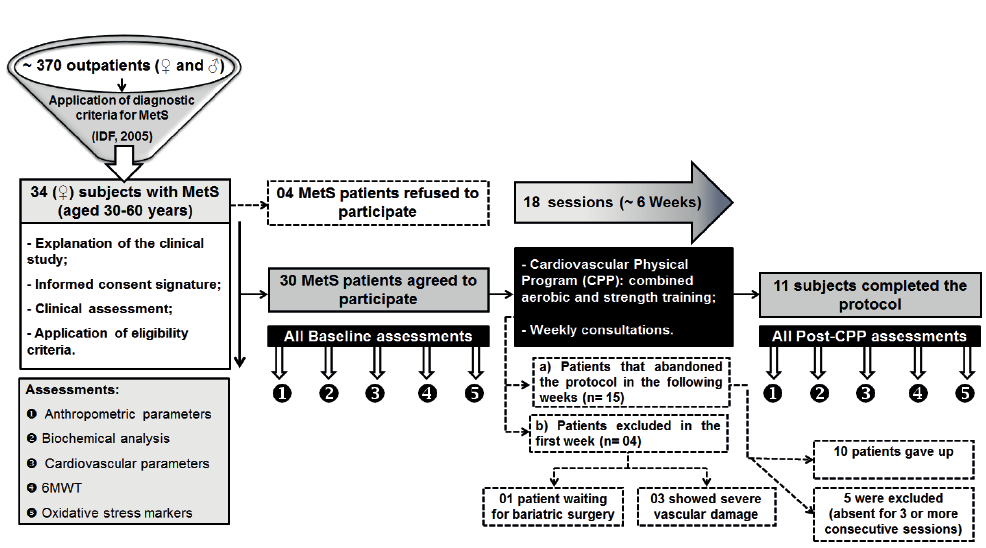
All patients were selected from the ambulatory of endocrinology at the Professor Alberto Antunes University Hospital (HUPAA), Federal University of Alagoas (UFAL). The service has a monthly flow of approximately 370 outpatients, of both sexes. Based on this initial population, over a period of 4 months, 34 female patients were diagnosed as presenting with MetS, established according to the International Diabetes Federation (2005) (2), being readily enrolled in the present study. All of them shared a similar sedentary occupational status and dietary habits (Fig. 1). Inclusion criteria were: patients aged 30–60 years, fulfilling the minimum criteria for MetS diagnosis. In this respect, for being diagnosed with MetS, at least 3 among 5 criteria should be found in women: (i) waist circumference ≥ 80 cm; (ii) triglycerides ≥ 150 mg/dl; (iii) high-density lipoprotein (HDL)-cholesterol < 50 mg/dl; (iv) systolic blood pressure ≥ 130 mmHg or diastolic blood pressure ≥ 85 mmHg; (v) fasting glucose ≥ 100 mg/dl. The following exclusion criteria were adopted: patients with severe metabolic or organic decompensation, chronic inflammatory diseases, amputees, patients with diseases that contraindicate physical training, patients with severe neurological diseases, the ones who did not attend 3 or more consecutive sessions, as well as those who, for any reason, did not agree to participate in this research protocol or wished to drop out. While explaining the protocol, 4 patients dropped out immediately due to work-related difficulties (2-shift working hours); thus a total of 30 sedentary female patients underwent the next evaluation stage. They were assessed by medical history, and clinical and laboratory examinations. At the beginning of the protocol, 4 patients were unable to participate in the study, once 1 of them was waiting for bariatric surgery and the remaining 3 had severe vascular damage. In the following weeks, 10 patients gave up and 5 were excluded because they were absent for 3 or more consecutive sessions. Thus, 11 patients completed the cardiovascular physical training protocol. The study protocols complied with the Guidelines of the ethics committee of the Federal University of Alagoas (protocol number 016516/2008-2) and conformed to the principles outlined in the Declaration of Helsinki (1964). All subjects signed an informed written consent form.
