Masahiro Tsujikawa, MD1, Yohei Otaka, MD1, 2, Rei Hasegawa, MD, PhD3, Kunitsugu Kondo, MD, PhD2 and Meigen Liu, MD, PhD1
From the 1Department of Rehabilitation Medicine, Keio University School of Medicine, Tokyo, 2Department of Rehabilitation Medicine, Tokyo Bay Rehabilitation Hospital, and 3Department of Cardiology, Yatsu Hoken Hospital, Chiba, Japan
OBJECTIVE: To evaluate cardiac function using echocardiography in patients with stroke admitted to subacute rehabilitation units.
DESIGN: Retrospective, cross-sectional study.
PARTICIPANTS: A total of 750 consecutive patients with stroke who were admitted to a suburban rehabilitation hospital. Mean age 67.5 years (standard deviation (SD) 12.3 years). Mean time since stroke 36.7 days (SD 13.2 days).
METHODS: Patients were assessed using transthoracic echocardiography within 7 days of admission. The prevalence of echocardiographic abnormalities was analysed and compared between cerebral infarction and haemorrhage using the Mantel-Haenszel method controlled for age.
RESULTS: Arrhythmias were found in 13.7% of the patients, 94.2% of whom had atrial fibrillation. Left atrial enlargement and left ventricular hypertrophy were found in 20.4% and 19.5% of all patients, respectively. Left ventricular asynergy was detected in 6.1% of all patients, but 47.8% of them had no history of myocardial infarction. Left ventricular ejection fraction was low in 12.2% of all patients. Abnormal rhythms and left atrial enlargement were significantly more frequent in patients with cerebral infarction than in those with cerebral haemorrhage (p < 0.01).
CONCLUSION: The prevalence of cardiac problems is high among patients with subacute stroke regardless of a history of heart disease, and this should be taken into account when planning rehabilitation programmes.
Key words: comorbidity; risk factor; convalescent hospital; cardiac function test; heart disease.
J Rehabil Med 2014; 46: 00–00
Correspondence address: Yohei Otaka, Department of Rehabilitation Medicine, Keio University School of Medicine, 35 Shinanomachi, Shinjuku-ku, Tokyo, Japan. E-mail: otaka119@mac.com
Accepted Jun 10, 2014; Epub ahead of print Sep 3, 2014
INTRODUCTION
Stroke is a leading cause of admission to subacute rehabilitation units in Japan (1). The risk factors for stroke, such as hypertension, diabetes mellitus and hyperlipidaemia, are also associated with heart disease, especially ischaemic heart disease. Thus, patients with stroke may also have cardiac problems, such as atrial fibrillation and chronic heart failure, which could be inhibitory factors in rehabilitation as well as risk factors for stroke recurrence. Therefore, understanding cardiac problems is very important in designing successful and safe rehabilitation programmes for patients with stroke.
Cardiac problems are usually assessed using electrocardiography and echocardiography. The latter is a particularly important tool because it can measure cardiac chamber size, ventricular mass and ventricular systolic function. Several studies have evaluated abnormalities on electrocardiograms from patients with stroke (2–10). However, only a few studies have evaluated echocardiographic abnormalities, and their aim was to detect sources of embolism (11–14).
The aims of the current study were to explore the prevalence of echocardiographic abnormalities in patients with stroke who were admitted to subacute rehabilitation units in Japan and to compare the prevalence of such abnormalities between patients with cerebral infarction and those with cerebral haemorrhage.
METHODS
Participants
A total of 767 consecutive Japanese patients with cerebral infarction or cerebral haemorrhage who were admitted to Tokyo Bay Rehabilitation Hospital within 60 days of onset from March 2007 to October 2009 were enrolled in the study. Of these, 17 were excluded from the analyses because of incomplete echocardiographic findings, due to inability to follow instructions (n = 13) or insufficient basic information due to early discharge (n = 4). Data from the remaining 750 patients were analysed. The study was approved by the ethics committee of Tokyo Bay Rehabilitation Hospital.
