Cintia Laura Pereira de Araujo, MSc1,2, Manuela Karloh, MSc1,3, Cardine Martins dos Reis, MSc1,2, Marina Palú, PT1,2 and Anamaria Fleig Mayer, PhD1,2
From the 1Center of Assistance, Education and Research in Pulmonary Rehabilitation, 2Graduate Program in Physiotherapy and 3Graduate Program in Human Movement Science, Santa Catarina State University (UDESC), Florianópolis, Brazil
BACKGROUND: Dynamic hyperinflation leads to dyspnoea and consequent limitations in functional capacity in chronic obstructive pulmonary disease. It has been shown that the response to pursed-lips breathing in terms of dynamic hyperinflation and lower-limb exercise capacity is variable, and its effects on activities of daily living are unknown. This study aimed to evaluate the effect of pursed-lips breathing on dynamic hyperinflation and functional capacity in patients with chronic obstructive pulmonary disease in a lower-limb exercise test and in a multiple-task activities of daily living test.
DESIGN: Randomized cross-over study.
Patients: Twenty-five patients with chronic obstructive pulmonary disease (16 men, mean age 64 ± 7 years, forced expiratory volume in 1 s (FEV1) = 41.7 ± 14.7% predicted).
METHODS: Patients randomly performed two 6-min walk tests (6MWT) with and without pursed-lips breathing (6MWTPLB and 6MWTNon-PLB) and two Glittre-ADL tests with and without pursed-lips breathing (TGlittrePLB and TGlittreNon-PLB). Inspiratory capacity was assessed at baseline and immediately after the tests.
RESULTS: The 6MWTNon-PLB and TGlittreNon-PLB induced similar magnitude dynamic hyperinflation (0.22 ± 0.24 l and 0.31 ± 0.23 l, respectively; p > 0.05). Pursed-lips breathing did not improve dynamic hyperinflation induced by the 6MWT (0.24 ± 0.20 and 0.22 ± 0.24 l, respectively, with and without pursed-lips breathing; p > 0.05). Dynamic hyperinflation in the TGlittrePLB was significantly lower than in the TGlittreNon-PLB (0.19 ± 0.20 l and 0.31 ± 0.23 l, respectively; p = 0.02). Pursed-lips breathing did not improve 6MWT or TGlittre performance.
CONCLUSION: Pursed-lips breathing reduced dynamic hyperinflation in the TGlittre, but not in the 6MWT. However, pursed-lips breathing did not improve functional capacity.
Key words: inspiratory capacity; positive-pressure respiration; exercise; activities of daily living; COPD.
J Rehabil Med 2015; 47: 957–962
Correspondence address: Anamaria F. Mayer, Departamento de Fisioterapia, Centro de Ciências da Saúde e do Esporte (CEFID), Universidade do Estado de Santa Catarina (UDESC), Rua Paschoal Simone, 358, CEP 88080-350, Florianópolis, SC, Brazil. E-mail: anamaria.mayer@udesc.br
Accepted Jun 22, 2015; Epub ahead of print Nov 4, 2015
INTRODUCTION
Dynamic hyperinflation (DH) plays an important role in effort limitation in patients with moderate-to-severe chronic obstructive pulmonary disease (COPD) (1, 2). This phenomenon is associated with expiratory flow limitation, shorter expiratory time, and pulmonary air trapping, leading to respiratory discomfort during exercise or activities of daily living (ADL) (2, 3). Studies have shown that ventilatory requirements imposed by different exercise test protocols cause DH (2–5). In addition, patients who develop DH during incremental exercise testing on the cycle-ergometer perform less daily physical activity, as measured using a triaxial accelerometer (2).
Regarding DH prevention, pursed-lips breathing (PLB) is widely used in pulmonary rehabilitation programmes in order to impose a positive expiratory pressure that aims to create resistance to expiratory flow, preventing early bronchial collapse (6, 7).
