Jasper T. Yen, PhD1,2 and Sheng Li, MD, PhD1,2
From the 1Department of Physical Medicine and Rehabilitation, The University of Texas Health Science Center – Houston, Houston, TX and 2NeuroRehabilitation Research Laboratory, TIRR Memorial Hermann Research Center, Houston, TX, USA
OBJECTIVE: To investigate the effect of spasticity and involuntary synergistic activation on force perception during voluntary activation of spastic paretic muscles.
METHODS: Eleven stroke subjects with spastic hemiparesis performed various isometric elbow-flexion force-matching tasks. Subjects were instructed to generate a target reference force with visual feedback using one arm (impaired or non-impaired) and then to produce a force with the other arm to match the magnitude of the reference force without visual feedback. The reference arm was at rest in unilateral exertion trials and maintained contraction in bilateral exertion trials during the matching force-production period.
RESULTS: Both force and effort mismatches occurred in most conditions, and there were asymmetries in force perception. When the non-impaired arm was the matching arm, effort and force overestimation occurred, but effort was matched better than force. When the impaired arm was the matching arm, force underestimation and effort overestimation occurred, but force was matched better than effort. No difference in matching performance was found between unilateral and bilateral exertion-matching tasks.
CONCLUSION: Overall, both force and effort misperceptions occur in stroke survivors with spasticity. Spasticity and spastic synergistic activation probably contribute to force and effort misperception during voluntary activation in chronic stroke.
Key words: spasticity; stroke; force perception.
J Rehabil Med 2015; 47: 917–923
Correspondence address: Sheng Li, University of Texas Health Science Center – Houston, 77030 Houston, TX, USA. E-mail: sheng.li@uth.tmc.edu
Accepted Aug 20, 2015; Epub ahead of print Nov 9, 2015
INTRODUCTION
Spasticity and weakness on the impaired side are common clinical presentations after stroke (1–4). Stroke survivors with spastic hemiparesis often have flexion synergy and a stereotyped posture in the upper limb with various degrees of combination of shoulder adduction and internal rotation, elbow flexion, forearm pronation and wrist/finger flexion.
Spasticity is traditionally assessed at rest using different clinical scales. Its role during volitional activation of spastic muscles is not fully understood. During volitional activation of spastic muscles in one part of the limb, muscle activation is also seen in other muscles of the limb, i.e. spastic synergistic activation. For example, shoulder abduction causes involuntary activation of distal wrist and finger flexors much more in the impaired limb than in the non-impaired limb and control limb (4). Synergistic activation may be related to bulbospinal activation as a result of disinhibition following cortical damage in stroke (4, 5). This divergent bulbospinal activation and resultant spontaneous motor unit activity are viewed as the primary underlying mechanism mediating post-stroke spasticity and related synergistic activation (6). Spontaneous motor unit discharges from resting spastic muscles have been observed in stroke survivors (2, 7, 8). The firing rate of these spontaneous motor units increases with the level of force during voluntary activation, e.g. spontaneous motor unit activities remain high after voluntary activation of the spastic muscle is stopped (2, 7). Activation of motor unit determines perception of force (9). Therefore, this spasticity-related “involuntary” activation of the intended spastic muscles and its synergistic muscles could be misperceived as voluntary force of the spastic muscles. Misperception of such “involuntary” activation is likely to be examined in a conventional force-matching paradigm.
Force perception is commonly studied in a contralateral force-matching paradigm. In this paradigm, subjects usually produce a reference force with visual feedback in 1 limb and then reproduce another force in the contralateral limb without visual feedback to match the magnitude of the reference force. In such a paradigm (10–22), the effort of the reference or matching force is defined as a percentage of its maximum strength. If there is no difference between efforts of the reference and matching forces, force perception relies mainly on “sense of effort”, efferent copy of the descending motor commands. In contrast, force perception relies on “sense of force” if absolute force magnitudes are matched between reference and matching forces, primarily based on sensory feedback from peripheral cutaneous receptors, muscle spindles and tendon organs. In neurologically intact subjects, it is reported that force perception relies primarily on the “sense of effort” when the strength of 1 muscle is altered via fatigue (11, 23), partial curarization (13), or changes in muscle length (10). Similar observations have been reported in hemiparetic stroke survivors (14, 24). In both studies, hemiparetic subjects generated a reference force with the impaired limb, and it was found that effort was matched by the non-impaired limb. However, stroke survivors with spastic hypertonia were excluded from participation in the previous study (24). Whether spasticity and related “involuntary” activation affect force perception remains unknown. Accordingly, we aimed to examine force perception in stroke survivors with mild to moderate spasticity using a contralateral force-matching paradigm. As mentioned above, activation of spontaneous motor units of the intended spastic muscle and involuntary activation of other synergistic spastic muscles may be misperceived as voluntary force in the impaired limb. No such involuntary activation is presumed in the non-impaired limb. As such, we hypothesized that spasticity-related involuntary activation would lead to force misperception in a contralateral force-matching task.