Cardiovascular physical programme
Thirty sedentary female patients, age range 30–60 years, were submitted to a cardiovascular physical training protocol 3 times a week, for 60 min per session, corresponding to 18 sessions (~6 weeks) (Fig. 1). The protocol was standardized in a preliminary study (unpublished data). The proposed cardiovascular programme consisted of combined aerobic and strength physical exercises, according to the following 4 phases:
• First phase. Warm-up: stretching and mobilization, aiming to reduce the risk of injury in later stages, and prepare the structures for musculoskeletal strength and aerobic training, lasting ~10 min. In this phase, 3 interventions were performed: shoulder and pelvic girdle dissociation, proprioception and balance training using warm-up sticks and Swiss balls, as well as self-stretching of the main anterior and posterior chain muscle.
• Second phase. Strength training: in order to improve functionality and strength, this step was carried out using dumbbells and exercise machines during ~15 min. One sort of exercise was performed for each of the considered muscles (concentrated on the main anterior chain). Thus, biceps and triceps underwent this phase on a double pulley system, while pectoral and abdominal oblique muscles were exercised using dumbbells. A leg extension machine was used for quadriceps muscle training. All equipments used were from the same make (Carci®, São Paulo, Brazil). In addition, the muscle strength and range of motion (ROM) test were also performed, aiming directly to evaluate the joints required in training, considering values from grades 5 to 0, according to the Oxford Scale (20). The maximum load and repetitions were evaluated using the protocol of 1-repetition maximum (1RM) (20). Thus, each patient performed 3 sets of 10 repetitions with previous determined weights. The strength training began with 50% of the maximum load determined at the 1RM test, being gradually increased from 7% to 10% every 6 sessions until the treatment ended.
• Third phase. Aerobic training: this step aimed to improve general physical function, and lasted ~30 min. For this, a treadmill (Moviment®, São Paulo, Brazil) without inclination was used, keeping the monitoring of cardiovascular parameters by using a heart rate monitor (Vantage XL Polar®, Finland), an oximeter (Onyx 9500® Nonin Medical Inc, Plymount, USA) and the rate of perceived exertion with the Borg scale (0 – no dyspnoea; 20 – maximal dyspnoea). The patients started with 50% of the heart rate reserve (HRR) for calculating the heart rate training (HRT), with progressive increases of 10% every 6 sessions until they reached 70% of HRT. The treadmill speed was controlled according to the heart rate assuming a normal range of 10% in either direction, and their perceived exertion, assessed at the 1st and the 5th min, and then every 5 min during the 30 min of the aerobic phase.
• Fourth phase. Cooling down: conducted to maintain the elasticity and promoting the return of blood pressure to resting values, and lasting ~5 min.
The physical training was carried out in a wide place, with groups of 2 or 3 people who completed all phases together, being constantly monitored at all stages under the supervision of a physical therapist. The whole protocol was accomplished at the School-Clinic of Rehabilitation of the Center for Higher Studies of Maceió (Centro de Estudos Superiores de Maceió; CESMAC, Maceió, Alagoas, Brazil).
Eleven subjects completed the training programme, and this group was further evaluated based on biochemical and cardiovascular parameters. All the measurements were taken before and post-cardiovascular physical programme (post-CPP) (Fig. 1).
Fig. 1. Study flow-chart. METS: metabolic syndrome; 6MWT: 6-min walk test.
Anthropometric parameters
Anthropometric measurements were taken in the standing position. Weight was measured on electronic scales to the nearest 0.1 kg, with patients wearing underwear. In addition, height was measured barefoot to the nearest 1 mm, using a stadiometer with the participant standing erect with the head in the anatomical position. Body mass index (BMI) was calculated as weight divided by the square of height (kg/m2). Waist circumference (WC) and waist-to-hip ratio (WHR) were measured with a heavy-duty inelastic fibreglass tape that was calibrated against a metal tape measure to ensure accuracy. The WC was measured at the part of the trunk located midway between the lower costal margin and the iliac crest at the end of a normal expiration and the WHR was calculated by measuring the smallest circumference of the waist, just above the umbilicus, and dividing it by the hip circumference at the widest part of the buttocks and hips.