Although the 6-min walk test (6MWT) reflects limitations in performance of ADL (8), it does not evaluate the influence of upper limb work or activities other than walking on ADL limitation. It seems that upper limb activities play an important role in developing DH (3, 9, 10). However, the effect of PLB in avoiding DH (11, 12) and improving effort tolerance has only been studied during single-task lower-limb exercise tests (12–15), which are less representative of ADL (16).
In this context, the purpose of this study is to investigate the acute effect of volitional PLB on DH in patients with COPD in a lower-limb exercise test and in a multiple-task ADL test and, as secondary aim, its effect on functional capacity.
METHODS
Subjects
Twenty-five patients with COPD were included in the study. None of the subjects had been engaged in any exercise-training programme at least one year before participating in the study. Patients were recruited from pulmonology outpatient services of public hospitals and private clinics. Inclusion criteria were: Global Initiative for Obstructive Lung Disease (GOLD) stages 2, 3 and 4 of severity of airflow limitation (17) with a history of smoking ≥ 20 pack-years, clinical stability in the 4 weeks prior to the study protocol and age ≥ 40 years. Exclusion criteria were: long-term oxygen therapy, current smoking, any pulmonary disease other than COPD, and comorbidities that would compromise ability to perform any of the evaluations in the study. From the variability of inspiratory capacity (IC) measurements obtained in a pilot study, a reduction in DH of 0.15 (0.23) l was expected to result from PLB on the basis of a power analysis for 25 subjects (α = 0.05 and β = 0.10 power). The study was approved by the Human Research Ethics Committee of the State University of Santa Catarina (protocol number 223/2011) and all participants signed an informed consent. This study was registered at ClinicalTrials.gov (Identifier NCT02203058).
Protocol
Subjects were assessed in a 3-day cross-over design. Anthropometric and lung function assessments were carried out for all subjects. On the same day, patients underwent a familiarization trial for both the 6MWT and the TGlittre and for PLB. During the familiarization it was observed whether the subjects performed PLB spontaneously. Also, they were asked if they routinely used PLB in their daily life. The respiratory frequency and the tidal volume were not controlled during the tests. On 2 separate days, 2 of the tests were randomly performed: 6MWT without PLB (6MWTNon-PLB), 6MWT with PLB (6MWTPLB), TGlittre without PLB (TGlittreNon-PLB), and TGlittre with PLB (TGlittrePLB). To avoid the learning effects, 2 6MWTs or 2 TGlittres were not permitted on the same day. Subjects were instructed on how to apply PLB according to standard guidelines (18) and were encouraged to perform PLB throughout the 6MWTPLB and TGlittrePLB. To inhibit PBL during the 6MWTNon-PLB and TGlittreNon-PLB, the subjects used a mouthpiece (2 cm diameter × 2 cm length) such as the one used in pulmonary function tests. It was not possible to blind the patients and the evaluators to the study protocol.
Pulmonary function testing. Lung function was tested using an EasyOne spirometer (NDD Medical Technologies Inc., Zurich, Switzerland), and calibration was checked before each evaluation. Spirometry was performed in accordance with American Thoracic Society (ATS)/European Respiratory Society (ERS) standards (19). The predicted values were calculated from the equations proposed by Pereira et al. (20).
Inspiratory capacity (IC) measurement. IC was measured with the patient in the sitting position, using an EasyOne portable spirometer, before and immediately after the tests, by the slow vital capacity manoeuvre starting from a stable end-expiratory volume, in accordance with ATS/ERS standards (19). IC was measured in the exact place where the patients finished. A minimum of 3 manoeuvres (maximum of 8) was performed and, in order to be considered reproducible, 2 curves could not vary by more than 5% or 150 ml (4, 21, 22). The higher value of 2 reproducible curves was used for analysis. DH, the primary outcome, was considered when the IC decreased 10% and/or 150 ml or more compared with the basal value (22). The measurements of IC took an average of 2 min 27 s; therefore, patients had no time to recover from DH (23).