Basic information
The following demographic and basic information about the participants were extracted from their charts: age, sex, diagnosis, time from stroke onset until admission, history of heart disease, Functional Independence Measure (FIM) score (15) on admission.
Echocardiographic parameters
Patients were assessed by transthoracic echocardiography within 7 days of admission using an Aplio SSA-700A (Toshiba Medical Systems Corp., Tokyo, Japan). One medical technologist with more than 10 years of echocardiographic experience evaluated all patients in the same manner. Heart rhythm was measured during echocardiography with a limb lead (II). Left atrial diameter was measured in the parasternal long-axis view, and interventricular septal wall thickness in the parasternal long-axis view. Asynergy was measured semi-quantitatively. Valvular lesions were recorded as mild, moderate or severe. Left ventricular (LV) ejection fraction was evaluated in the long-axis view using the Teichholz method (16), or modified Simpson’s rule (17) if asynergy was present. In patients with atrial fibrillation, mean values of LV ejection fraction during 3 consecutive heartbeats were further analysed, except when the examiner judged it difficult to measure due to a severely irregular heartbeat. Echocardiograms with ambivalent findings were reassessed by a board-certified cardiologist.
Determination of abnormal values in the echocardiographic parameters
Abnormal echocardiographic parameters were determined as follows. Data for healthy Japanese persons described by Daimon et al. (18) were used as the normal reference. Values were considered abnormal when they deviated from mean ± 2 standard deviations (SD) of the healthy values for each age group (20–29, 30–39, 40–49, 50–59, 60–69 and 70–79 years) and sex (see Appendix I for details). Because the age of healthy persons in the Daimon report ranged from 20 to 79 years, we did not have normal references for patients aged ≤ 19 (n = 1) and ≥ 80 (n = 120) years, and thus data from healthy persons aged 20–29 and 70–79 years were used as the respective references for these patients.
Only severe valvular lesions were classified as abnormal (19), because insignificant degrees of valvular regurgitation, as detected by echocardiography, are very common among elderly persons (20).
Statistical analyses
Basic values were compared between types of stroke using the χ2 test for categorical variables, unpaired t-tests for parametric variables and the Mann-Whitney U test for non-parametric variables. The prevalence of echocardiographic abnormalities was compared between stroke types using the χ2 test for simple comparisons of each parameter. The confounding effect of age was removed using the Mantel-Haenszel test. All data were analysed using SPSS version 21 (IBM Corp., Armonk, NY, USA), with p < 0.05 considered statistically significant.
RESULTS
Patient characteristics
Table I summarizes the basic information for the 750 patients. On admission, 167 (22.3%) patients had a history of heart disease, with a higher proportion in the group with cerebral infarction than in the group with cerebral haemorrhage. Patients with cerebral infarction were significantly older and had higher FIM scores at admission than those with cerebral haemorrhage. The male:female ratio and time from stroke onset until admission did not differ significantly between the 2 groups.
|
Table I. Patients’ characteristics |
||||
|
All n = 750 |
Infarction n = 448 |
Haemorrhage n = 302 |
p-value |
|
|
Age, years, mean (SD) |
67.5 (12.3) |
70.3 (11.6) |
63.3 (12.1) |
< 0.01a |
|
Female/male, n |
287/463 |
160/288 |
127/175 |
0.08b |
|
Time since stroke, days, mean (SD) |
36.7 (13.2) |
37.1 (13.2) |
36.2 (13.2) |
0.41c |
|
History of heart disease, n (%) |
167 (22.3) |
136 (30.4) |
31 (10.3) |
< 0.01b |
|
FIM score at admission, mean (SD) |
73.9 (30.0) |
76.1 (29.9) |
70.6 (30.0) |
< 0.05c |
|
aUnpaired t-test. bχ2 test. cMann–Whitney U test. FIM: functional independence measure; SD: standard deviation. |
||||
History of heart disease
Table II shows the subjects’ history of heart disease. The most frequent type of heart disease was arrhythmia, for which 104 (13.9%) patients had a history. Atrial fibrillation was the most common type of arrhythmia in 88 (84.6%) of these 104 patients, and 4 had prior paroxysmal atrial fibrillation. A history of ischaemic heart and valvular disease was found in 58 (7.7%) and 10 (1.3%) of all patients, respectively.