Six-min walk test protocol
Functional exercise capacity was assessed by the 6MWT, according to the American Thoracic Society (ATS) guideline (21), with subjects advised to abstain from caffeine or alcohol for at least 4 h before performing the test. In brief, the protocol was conducted in an internal 50 m length closed and flat corridor without obstructions and foot traffic for 6 min (21). Instructions, such as “wear light clothes”, “walk as long and as fast as possible” and “slow down, stop, and rest as necessary” were given in advance. In addition, 5 min before the test, blood pressure was recorded in the sitting position with an oscillometric electronic pulse device (Microlife®,Widnau, Switzerland). Heart rate (HR) and oxygen saturation (SpO2) were recorded using a pulse oximeter (Onyx 9500®, Nonin Medical Inc, Plymouth, USA ) placed on the second finger of the right hand, these measurements corresponding to the zero time brands. Later, the same data acquisition was performed at the sixth minute regarding the end of the test, and after 5 min of rest. All participants were supervised by a physical therapist, the latter regularly encouraging the patients with standardized phrases in order to keep them walking. Besides the aforementioned measurements, respiratory rate (RR), systolic and diastolic blood pressure (SBP and DBP, respectively), and perceived exertion by the Borg scale (22) were also assessed at the beginning and end of the test. The total walking distance was measured in meters, and the mean walking speed lap by lap was calculated in m/s. Functional exercise capacity was evaluated using a standardized formula for women, as follows: 6MWD = (2.11 × height in cm) – (2.29 × weight in kg) – (5.78 × age) + 667 m. To define the lower limit of normal (LLN), 139 m is subtracted from the found value (23).
Blood and urine samples
Twelve-hour overnight fasting venous blood was drawn into cold tubes containing ethylenediaminetetraacetic acid (EDTA) and was centrifuged immediately at 4000 rpm for 10 min at 4ºC to separate plasma from blood cells. General biochemical profile and malondialdehyde (MDA) measurement were assessed in plasma, and enzymatic activities in erythrocytes. Twenty-four-hour urine was collected and stored in aliquots. All samples were frozen at −80ºC until analysis.
Biochemical analysis
Plasma fasting glucose, triglycerides, total cholesterol, very low-density lipoprotein (VLDL)-cholesterol and HDL-cholesterol were determined enzymatically (Cobas Mira Roche®, Basel, Switzerland). Concentrations of LDL-cholesterol were determined by the Friedewald method (24). Serum uric acid, glycosylated haemoglobin (HbA1C), and urine creatinine levels were measured at the Laboratory Medicine Unit of the HUPAA/UFAL by routine methods using an Olympus® AU 400 (Beckman Coulter, USA).
Cardiovascular parameters
The subjects were studied in the morning after an overnight fast. Studies were conducted in a quiet, temperature-controlled room (mean 22 ºC (standard deviation (SD) 2). All the haemodynamic records were made in triplicate. Three sets of records were made during a 45-min period of seated position, the last one taken as the baseline.
Blood pressure parameters (systolic blood pressure (SBP); diastolic blood pressure (DBP); mean arterial pressure (MAP); and pulse pressure (PP)) and heart rate (HR) were recorded using a validated oscillometric sphygmomanometer connected to an integrated software system. Indirect assessment of arterial stiffness was estimated by the AI (9, 27, 28) by applying the technique of applanation tonometry to the radial artery after resting for ≥ 15 min, using a HEM-9000AI device (Omron Corp®, Bangalore, India).
Oxidative stress markers assessment
In order to investigate the involvement of oxidative stress in the effects of cardiovascular physical rehabilitation, we evaluated plasma lipid peroxidation, measured by thiobarbituric acid reactive substances (TBARS) levels, and superoxide dismutase (SOD) and catalase (CAT) activities in erythrocyte lysate.
Measurement of lipid peroxidation. Lipid peroxidation was determined by measuring TBARS in plasma, as described by Ohkawa et al. (25). In parallel, MDA standards were diluted in the range of 0–4 μmol/l. TBARS values are expressed as MDA equivalents in nmol/ml. Absorbance was read at 532 nm and normalized by protein concentration.