Six-minute walk test. The 6MWT was performed according to the guidelines of the American Thoracic Society (ATS) (24). Pulse oxyhaemoglobin saturation (SpO2; Oxi-Go, Oximeter Plus, Roslyn Heights, NY, USA) and dyspnoea (Borg CR10 scale) (25) were measured at the beginning and at end of the test. Predicted values for walking distance were calculated according to Iwama et al. (26) and performance improvement cut-off was set as an increase of 25 m or more in the distance walked (27).
Glittre-ADL test. The TGlittre consists of completing a circuit, as follows: from a sitting position, the subject stands up and walks along a flat 10-m long course, in the middle of which there is a 2-step ladder (each step 17 cm high × 27 cm deep) to be climbed; after completing the 10 m, the subject faces a shelf containing 3 1-kg objects positioned on the top shelf (shoulder height) and moves them one by one to the bottom shelf (waist height) and then to the floor; the objects are then returned to the bottom shelf and, finally, to the top shelf again; the subject walks back, climbing up and down the steps, until reaching the starting point (chair), sits down and immediately begins the next lap. The subjects carried a weighted backpack (2.5 kg for women, 5.0 kg for men) and were instructed to complete 5 laps on this circuit as quickly as possible (9). SpO2 and dyspnoea index (25) were measured at the beginning of the test, at the end of each lap, and at the end of the test. Since the minimal important difference for TGlittre remains unclear, a reduction of 0.89 min in performing the test was used as an indication of performance improvement based on the responsiveness value found by Skumlien et al. (9).
Statistical analysis
Normal distribution was checked with the Shapiro–Wilk test. Data were reported as mean (SD) or median (inter-quartile range; IQR). The paired Student t-test or the Wilcoxon test was used to compare data, with and without PLB, between the 6MWT and TGlittre, and the percentage of difference in performance and DH [(PLB - Non-PLB)∕Non-PLB]∙100) between the 6MWT and TGlittre. The Pearson or Spearman’s correlation test was performed to verify the association between DH, test performance, and dyspnoea index in the 6MWT and TGlittre. The comparison between subjects who did and did not perform PLB spontaneously was assessed using Student’s t-test for independent samples or the Mann-Whitney test. Associations between qualitative variables, such as spontaneous and non-spontaneous use of PLB, reduction in DH, and development of DH with PLB, were verified using the χ2 test. Data analysis was performed with SPSS 20.0 (SPSS Inc., Chicago, IL, USA) and the graphs were made with GraphPad Prism 5 (GraphPad Software Inc., La Jolla, CA, USA). Statistical significance was set at p < 0.05.
RESULTS
From a total of 32 patients with COPD, 25 were considered eligible and were included in the study between March 2012 and March 2013. The reasons for ineligibility were: bronchiectasis (n = 2), abdominal hernia (n = 1), knee arthrosis (n = 1), recent exacerbation (n = 1), GOLD stage 1 (n = 1), and one patient declined to participate. The anthropometric and spirometric characteristics of the 25 patients are shown in Table I.