|
Table II. Patients’ history of heart disease |
||||
|
All n = 750 n (%) |
Infarction n = 448 n (%) |
Haemorrhage n = 302 n (%) |
pa |
|
|
Arrhythmias |
104 (13.9) |
90 (20.1) |
14 (4.6) |
< 0.01 |
|
Atrial fibrillation |
88 |
77 |
11 |
|
|
Otherb |
10 |
10 |
0 |
|
|
Uncertain |
10 |
7 |
3 |
|
|
Ischaemic heart disease |
58 (7.7) |
40 (8.9) |
18 (6.0) |
0.14 |
|
Valvular diseases |
10 (1.3) |
8 (1.8) |
2 (0.7) |
0.16 |
|
Othersc |
22 (2.9) |
21 (4.7) |
1 (0.3) |
< 0.01 |
|
Total |
167 (22.3) |
136 (30.4) |
31 (10.3) |
< 0.01 |
|
aχ2 test. bOthers: sick sinus syndrome (n = 7), atrioventricular block (n = 2), paroxysmal supraventricular tachycardia and complete right bundle branch block (n = 1 each). cOthers: heart failure (n = 17), atrial septal defect, hypercardia, hypertrophic cardiomyopathy, pericarditis and probe patency of foramen ovale (n = 1 each). |
||||
Echocardiographic abnormalities
Abnormal rhythms. A total of 103 (13.7%) patients had abnormal rhythms at the time of echocardiography (Table III). Among these patients, atrial fibrillation was the most frequent (n = 97; 94.2%), and 35 (36.1%) had not been previously diagnosed with atrial fibrillation. Sinus rhythm was present in 23 of the patients with a history of atrial fibrillation at the time of evaluation. Significantly more arrhythmias were identified in patients with cerebral infarction than in those with cerebral haemorrhage, even after adjusting for age (p < 0.01; Fig. 1).
|
Table III. Echocardiographic abnormalities |
|||||
|
All n = 750 n (%) |
Infarction n = 448 n (%) |
Haemorrhage n = 302 n (%) |
pa |
pb |
|
|
Abnormal rhythm |
103 (13.7) |
87 (19.4) |
16 (5.3) |
< 0.01 |
< 0.01 |
|
Atrial fibrillation |
97 |
81 |
16 |
< 0.01 |
< 0.01 |
|
Atrial fibrillation with pacemaker rhythm |
2 |
2 |
0 |
||
|
Pacemaker rhythm |
2 |
2 |
0 |
||
|
Junctional rhythm |
2 |
2 |
0 |
||
|
LA enlargementc |
152 (20.4) |
114 (25.6) |
38 (12.7) |
< 0.01 |
< 0.01 |
|
with atrial fibrillation |
78 |
65 |
13 |
||
|
LV hypertrophyb |
145 (19.5) |
72 (16.2) |
73 (24.3) |
< 0.01 |
0.20 |
|
Symmetrical hypertrophy |
135 |
66 |
69 |
||
|
Hypertrophic cardiomyopathyd |
11 |
7 |
4 |
||
|
Ventricular asynergye |
46 (6.1) |
34 (7.6) |
12 (4.0) |
< 0.05 |
0.22 |
|
Severe valvular diseasef |
7 (0.9) |
4 (0.9) |
3 (1.0) |
0.59 |
– |
|
Aortic insufficiency |
1 |
1 |
0 |
||
|
Aortic stenosis |
0 |
0 |
0 |
||
|
Mitral insufficiency |
5 |
2 |
3 |
||
|
Mitral stenosis |
1 |
1 |
0 |
||
|
Low left ventricular ejection fractiong |
90 (12.2) |
59 (13.6) |
31 (10.3) |
0.18 |
0.30 |
|