SOD activity. SOD activity in erythrocyte lysates was measured with the SOD Assay Kit (Fluka® SOD Assay Kit BioChemika, Sigma-Aldrich, St. Louis, USA) according to the manufacturer’s protocol. SOD activity was calculated and expressed as units per mg of Hb.
CAT activity. CAT activity in erythrocyte lysates was measured spectrophotometrically as the rate of decomposition of H2O2, as described elsewhere (26), with appropriate adjustments for erythrocyte lysates. One unit of CAT activity was calculated kinetically at two time-points and defined as the rate constant of the first-order reaction (k). Relative activity was expressed as k/mg haemoglobin.
Statistics
For all the variables considered, normality was assessed by the Shapiro–Wilk test. Parametric variables were evaluated using the paired t-test. The Wilcoxon’s test was used for assessing the non-parametric variables. Results are presented as mean (SD) for those with normal distribution and as median values and interquartile range for the non-parametric ones. A p-value of less than 0.05 was considered statistically significant. GraphPad® Prism version 4.0 for Windows software (San Diego, CA, USA) was used.
Results
Clinical characteristics and biochemical measurements
Among the 30 women (mean 46.68 years (SD 3.43 years) diagnosed with MetS who participated in the 18-session physical protocol, only 11 completed the programme. The analysis of the biochemical profile of these subjects after the cardiovascular physical training revealed no differences in comparison with their baseline parameters (Table I).
|
Table I. Baseline and post-cardiovascular physical programme (post-CPP) biochemical parameters of the 11 subjects who completed the cardiovascular training |
||
|
Parameter |
Baseline |
Post-CPP |
|
Fasting plasma glucose, mg/dl, median (IQR)a |
84.90 (56.00) |
98.70 (13.00) |
|
Postprandial glycaemia, mg/dl, median (IQR)a |
114.10 (124.8) |
132.60 (26.70) |
|
Glycosylated haemoglobin, %, mean (SD)b |
6.25 (1.18) |
5.87 (1.23) |
|
Triglycerides, mg/dl, mean (SD)b |
153.63 (51.49) |
149.80 (43.83) |
|
Total cholesterol, mg/dl, median (IQR)a |
198.00 (52.00) |
209.00 (38.00) |
|
VLDL-cholesterol, mg/dl, median (IQR)a |
31.00 (19.00) |
26.80 (13.00) |
|
LDL-cholesterol, mg/dl, median (IQR)a |
117.00 (31.00) |
140 (41.00) |
|
HDL-cholesterol, mg/dl, mean (SD)b |
41.73 (9.62) |
41.09 (7.62) |
|
Creatinine, mg/dl, mean (SD)b |
0.55 (0.06) |
0.58 (0.06) |
|
Uric acid, mg/dl, mean (SD)b |
4.05 (0.78) |
4.29 (1.12) |
|
aWilcoxan test; bPaired t-test. VLDL-cholesterol: very low density lipoprotein-cholesterol; LDL-cholesterol: low density lipoprotein cholesterol; HDL-cholesterol: high density lipoprotein-cholesterol; SD: standard deviation; IQR: interquartile range. |
||
Effects of short-term cardiovascular physical programme on waist circumference
Body weight, BMI, WHR and WC measures before and after training indicated that the cardiovascular physical programme did not change the first 3 parameters (Table II). These characteristics were similar at 0 and 18 sessions of follow-up. On the other hand, WC was significantly decreased in comparison with baseline conditions (Table II).