|
Table I. Characteristics of the study group |
|
|
Characteristics |
|
|
Age, years, mean (SD) [95% CI] |
64 (7) [61–66] |
|
Male (female), n |
16 (9) |
|
Spontaneous PLB, n |
11 |
|
Smoking history, pack-years, mean (SD) [95%] |
57.8 (34.3) [44.5–71.2] |
|
GOLD 2, n |
8 |
|
GOLD 3, n |
12 |
|
GOLD 4, n |
5 |
|
mMRC*, median [IQR] |
1 [1–2] |
|
BMI, kg/m2, mean (SD) [95% CI |
27.6 (5.13) [25.5–29.7] |
|
FEV1/FVC, %, mean (SD) [95% CI] |
0.47 (0.10) [0.43–0.51] |
|
FEV1, l, mean (SD) [95% CI] |
1.26 (0.51) [1.05–1.47] |
|
FEV1, %pred, mean (SD) [95% CI] |
41.7 (14.7) [35.6–47.8] |
|
FVC, l, mean (SD) [95% CI] |
2.62 (0.74) [2.31–2.93] |
|
FVC, %pred, mean (SD) [95% CI] |
69.0 (16.5) [62.3–75.8] |
|
GOLD: Global Initiative for Obstructive Lung Disease; 95% CI: 95% confidence interval; PLB: pursed-lips breathing; spontaneous PLB: patients who spontaneously performed PLB during familiarization tests; mMRC: modified Medical Research Council scale (37); BMI: body mass index; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; %pred: percentage of predicted value; IQR: interquartile range; 95% CI: 95% confidence interval. |
|
PLB and dynamic hyperinflation
Baseline IC was similar between PLB and Non-PLB tests and between the 6MWT and TGlittre. All subjects developed DH in at least one test (6MWTNon-PLB, 6MWTPLB,TGlittreNon-PLB or TGlittrePLB). DH induced by the 6MWTNon-PLB and TGlittreNon-PLB were not statistically different (mean difference: 0.08 ± 0.29 l; p = 0.16) and no significant difference between DH induced by the 6MWTNon-PLB and 6MWTPLB was found. The DH in the TGlittrePLB was significantly lower than in the TGlittreNon-PLB (Table II; Fig. 1). Although PLB did not affect DH in the 6MWT, the percentage of difference in DH correlated positively between the 6MWT and TGlittre (r = 0.63; p = 0.001), indicating a similar behaviour in the PLB effect on DH.
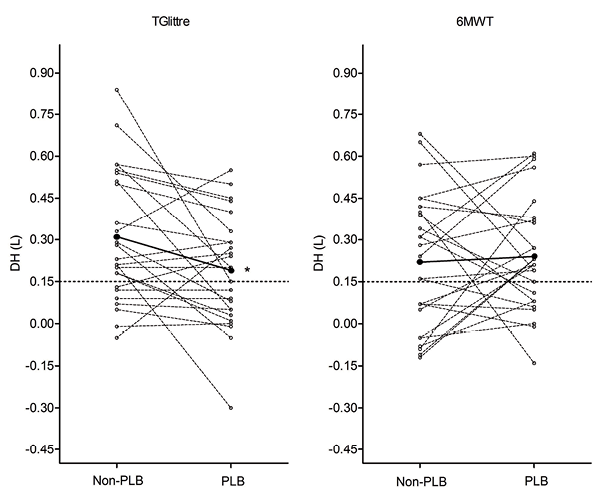
PLB and functional capacity
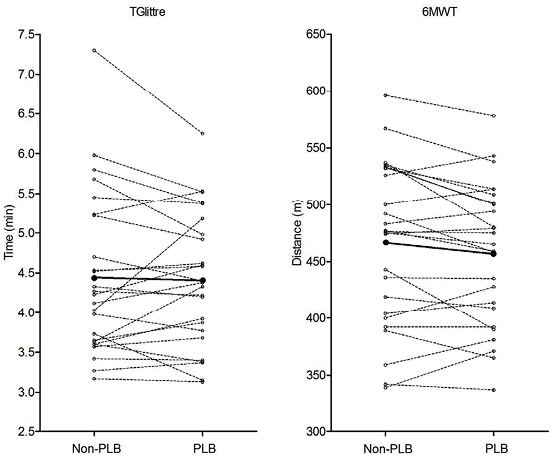
The performances in the 6MWT and TGlittre with and without PLB are described in Fig. 2 and Table II. No significant difference between tests with and without PLB was found. Two subjects achieved performance improvement cut-off in the 6MWTPLB and one subject in the TGlittrePLB.
Functional capacity and dynamic hyperinflation
Performances in the 6MWTNon-PLB, 6MWTPLB, TGlittreNon-PLB or TGlittrePLB and their respective DH did not show significant correlation (p > 0.05). DH between the TGlittreNon-PLB and TGlittrePLB correlated moderately (r = 0.44; p = 0.03), while a non-significant correlation was found in DH between the 6MWTNon-PLB and 6MWTPLB (p > 0.05). The change in performance and the change in DH in both tests did not show significant correlation (p > 0.05).