aχ2 test. bAdjusted for age with Mantel-Haenszel method in all age groups (except severe valvular disease due to very small numbers). Data from the following patients were excluded: 3 with infarction and 2 with haemorrhagec; 1 with infarctione; 6 with infarction and 1 with haemorrhagef; 13 with infarction and 1 with haemorrhageg because of technical difficulties with measurements. dOne patient with cerebral infarction and hypertrophic cardiomyopathy did not have left septal hypertrophy. LA: left atrial; LV: left ventricular. |
|||||
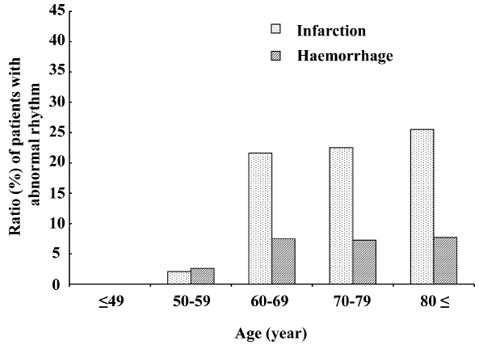
Fig. 1. Proportions of patients in each age group with cerebral infarction and cerebral haemorrhage who had abnormal rhythms. More patients with cerebral infarction aged ≥ 60 years had abnormal rhythm. Proportions differed significantly between types of stroke after Mantel-Haenszel adjustment for age (p < 0.01).
Left atrial enlargement. Overall, 152 (20.4%) patients had left atrial enlargement, 78 (51.3%) of whom had atrial fibrillation at the time of evaluation. Among the patients with cerebral infarction without atrial fibrillation at the time of evaluation, or with a history of atrial fibrillation, 11.1% had left atrial enlargement. The proportion of patients with left atrial enlargement increased with age among those with cerebral infarction, but not among those with cerebral haemorrhage (Fig. 2). The proportion of patients with left atrial enlargement after adjusting for age remained significantly higher in patients with cerebral infarction than in those with cerebral haemorrhage (p < 0.01).
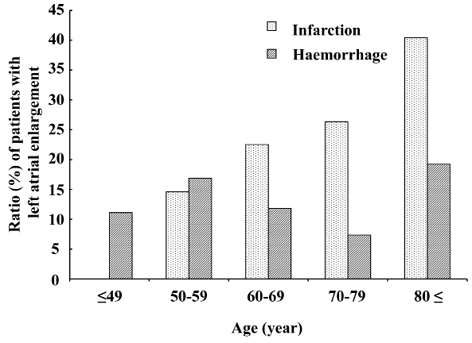
Fig. 2. Proportions of patients in each age group with cerebral infarction and cerebral haemorrhage who had left atrial enlargement. The proportion of left atrial enlargement increased with age in patients with cerebral infarction. Proportions significantly differed between types of stroke after Mantel-Haenszel adjustment for age (p < 0.01 in all age groups and in patients aged 20–79 years with normal reference from the same age group).
Interventricular septal wall thickening. Overall, interventricular septal wall thickening was found in 145 (19.5%) patients, of whom 11 had asymmetrical left ventricular hypertrophy. Increased interventricular septal wall thickness was more frequent in younger patients with both types of stroke (Fig. 3). The difference between the stroke types did not reach statistical significance after adjusting for age.
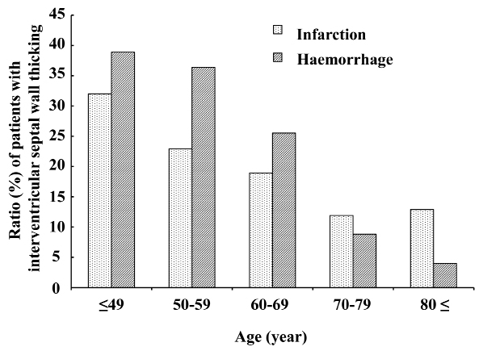
Fig. 3. Proportions of patients in each age group with cerebral infarction and cerebral haemorrhage who had increased interventricular septal wall thickness. The proportion of interventricular septal wall thickness increased more in younger groups with both types of stroke. Proportions did not significantly differ between types of stroke after Mantel-Haenszel adjustment for age (p = 0.36 in all age groups and p = 0.20 in patients aged 20–79 years, with normal reference from the same age group).