|
Table II. Baseline and post-cardiovascular physical programme (post-CPP) anthropometry of the 11 subjects who completed the cardiovascular training |
||
|
Parameter |
Baseline Mean (SD) |
Post-CPP Mean (SD) |
|
Body weight, kg |
83.18 (26.00) |
81.45 (21.03) |
|
Body mass index, kg/m2 |
34.93 (7.54) |
34.2 (7.87) |
|
Waist-to-hip ratio |
0.83 (0.05) |
0.83 (0.02) |
|
Waist circumference, cm |
105.80 (16.58) |
102.4 (15.32)* |
|
*p < 0.05. SD: standard deviation. |
||
Cardiovascular parameters
To further investigate the effect of short-term physical training, we evaluated cardiovascular parameters and the variation in the AI by tonometry. Cardiovascular physical programme was effective in beneficially diminishing several cardiovascular parameters, including blood pressure levels, such as SBP (baseline: 122.00 (SD 37.00); post-CPP: 112.00 (SD 12.00); p = 0.001; Fig. 2A), DBP (baseline: 75.30 (SD 9.00); post-CPP: 69.50 (SD 9.33); p = 0.0087; Fig. 2B), PP (baseline: 48.50 (SD 25.30); post-CPP: 41.50 (SD 12.70); p = 0.0098; Fig. 2C), and MAP (baseline: 90.66 (SD 19.88); post-CPP: 83.72 (SD 9.95); p = 0.0010; Fig. 2D). However, there was no significant change in heart rate (Fig. 2E). On the order hand, the AI, a marker of arterial stiffness, was shown to be decreased after the cardiovascular physical programme (baseline: 93.06 (SD 15.23); post-CPP: 86.27 (SD 12.48); p = 0.026; Fig. 2F).
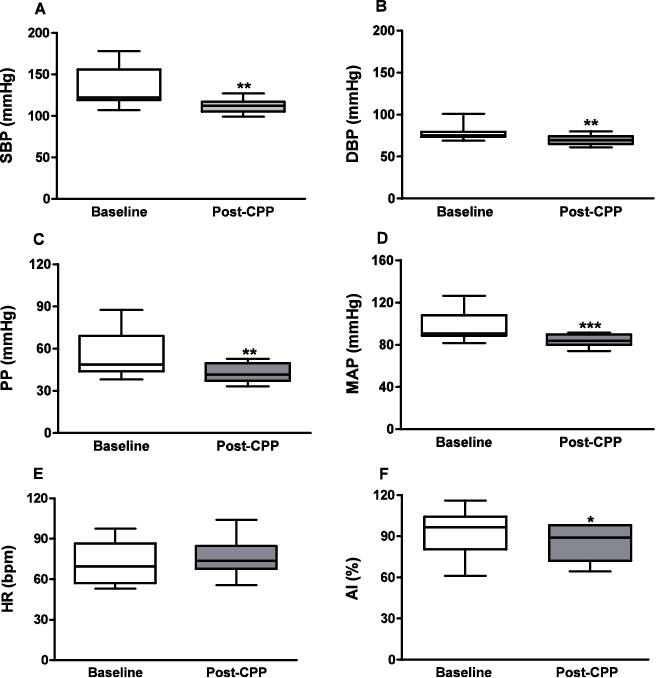
Fig. 2. Cardiovascular data: (A) systolic blood pressure (SBP); (B) diastolic blood pressure (DBP); (C) pulse pressure; (D) heart rate (HR); (E) mean arterial pressure (MAP); (F) Augmentation Index (AI) at baseline and post-cardiovascular physical programme ((post-CPP); n = 11) of patients with metabolic syndrome (MetS). Box plots represent mean values (standard deviation) for AI (paired t-test), and median values ± interquartile range (IQR) for the remaining variables (Wilcoxon test). *p < 0.05; **p < 0.01; ***p < 0.001.
Short-term cardiovascular training improved functional exercise capacity
For the functional exercise capacity assessment, our results indicated that the walking distance (baseline: 544.30 (SD 70.50); post-CPP: 577.10 (SD 70.25); in m; p = 0.0195; Fig. 3A) and speed (baseline: 1.51 (SD 0.19); post-CPP: 1.60 (SD 0.19); in m/s; p = 0.0230; Fig. 3B) were significantly higher after the cardiovascular training. In addition, functional exercise capacity was significantly increased after the CPP (baseline: 103.00 (SD 10.48); post-CPP: 109.20 (SD 11.23); p = 0.0322; Fig. 3C).