There are unique behaviours of interactions between the impaired and non-impaired limbs when both the impaired and non-impaired limbs act simultaneously in bilateral exertion. Studies suggest that forces of each limb are accurately perceived and modulated in a task-specific manner, despite weakness in the impaired limb and strength asymmetry in stroke survivors with hemiplegia (25–30). Instead of even force distribution between 2 limbs, as observed in healthy subjects, the impaired and non-impaired limbs share the total force with a unique sharing pattern in bilateral force-production tasks, i.e. each limb produces the same relative force with respect to its maximum strength over the tested range. In other words, each limb uses the same effort. This sharing pattern has been consistent over a wide range of force (5–65% of maximal strength) with and without specific instructions to produce the same magnitude of force regardless of visual feedback (25–27). This sharing pattern is maintained even when visual feedback of force from one limb is distorted (28). However, the sharing pattern changes when 2 limbs produce force together at high intensities or at the maximal level. The impaired side contributes disproportionally more to the total force (25, 29, 30). Both impaired and non-impaired limbs are able accurately to distinguish different levels of effort (31). Furthermore, it is known that there is bilateral cortical activation during unilateral exertion (32). However, greater cortical areas are activated during simultaneously bilateral exertion (33). These studies suggest that bilateral exertion tasks are not simply a superposition of the activity of 2 unilateral tasks. In this study, 2 force-matching paradigms (unilateral vs bilateral exertion) were used to examine the possible effect of bilateral exertion on force perception.
METHODS
Subjects
Eleven individuals (7 females and 4 males, mean age 60.1 years (standard deviation (SD) 12.7 years)) with post-stroke spastic hemiparesis volunteered to participate in this study (see Table I for details). Inclusion criteria were: (i) upper extremity paresis resulting from a single ischaemic or haemorrhagic stroke at least one year earlier; (ii) residual volitional elbow flexion torque on the impaired side; (iii) mild to moderate spasticity in elbow flexors, ranging from 1 (mild) to 3 (severe) on the modified Ashworth scale (MAS); (iv) full passive range of motion in the impaired shoulder and elbow joints; (v) sufficient cognitive ability to give informed consent and understand instructions related to the experiment; and (vi) able to see the visual targets. Exclusion criteria were: (i) a history of multiple strokes or bilateral involvement; (ii) presence of contracture that would limit full elbow range of motion on the impaired side; (iii) visuospatial neglect and impairment; (iv) elbow flexor spasticity rated at MAS 4 that limits passive range of motion and positioning or no spasticity (flaccid or fully recovered); (v) elbow flexor and extensor co-contraction; (vi) cognitive deficits that patients could not follow instructions for the experiments and/or give informed consent. Subjects were asked not to change the total daily dose of any anti-spasticity medications for at least 2 weeks before participation. Referring physicians or therapists conducted the MAS test. They were blind to the primary experiment. All subjects gave informed consent prior to participation.