Left ventricular asynergy. Left ventricular asynergy was found in 46 (6.1%) of the 750 enrolled patients. Twenty-two (47.8%) patients with asynergy had no history of ischaemic heart disease. Among the 46 patients, 73.9% had cerebral infarction, but the difference between the stroke types did not reach statistical significance after adjusting for age (Fig. 4).
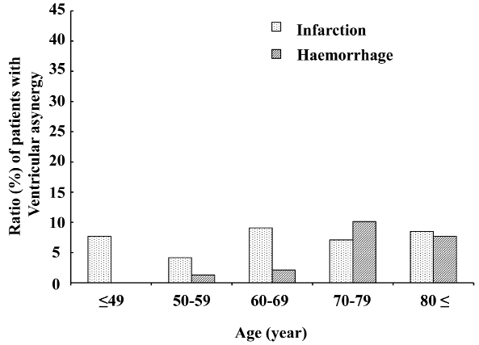
Fig. 4. Proportions of patients in each age group with cerebral infarction and cerebral haemorrhage who had ventricular asynergy. Ventricular asynergy did not seem to change with age, and did not significantly differ between types of stroke after Mantel-Haenszel adjustment for age (p = 0.22).
Valvular lesions. Valvular lesions were severe in 7 (0.9%) patients, among whom mitral insufficiency was most frequent (n = 5; 0.7%) followed by aortic insufficiency (n = 1; 0.1%) and mitral stenosis (n = 1; 0.1%).
Low left ventricular ejection fraction. Low LV ejection fraction was identified in 90 (12.2%) of the 750 patients (Fig. 5). The prevalence of low LV ejection fraction was higher (26.9%) among younger (age ≤ 49 years) patients with cerebral infarction, Overall, the prevalence of LV ejection fraction did not differ significantly between patients with cerebral infarction and those with cerebral haemorrhage.
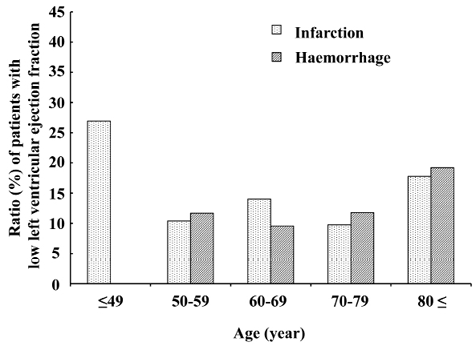
Fig. 5. Proportions of patients in each age group with cerebral infarction and cerebral haemorrhage who had low left ventricular ejection. Left ventricular (LV) ejection fraction was low in approximately 10% of patients aged 50–79 years. Although no particular trend was evident for (LV) ejection fraction with age, the prevalence of low LV ejection fraction was higher (26.9%) in patients with cerebral infarction aged ≤ 40 years. The prevalence of low LV ejection fraction was also somewhat higher in those aged ≥ 80 years. However, they were compared with healthy individuals aged 70–79 years because data for healthy persons in this age group are unavailable.
DISCUSSION
To our knowledge, this is the first study to evaluate abnormalities of echocardiographic parameters in a relatively large sample of patients with stroke admitted to a subacute rehabilitation unit and to compare these abnormalities between patients with cerebral infarction and those with cerebral haemorrhage.
Atrial fibrillation was identified in 12.9% of the patients in this study, and more frequently in those with cerebral infarction than with cerebral haemorrhage. One reason for this may be that atrial fibrillation increases the risk of cerebral infarction. In addition to its importance as a risk factor for cerebral infarction, several studies have shown that atrial fibrillation is related to functional outcomes of stroke (21–24). Karataş et al. (21) and Giaquinto et al. (22) reported that admission and discharge FIM scores were significantly lower, and that FIM gain was also lower, among stroke patients with, than without, atrial fibrillation. Lin et al. (23) and Cisternino et al. (24) reported that these differences in activities of daily living disappeared 1 year after onset or discharge from a rehabilitation centre. Further study is needed to define how atrial fibrillation influences the functional outcomes of patients with stroke.