Fig. 3. The 6-min walk test (6MWT) performance: (A) distance walked during the 6MWT; (B) walking speed; and (C) functional exercise capacity at baseline and post-cardiovascular physical programme (post-CPP; n = 11) of the patients with metabolic syndrome. Each box plot represents mean values (standard deviation) (*p < 0.05, paired t-test).

The 18-session cardiovascular physical programme decreased systemic oxidative stress
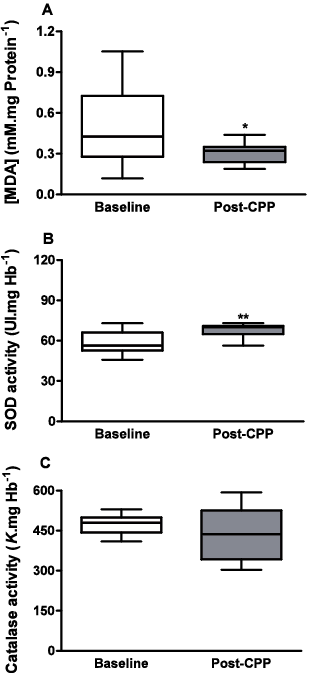
The results indicated that plasma TBARS levels were significantly decreased in the patients with MetS after the 18-session physical programme in comparison with baseline conditions (baseline: 0.48 (SD 0.28); post-CPP: 0.30 (SD 0.08); p = 0.492; Fig. 4A). Interestingly, SOD activity was increased in erythrocyte lysates obtained from these patients (baseline: 56.43 (SD 13.68); post-CPP: 70.03 (SD 6.23); p = 0.0098; Fig. 4B). Nevertheless, we observed no significant change in CAT activity (baseline: 469.40 (SD 37.42); post-CPP: 449.80 (SD 97.24); p = 0.3587; Fig. 4C).
Fig. 4. Oxidative stress and reactive oxygen species degrading enzymes. (A) Plasma concentration of the thiobarbituric acid reactive substances (TBARS) malondialdehyde (MDA) at baseline and post-cardiovascular physical programme (post-CPP; n = 11) in the patients with metabolic syndrome; (B) superoxide dismutase (SOD); (C) catalase (CAT) activity in erythrocyte lysate in similar conditions. Box plots represent mean (standard deviation (SD)) for TBARS and catalase (paired t-test), and median values ± interquartile range for SOD (Wilcoxon test). *p < 0.05.
Discussion
The major finding of this study indicates that short-term cardiovascular training improves cardiovascular function and performance in the 6MWT in women with MetS, and that these effects trigger a reduction in the markers of systemic oxidative stress.
This study found that the 18-session (~6 weeks) cardiovascular physical rehabilitation was efficient in improving several cardiovascular parameters, and that short-term physical rehabilitation not only lowers blood pressure, but also improves the AI, a quantitative index of systemic arterial stiffness. In contrast, the heart rate values were not affected by the training.
A number of other studies of patients with cardiovascular diseases have found a close relationship between various indices of arterial stiffness and endothelial dysfunction (6–9, 27–29). However, arterial stiffness evaluation often requires invasive procedures and/or highly specialized instruments and operators. McEniery et al. (9) demonstrated that endothelial function is associated with pulse pressure, pulse wave velocity, and AI in healthy humans. Therefore, evaluation of AI by arterial tonometry may represent a new, relatively simple non-invasive technique, which may serve to assess the predictive value of endothelial function, since the latter is associated negatively with the AI (9–11). Hence, this study and others (9–14) suggest that a peripheral AI, assessed by applanation tonometry, may be a useful alternative method to estimate improved vascular function. To the best of our knowledge, we are the first to apply this method to assess the effects of physical programme on patients with MetS. Therefore, we believe that this technique is suitable for assessing endothelial function in a large number of patients, and can answer the important question of the predictive value of endothelial function, including assessment of the effects of physical rehabilitation.