|
Table I. Characteristics of stroke survivors |
||||||||
|
Subject number |
Age (years) /Sex |
Premorbid handedness |
Hemiplegia |
Stroke (months) |
Haemorrhagic (H) or ischaemic (I) |
Elbow flexor MAS |
Strength – impaired side (n) |
Strength – non-impaired side (n) |
|
1 |
65/F |
R |
L |
59 |
H |
1 |
12.9 |
59.1 |
|
2 |
55/F |
R |
L |
55 |
H |
1+ |
63.2 |
96.8 |
|
3 |
59/F |
R |
L |
143 |
I |
1 |
47.4 |
144 |
|
4 |
75/M |
R |
R |
121 |
I |
1+ |
41.5 |
71.2 |
|
5 |
49/M |
R |
L |
16 |
I |
1 |
83.9 |
253.1 |
|
6 |
57/F |
R |
R |
50 |
H |
1 |
49.3 |
142 |
|
7 |
76/M |
R |
R |
70 |
H |
1 |
91 |
109.5 |
|
8 |
79/F |
R |
L |
51 |
H |
1 |
33.9 |
98.5 |
|
9 |
79/M |
R |
L |
24 |
H |
1 |
108 |
119.2 |
|
10 |
50/M |
R |
R |
27 |
H |
2 |
54.8 |
171 |
|
11 |
50/F |
R |
L |
120 |
H |
1 |
67.7 |
86.8 |
|
Mean (SD) |
63.1 (12.2) |
66.9 (42.8) |
59.4 (27.3) |
122.8 (54.4) |
||||
|
SD: standard deviation; R: right; L: left; MAS: Modified Ashworth Scale; N: Newton. |
||||||||
The study was approved by the Committee for the Protection of Human Subjects at the University of Texas Health Science Center at Houston and TIRR Memorial Hermann Hospital.
Procedure
Similar to previous experimental settings (28, 34), subjects sat on a height-adjustable chair and rested their upper limbs on mechanically grounded supports. Limbs were symmetrically positioned with the shoulder slightly flexed and abducted to approximately 45°, the elbow flexed to approximately 90°, and the wrist in a neutral position. As the elbow of the impaired arm flexed, the exact amount of wrist pronation varied between subjects. Each individual’s pronation tendency was used for the initial posture and for all trials. The wrist of the non-impaired arm matched that of the impaired arm.
Stabilization straps secured both upper limbs to the supports at both proximal and distal forearm. Load cells (208C02; PCB Piezotronics, Depew, NY, USA) at the distal end of each forearm measured elbow flexion forces. Sensor distances from the elbows were symmetrical. Force signals were amplified (PCB, Depew Piezotronics, NY, USA) and digitized at 1,000 Hz (PCI-6229, National Instruments, Austin, TX, USA) and recorded with LabVIEW (National Instruments, Austin, TX, USA).
Maximum voluntary contractions. Subjects first performed isometric maximum voluntary contractions (MVCs) of elbow flexors. Subjects were asked to refrain from moving their trunk, shoulders or wrists. Each side performed 3 MVC trials of 5-s duration per trial in a randomized order. The highest force value was used as the MVC force for the respective arm. Visual targets of submaximal forces (10%, 20%, and 30% of this MVC force) were subsequently created for the matching tasks described below.
Matching tasks. The main experimental tasks were submaximal isometric elbow flexion matching tasks. One arm generated a reference force and the other arm generated a matching force. Subjects only received real-time visual feedback of the reference force and a submaximal target. Instructions were to generate and maintain a reference force following the force target with the reference arm, and to match the absolute magnitude of force, not the effort, with the other arm, i.e. subjects were explicitly instructed not match how hard they pushed (Fig. 1). To encourage subject motivation, feedback in the form of a numerical score was given after each trial. The numerical score constituted the percentage error of matching the target over last 3 s. Accepted trials were those in which subjects indicated that they thought the forces matched just after the trial.
There were 2 matching conditions: unilateral exertion and bilateral exertion (Fig. 1). In unilateral exertion-matching tasks, subjects were instructed to generate a reference force with one arm by following the visual target, and then generate a matching force with the other arm, i.e. 1 at a time. Unilateral exertion trials were 20 s in duration. Starting at the 4-s mark, subjects generated the reference force for 6 s. The start and end were signalled by visual and auditory cues. Then, after a 4-s rest, the subject flexed the elbow of the other arm to produce the matching force. In the bilateral exertion-matching tasks, subjects generated and maintained the reference force with 1 arm while generating a matching force simultaneously later in the trial. Bilateral exertion trials were 16 s in duration. Subjects were cued to start generating the matching force at the 8-s mark while maintaining the reference force. Subjects received no visual feedback of the force of the matching arm, but were provided with visual feedback of the reference arm for the reference force period.