Left atrial enlargement was identified in 20.4% of the 750 patients with stroke, and the proportion of left atrial enlargement increased with age in patients with cerebral infarction. Approximately half of the patients with left atrial enlargement had atrial fibrillation and the remainder had sinus rhythm. Left atrial enlargement was evident in 11.1% of patients with cerebral infarction who had no history of atrial fibrillation and who had sinus rhythm at the time of evaluation. These patients might have had paroxysmal atrial fibrillation. An enlarged atrium can be a consequence (25) or a cause (26) of atrial fibrillation. Psaty et al. (26) reported that the size of the left atrium was closely and independently associated with the incidence of atrial fibrillation, and that the relative risk was 4.05 for individuals with a left atrium > 5.0 cm, compared with ≤ 3.0 cm. The results of these reports and the present findings suggest that 10% of patients with cerebral infarction accompanied by sinus rhythm at the time of evaluation on admission to subacute rehabilitation units might have extant paroxysmal atrial fibrillation or will develop atrial fibrillation in the future, especially among elderly patients.
Symmetrical thickening of the interventricular septal wall was identified in 18.1% of the patients in this study, and the proportion of increased interventricular septal wall thickness increased in younger groups regardless of stroke type. Because none of the patients in this study had severe aortic stenosis, we supposed that the main cause of septal wall thickening was hypertension. Therefore our result suggests that hypertension exerts a more powerful effect in younger patients. Others have associated left ventricular hypertrophy not only with risk of stroke but also with exaggerated exercise systolic blood pressure (27, 28). Ren et al. (27) correlated LV mass with exercise systolic blood pressure or the change from resting to exercise systolic blood pressure in healthy persons and in patients with hypertension. Lim et al. (28) suggested that LV wall thickness and mass correlate with systolic blood pressure during submaximal exercise (4.7 metabolic equivalents (METS) and 7.1 METS) in patients with hypertension. Lauer et al. (29) reported that the prevalence of LV hypertrophy was higher in healthy individuals with an exaggerated than with a normal systolic blood pressure response, but the difference did not reach statistical significance after adjusting for age, resting systolic blood pressure and body mass index. Although the relationship between LV hypertrophy and exaggerated exercise systolic blood pressure remains to be explored, the possibility that patients with stroke have exaggerated exercise systolic blood pressure should be considered when prescribing rehabilitation programmes.
The presence of LV asynergy indicates a high likelihood of prior ischaemic heart disease. However, 47.8% of stroke patients with asynergy in our study had never been diagnosed with ischaemic heart disease. Di Pasquale et al. (30) also reported that the maximal exercise treadmill test was positive in 26% of patients with cerebral ischaemia, but without symptoms or electrocardiographic signs of ischaemic heart disease. Their results and the present findings suggest that a medical history, physical findings and electrocardiography are insufficient to rule out current or prior ischaemic heart disease. Information about ischaemic heart disease is important for more accurate predictions of stroke rehabilitation outcomes, as well as for preventing recurrences of both ischaemic heart disease and ischaemic stroke. Roth et al. (31) found that functions improved significantly less among patients with thrombotic or embolic stroke accompanied by coronary artery disease and congestive heart failure than in those without coronary artery disease. They also showed that performance, in terms of rolling, moving in bed, transferring from a wheelchair to a bed and walking, was lowest among patients with coronary artery disease.
Although valvular lesions themselves do not increase the risk of stroke recurrence in patients with ischaemic stroke (32), they are important factors that influence the outcome of stroke rehabilitation (6). However, very few of our patients had severe valvular lesions.
LV ejection fraction was low in approximately 10% of all patients aged 50–79 years and in approximately 20% of patients with cerebral infarction aged ≤ 40 years. Low LV ejection fraction was independently associated with an increased risk of ischaemic stroke (33, 34). Although cardiac load is not high at comfortable walking speed compared with normal individuals (35), a low LV ejection fraction should be considered when more strenuous rehabilitation programmes are prescribed in order to achieve further functional gain. In addition, LV ejection fraction is an important predictor of stroke rehabilitation outcomes. Kevorkian et al. (36) reported lower discharge FIM scores, FIM gains and FIM efficiency in patients with stroke and low LV ejection fractions than in those with high LV ejection fractions.