Recent epidemiological studies have demonstrated that the increase in pulse pressure, determined by ventricular interaction with the elastic properties of the large arteries and the magnitude of wave reflection (11, 27), is independently associated with adverse cardiovascular events (9, 11, 27–29). In fact, MetS is recognized as a cluster of cardiovascular risk factors, which are frequently associated with insulin resistance and hyperglycaemia. Moreover, arterial stiffness is significantly related to MetS, as well as to other diseases, such as ischaemic events, hypertension and diabetes (6, 13, 27–29).
In addition, Moon et al. (28) showed that an increase in the MetS score is an independent determinant of increased PP. In our study, we observed a decrease in PP after 18 rehabilitation sessions. Both PP and AI were lower after the physical programme compared with baseline conditions (Fig. 2C and 2F, respectively). Furthermore, this study showed significant concomitant changes in systolic, diastolic and mean arterial pressure (Fig. 2A, 2B, and 2D, respectively). It is important to emphasize that there is a positive correlation between arterial stiffness and AI (9, 11). We suggest that the significant reduction in the AI observed in our study after physical rehabilitation may be a consequence of improved adaptation of blood vessels to blood flow through a better physiological response to this mechanical force, which seems to improve the shear stress. We believe there is a close relationship between this improved vascular function and the functional physical capacity of patients after training (once circulation improves, the muscles are also able to work more efficiently). Thus, we suggest that the short-term physical programme may be a useful non-pharmacological approach to reduce MetS risk factors by improving vascular function.
The balance between superoxide anions and •NO released within the endothelial environment plays a critical role in maintaining normal endothelial function (3, 6–8). It has been shown that aerobic exercise augments •NO release (30) through vascular shear stress (14–16). On the other hand, local metabolic adaptations that occur in skeletal muscle could improve the pathways of lipid metabolism (31). These events may be associated with reduction in WC and improvement in cardiovascular parameters, such as the decrease in both systemic blood pressure and AI (Fig. 2) observed in the present study. Thus, a possible pathway whereby short-term aerobic and endurance exercise augments •NO release is via an increase in vascular shear stress as a result of increased blood flow. These effects could be correlated with the reduction in oxidative stress observed after 18 physical training sessions (Fig. 4).
Our findings are consistent with those of Niebauer & Cooke (32), who demonstrated that exercise-induced increases in shear stress can lead to functional and histological alterations in the vascular endothelium, resulting in enhanced vascular structure and function. Consistent with this observation, Sonnenschein et al. (33) recently applied an 8-week exercise training programme (5 days/week, 30 min/day on a bicycle ergometer) and found that regular moderate physical exercise was effective in restoring impaired in vivo endothelial capacity of early endothelial progenitor cells in subjects with MetS. They identified decreased superoxide production and increased •NO production of such cells, as well as improved endothelium-dependent vasodilation. Furthermore, other authors have demonstrated changes in microvascular function after exercise training, based on the measured coronary flow reserve ratio in response to adenosine (31). Patients showed a 29% increase in coronary flow reserve after exercise, which is also indicative of enhanced sensitivity of coronary microcirculation to adenosine.
Fujita et al. (5) showed that the increase in systemic oxidative stress observed in MetS subjects was strongly correlated with visceral fat accumulation. Moreover, plasma MDA, an index of lipid peroxidation in the circulating blood and endothelial membrane, was significantly and positively correlated with BMI and WC, indicating that fat accumulation is very closely correlated with oxidative stress in humans. Hence, the authors suggest that systemic oxidative stress might represent a crucial connection in the positive cycle created by accumulated visceral fat and MetS. We observed a reduction in both TBARS (Fig. 4A) and WC (Table II), suggesting an improvement in the cardiovascular status of patients with MetS after the short-term rehabilitation programme. In addition to lower TBARS levels, we also found significantly higher SOD activity, measured in erythrocyte lysates in the post-CPP period (Fig. 4B). This enzyme represents the first defence against superoxide anions, converting superoxide radicals into hydrogen peroxide (3). However, CAT activity remained unchanged (Fig. 4C). In addition, although no changes were observed in other anthropometric parameters (Table II) in our study, both TBARS and WC were decreased, indicating an improvement in physiological status of the patients with MetS after the short-term physical programme.