DH, dyspnoea and pulmonary function
PLB did not improve breathlessness at the end of the tests (Table II). However, the dyspnoea index correlated positively with DH in the TGlittrePLB (r = 0.43; p = 0.03). No correlations were found between the absolute change (Non-PLB minus PLB) in dyspnoea and the absolute change in DH in the 6MWT or TGlittre. Pulmonary function (FEV1 and FEV1/FVC) did not correlate significantly (p > 0.05) with DH or its changes in the tests.
|
Table II. Comparison between functional capacity tests and dynamic hyperinflation |
|||||||
|
Non-PLB |
PLB |
Mean difference |
p |
||||
|
Mean (SD) |
95% CI |
Mean (SD) |
95% CI |
||||
|
6MWT, m |
466 (71.5) |
437–496 |
457 (62.8) |
431–483 |
9.24 (23.8) |
0.06 |
|
|
6MWT, %pred |
86.4 (11.7) |
81.6–91.2 |
84.8 (11.0) |
80.3–89.3 |
|||
|
Baseline IC, l |
2.39 (0.75) |
2.08–2.69 |
2.42 (0.69) |
2.14–2.71 |
0.43 |
||
|
DH, l |
0.22 (0.24) |
0.12–0.32 |
0.24 (0.20) |
0.16–0.33 |
–0.02 (0.27) |
0.71 |
|
|
Dyspnoeaa |
3 |
0.75–4.0 |
3 |
1.0–4.0 |
–0.06 (–2; +3.5) |
0.65 |
|
|
TGlittre, mina |
4.23 |
3.62–5.23 |
4.38 |
3.73–6.08 |
0.04 (0.45) |
0.67 |
|
|
Baseline IC, l |
2.46 (0.74) |
2.15–2.76 |
2.41 (0.71) |
2.12–2.71 |
0.32 |
||
|
DH, l |
0.31 (0.23) |
0.21–0.40 |
0.19 (0.20) |
0.13–0.27 |
0.12 (0.23) |
0.02 |
|
|
Dyspnoeaa |
3 |
1.0–3.5 |
3 |
1.5–4.0 |
–0.2 (–3; +2) |
0.44 |
|
|
amedian (IQR 25th–75th) PLB: pursed-lips breathing; Non-PLB: test performed without PLB; PLB: test performed with PLB; 95% CI: 95% confidence interval; SD: standard deviation; DH: dynamic hyperinflation; Mean difference: Non-PLB minus PLB; 6MWT: 6-min walk test; %pred: percentage of the predicted value; Baseline IC: inspiratory capacity at baseline; DH: change in IC (baseline minus final values); Dyspnoea: dyspnoea measured at the end of the test by Borg CR10 scale; TGlittre: Glittre ADL-test; p: significance level; p: comparison between Non-PLB and PLB. No significant differences in baseline IC, DH, and dyspnoea were found between the 6MWT and TGlittre with and without PLB. |
|||||||
PLB spontaneous and non-spontaneous
No significant associations were found between spontaneous and non-spontaneous use of PLB, reduction in DH, and non-development of DH with PLB (p > 0.05) (Table III).