Several studies have evaluated cardiac function in patients with stroke during the acute phase in Japan (37) and elsewhere (38, 39). Wolber et al. (39) assessed 775 patients with acute ischaemic stroke using echocardiography and found left atrial dilation in 30%, LV hypertrophy in 23%, atrial fibrillation in 7%, low (< 45%) LV ejection fraction in 5%, aortic stenosis in 1%, aortic regurgitation in 0.9%, mitral regurgitation in 0.9%, silent myocardial infarction in 0.3%, and none with mitral stenosis. Our findings tended to be similar to theirs in terms of a high prevalence of left atrial dilation and LV hypertrophy and a low prevalence of valvular lesions, but differed in other respects. We suppose that these were due to differences in race, time from stroke onset and age. The proportion of patients aged ≤ 49 years in the study by Wolber and in the present study were 18.1% and 8.3%, respectively.
The present study has several limitations. First, the study was conducted at a single institution and thus the patients might not accurately reflect the entire stroke population in subacute rehabilitation units. Secondly, we used data from healthy individuals aged 70–79 years as a reference for patients aged ≥ 80 years because data for healthy Japanese subjects in this age group are unavailable. This might have led to overestimations of echocardiographic abnormalities. Finally, we used simple echocardiography in this study because it is convenient and non-invasive. However, cardiac function under exercise stress cannot be assessed with simple echocardiography alone; this requires dobutamine stress echocardiography or exercise echocardiography.
Despite these limitations, we believe that our findings offer important and useful information for stroke rehabilitation management. Patients with stroke in the subacute rehabilitation phase have a specific prevalence of cardiac abnormalities, irrespective of a history of heart disease such as left atrial enlargement in elderly patients and increased LV wall thickness and low ejection fraction in younger patients. These should be considered when prescribing rehabilitation programmes.
ACKNOWLEDGEMENTS
The authors would like to thank Chika Kawashima, MT, for conducting the echocardiographic studies.
APPENDIX
Appendix I. Cut-off values used to determine abnormal echocardiographic parameters in each age group for each sex | ||||||||
Age group | ||||||||
≤ 19 years | 20–29 years | 30–39 years | 40–49 years | 50–59 years | 60–69 years | 70–79 years | ≥ 80 years | |
Males | ||||||||
LA diameter (cm)a in parasternal long-axis view | 3.9d | 3.9 | 4.0 | 4.3 | 4.1 | 4.1 | 4.5 | 4.5d |
Interventricular septal wall thickness (cm)b | 1.0d | 1.0 | 1.0 | 1.0 | 1.1 | 1.1 | 1.2 | 1.2d |
LV ejection fraction (%)c | 56d | 56 | 54 | 55 | 52 | 56 | 54 | 54d |
Females | ||||||||
LA diameter (cm)a in parasternal long-axis view | 3.5d | 3.5 | 3.6 | 3.8 | 3.9 | 4.1 | 4.2 | 4.2d |
Interventricular septal wall thickness (cm)b | 0.9d | 0.9 | 0.9 | 1.0 | 1.0 | 1.0 | 1.1 | 1.1d |
LV ejection fraction (%)c | 56d | 56 | 57 | 56 | 56 | 53 | 59 | 59d |
Cut-off values determined as mean +2 SD of values from healthy persons in each age group (18) for aleft atrial (LA) enlargement and bincreased interventricular septal wall thickness, and mean –2SD for clow left ventricular (LV) ejection fraction. dBecause the age of healthy persons in Daimon’s report (18) ranged from 20 to 79 years, normal references for patients aged ≤ 19 years (n = 1) and ≥ 80 years (n = 120) years were unavailable. Data from healthy persons aged 20–29 years and 70–79 years, respectively, were therefore used for these comparisons. | ||||||||
REFERENCES