The findings of our study are consistent with those of previous observations cited above, in that they reveal a concomitant increase in the antioxidant defence mechanism, as indicated by the enhanced SOD activity, and decrease in endogenous oxidative stress. These data provide strong evidence of the role of oxidative stress markers in improved cardiovascular function after the short-term cardiovascular training. In addition, our group found that the serum nitrite concentration increased after the physical programme (unpublished data), which is noteworthy, since nitrite appears as an indirect indicator of release of the vasodilator •NO (6, 34).
In an elegant study, Kinugawa et al. (35) showed that exercise capacity is reduced in conditions in which superoxide anion is increased, and that this reduction is associated with a greater increase in whole-body oxygen consumption in SOD2+/– compared with SOD2+/+ mice. In their study, intraperitoneal administration of Tempol, a stable membrane permeable and metal-independent SOD mimetic, over a period of one week improved exercise capacity and the control of whole-body oxygen consumption (VO2) in SOD2+/– mice. However, they did not observe any effect in wild-type mice, highlighting the potential role of this enzyme in functional exercise capacity during certain pathological states.
The 6MWT is a quick and inexpensive way to measure physical function, and enables the estimation of an important component of the quality of life, since it reflects the capacity to undertake activities of daily living (19, 22, 23). Moreover, the results of our study indicate that the 6MWT could be useful, representing a cost-effective alternative for the assessment of functional exercise capacity in patients with MetS. In addition, the improvement in cardiovascular parameters demonstrated in this study after an 18-session physical rehabilitation programme is probably the main factor responsible for the improved functional exercise capacity evaluated by the 6MWT (Fig. 3C). However, the exact cellular mechanism by which the short-term cardiovascular training enhanced the cardiovascular profile in women with MetS merits further investigation. A potential pathway underlying these effects probably involves a reduction in oxidative stress and an improvement in endothelial function (34, 35). Nevertheless, additional data on this issue are needed to clarify the pathways involved in these effects. In addition, such studies could elucidate the significance of endogenous free radicals in the regulation of vascular function in both healthy individuals and those with cardiovascular disease.
There are some limitations to the present study. Firstly, a small number of patients was enrolled, which does not allow us to ensure that the lack of statistical significance for some variables would occur in the general population. Notwithstanding, the significant differences observed for other variables point to the important role of the combined cardiovascular physical programme proposed on cardiometabolic and oxidative parameters. Secondly, measurement variation may occur when assessing anthropometric data, such as waist circumference. However, despite the fact that it is impossible to rule out the possibility of this occurrence, the authors highlight that all measurements were taken by a single researcher broadly familiarized with the protocol and extensively trained for collecting anthropometric data. Furthermore, the same instrument (a validated inelastic fibreglass tape) was used for measuring the waist circumference before and after the proposed protocol. Regarding the oxidative stress experiments, as well as for all other data collection, the same researcher was responsible for the analysis steps. Taken together, these statements considerably reduce the possibility of measurement bias in the study.
In summary, the combined cardiovascular physical programme was effective in diminishing systemic arterial pressure levels, pulse pressure and AI, but did not modify heart rate or biochemical parameters. Moreover, after training, the increased physical capacity was accompanied by a reduction in abdominal fat, suggesting a close correlation with the markers of systemic oxidative stress. More importantly, these physiological effects provide novel insights into cardiovascular physical programmes, demonstrating that short-term physical training may represent an important non-pharmacological approach for treating individuals with MetS.
AcknowledgEments
The authors are indebted to Professor Ana Lúcia Gusmão and Professor Dr Mauro Guilherme Barros Quirino Martins for providing the physical space required for this study at the Rehabilitation Clinic of the Center for Higher Studies of Maceió (CESMAC). The authors would also like to thank the subjects of this study for their participation, as well as Valéria Nunes de Souza for providing technical assistance and Maria Lúcia F. Leite for her contribution to the statistical analysis.
This work was supported by grants from Brazil’s Ministry of Health /CNPq/SESAU-AL/FAPEAL (PPSUS-2006 PROJ_893_11388592) and PROCAD-NF/CAPES (PROCAD-NF 2450/2008).
References