|
Table III. Comparison between subjects who performed spontaneous and non-spontaneous pursed-lips breathing (PLB) |
||||
|
Non-spontaneous PLB (n = 14) Mean (SD) |
Spontaneous PLB (n = 11) Mean (SD) |
Mean difference (SE) |
p |
|
|
FEV1/FVC, % |
0.49 (0.10) |
0.44 (0.10) |
0.05 (0.04) |
0.19 |
|
FEV1, l |
1.32 (0.52) |
1.18 (0.51) |
0.14 (0.21) |
0.50 |
|
FEV1, %pred |
40.9 (14.9) |
42.7 (15.2) |
–1.87 (6.04) |
0.76 |
|
FVC, l |
2.60 (0.75) |
2.64 (0.77) |
–0.04 (0.31) |
0.90 |
|
FVC, %pred |
63.5 (14.4) |
76.0 (17.0) |
–12.5 (6.29) |
0.06 |
|
6MWT |
||||
|
6MWTPLB, m |
461.8 (71) |
451.3 (53) |
10.51 (25.75) |
0.69 |
|
Baseline IC, l |
2.46 (0,80) |
2.39 (0.57) |
0.07 (0.28) |
0.80 |
|
DH, l |
0.21 (0.21) |
0.29 (0.20) |
–0.09 (0.08) |
0.31 |
|
6MWTNon-PLB, m |
475.7 (76) |
454.5 (67) |
21.17 (29.1) |
0.47 |
|
Baseline IC, l |
2.36 (0.84) |
2.42 (0.65) |
–0.06 (0.31) |
0.84 |
|
DH, l |
0.24 (0.23) |
0.20 (0.27) |
0.04 (0.10) |
0.68 |
|
∆6MWT, % |
–2.72 (5.13) |
–0.19 (5.24) |
–2.53 (2.08) |
0.24 |
|
∆DH 6MWT, l |
0.04 (0.25) |
–0.09 (0.29) |
0.13 (0.11) |
0.25 |
|
TGlittre |
||||
|
TGlittrePLB, min |
4.34 (0.87) |
4.59 (0.84) |
–0.16 (0.35) |
0.66 |
|
Baseline IC, l |
2.45 (0.80) |
2.36 (0.60) |
0.07 (0.29) |
0.81 |
|
DH, l |
0.16 (0.16) |
0.22 (0.24) |
–0.06 (0.08) |
0.46 |
|
TGlittreNon-PLB, min |
4.0 (3.59–4.83)a |
4.5 (0.28) |
4.2 (3.6–5.2)a |
0.41b |
|
Baseline IC, l |
2.49 (0.80) |
2.42 (0.69) |
0.07 (0.30) |
0.82 |
|
DH, l |
0.27 (0.22) |
0.36 (0.25) |
–0.09 (0.09) |
0.34 |
|
∆TGlittre, % |
0.31 (–6.11–3.94)a |
–0.22 (3.00) |
–1.05 (–6.2–5.83)a |
0.87b |
|
∆DH TGlittre, l |
0.11 (0.16) |
0.14 (0.31) |
–0.03 (0.10) |
0.76 |
|
aMedian (interquartile range 25th–75th); p: comparison between non-spontaneous PLB and spontaneous PLB. Mean difference: mean difference between subjects who performed spontaneous and non-spontaneous PLB (independent Student’s t-test and bMann-Whitney test); PLB: pursed-lips breathing; DH: dynamic hyperinflation; SE: standard error; SD; standard deviation; FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; %pred: percentage of predicted value; Non-PLB: test performed without PLB; PLB: test performed with PLB; 6MWT: 6-min walk test; TGlittre: Glittre ADL-test; Baseline IC: inspiratory capacity at baseline; DH: dynamic hyperinflation; ∆6MWT, %: percentage of difference in 6MWT performance ([(PLB – Non-PLB)∕Non-PLB]∙100); ∆DH 6MWT, %: percentage of difference in DH induced by the 6MWT; ∆DH 6MWT, l: mean absolute change in DH induced by the 6MWT (Non-PLB – PLB); ∆TGlittre, %: percentage of difference in TGlittre performance ([(PLB – Non-PLB)∕Non-PLB]∙100); ∆DH TGlittre, %: percentage of difference in DH induced by the TGlittre; ∆DH TGlittre, l: mean absolute change in DH induced by the TGlittre (Non-PLB – PLB). |
||||
DISCUSSION
This study showed that PLB reduces DH induced by the TGlittre, while it does not affect DH induced by the 6MWT in a group of patients with moderate to very severe COPD. Besides, PLB did not affect the performance in either test.
Previous studies have reported that the 6MWT leads to similar DH to that found in the current study (5, 28) but, as far as we know, the magnitude of DH induced by a specific multiple-task test using activities besides walking, such as the TGlittre, has not been studied.
Porto et al. (3) have shown that patients with COPD hyperinflate more in upper-limb than in lower-limb exercise, with higher minute ventilation at the same metabolic demand. Hannink et al. (10) compared DH in upper- and lower-limbs tests at steady-state isoventilation and found it was not statistically different in patients with COPD. Recently, we have demonstrated a slightly higher oxygen uptake in the TGlittre, but also similar ventilatory and cardiovascular responses compared with the 6MWT (29).
Although the TGlittre includes unsupported upper-limb activities, in the current study this test did not lead to higher magnitude of DH in patients with COPD, possibly due to its similar ventilatory response to the 6MWT.
Another mechanism that may be related to DH in patients with COPD is dyssynchronous thoracoabdominal breathing, observed during activities performed with unsupported arms (30, 31), but not during unloaded leg cycling (30). These arm activities are required during TGlittre, which could lead to the DH. In addition, TGlittre requires other activities, such as bending the body, squatting, and weight carrying, which could also aggravate the dyssynchrony. Thus, the effect of PLB on DH induced by the TGlittre and not by the 6MWT may be explained by attenuation in the dyssynchronous thoracoabdominal breathing pattern.
Similarly, Spahija et al. (11) did not find reduction in end-expiratory lung volume, signalling DH, during constant-work-rate leg cycling with PLB. Meanwhile, more recently, Cabral et al. (12) showed that PLB reduced DH and increased exercise tolerance during high-intensity endurance leg cycling test, but only in COPD patients with reduced peak expiratory flow (40.2 ± 8.6% predicted), whom the authors considered as “improvers”.
Bianchi et al. (32) found that patients with more severe air flow obstruction and higher degree of static hyperinflation deflated during PLB at rest, although in our study no significant correlation was found between COPD severity and DH or the change in DH with and without PLB (p > 0.05). In addition to the significant mean reduction in DH induced by the TGlittrePLB, more patients responded positively to PLB (responders), while some patients presented an increase in DH induced by the 6MWTPLB (Fig. 1). However, further investigation is needed in this regard.
The supposed attenuation of dyssynchronous thoracoabdominal breathing may explain the effect of PLB on DH, but not the lack of improvement in exercise or ADL performance and dyspnoea during these activities. A beta error may have occurred for the functional capacity outcomes, since it was the study’s secondary aim and a sample size calculation was not performed for these outcomes.
Previous studies disagree on the effect of PLB on exercise performance. The conflicting findings among studies (12–15) on the effect of PLB on exercise performance could be explained by the way in which chest wall muscles adapt to the change in lung mechanics produced by DH (33). Patients with stable COPD who develop DH during a lower-limb exercise test seem to have lower expiratory muscle recruitment and respiratory work during a lower-limb exercise test (34) and better exercise performance in comparison with patients who do not develop DH (34, 35). It is also important to state that another reason for the inconsistent finding could be the different methodologies used in each study, such as exercise protocols and PLB spontaneity.
For future studies, subjects who present DH should be selected or analysed separately from those without DH. Based on conflicting studies, it is relevant to question how much it is necessary to decrease DH so that it can be perceived by the patient and have a clinical impact. Another interesting feature for investigation in further studies could be the reversibility of DH during recovery time.
This study has some limitations. Although the precise evaluation of DH requires the measurement of total lung capacity and of the ratio of functional residual capacity, in the current study DH was measured by the change in the IC using the slow vital capacity manoeuvre. In addition to its low cost, this method reflects dynamic residual volume alterations in a reproducible and accurate way (21, 36).
In summary, PLB decreases DH in the TGlittre, but not in the 6MWT in stable patients with moderate to very-severe COPD. Further studies are necessary to fully understand the mechanisms of DH improvement during PLB, the impact of DH reduction on exercise and ADL performance, and which patients might benefit from this breathing strategy.
ACKNOWLEDGEMENTS
The authors would like to thank those who supported the data acquisition and to all the pulmonologists who referred their patients to the study protocol, and the Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior–CAPES.
Financial support: This research was supported in part by the Coordenação de Aperfeiçoamento de Pessoal de Ensino Superior–CAPES, Brazil.
The authors declare no conflicts of interest.
REFERENCES