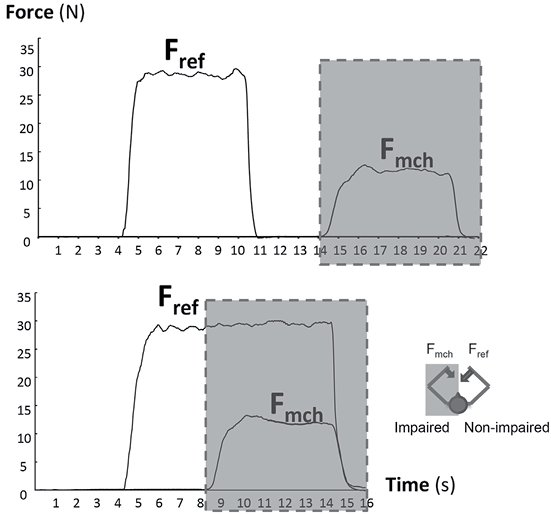
Fig. 1. Representative trials of unilateral (upper) and bilateral (lower) exertion force matching tasks. Shaded areas indicate no visual feedback available to subjects. Fref: reference force; Fmch: matching force.
In total, 4 blocks of matching tasks were performed. The order of the blocks was randomized, and the order of the trials within each block was randomized. Each block consisted of 3 trials at 3 target force levels (10%, 20%, and 30% of the MVC of the reference arm). Each level had 2 repetitions. The averaged forces were used for further analysis. For all subjects, the MVC of the impaired arm was greater than 30% MVC of the non-impaired arm. Thus subjects were capable of matching force at all target levels between arms. The entire experiment, including the MVC trials and matching trials, took approximately 2 h. Sufficient rest time was allowed between trials in order to minimize possible fatigue effect.
Data analysis
For the matching trials, the mean force in a 1.5-s window of each contraction for reference and matching forces was calculated. The end of this window was 0.5 s before the end of the contraction. Fmch was the mean matching force, and Fref was the mean reference force. The force mismatch error was quantified by the log of the force ratio (FR).
The ratio was used to average across different target force levels. The ratio itself does not allow for comparison between underestimation (bounded between 0 and 1) and overestimation (bounded between 1 and infinity). Therefore, we used the natural log of the ratio so that underestimation value (negative values) mirrors overestimation (positive values).
FR = log(Fmch/Fref) (Eq. 1)
To test whether the relative strength of the arms accounts for mismatch errors, we normalized the force ratio by the arms’ MVC ratio to yield an effort ratio (ER). ER served as an estimate of effort mismatching.
ER = log((Fmch/Fref)/ (MVCmch/MVCref)) = log(Emch/Eref) (Eq. 2)
where MVCmch is the MVC force of the matching arm, MVCref is the MVC force of the reference arm, Emch is Fmch/MVCmch, and Eref is Fmch/MVCmch.
Statistical analysis
Two-way repeated measures analysis of variance (ANOVAs) with factors of ARM (matching arm, × 2, impaired vs non-impaired) and TYPE (exertion type, × 2, unilateral vs bilateral) were used to assess whether mismatch errors (FR and ER) depended on factors matching arm and exertion type by collapsing data across all force levels. One-way ANOVA tests were then performed to determine force target level effects (LEVEL, × 3) separately for each ARM and TYPE. Paired 1-way t-tests were used to assess whether ER was less than FR (i.e. whether effort mismatches were less than force mismatches). All tests used a Bonferroni-adjusted alpha level of 0.025 per test (0.05/2).
RESULTS
Overall, there were mismatches in both force and effort in the majority of conditions. Mismatches, as quantified by log ratios of the matching arm to reference arm, or FR = log(Fmch/Fref) and ER = log(Emch/Eref), depended on which arm was the matching arm (non-impaired vs impaired), but did not depend on the exertion type (unilateral vs bilateral) (Fig. 2). Two-way repeated measures ANOVA on FR revealed a significant effect of ARM (F = 330.73, p < 0.001), a non-significant effect of TYPE (F = 1.53, p = 0.217), and a non-significant interaction effect between ARM and TYPE (F = 0.11, p = 0.745). Analysis of MVC-normalized force ratios, ER, showed the same pattern. Two-way repeated measures ANOVA on ER revealed a significant effect of ARM (F = 18.37, p < 0.001), a non-significant effect of TYPE (F = 1.84, p = 0.176), and a non-significant interaction effect between ARM and TYPE (F = 0.14, p = 0.707).
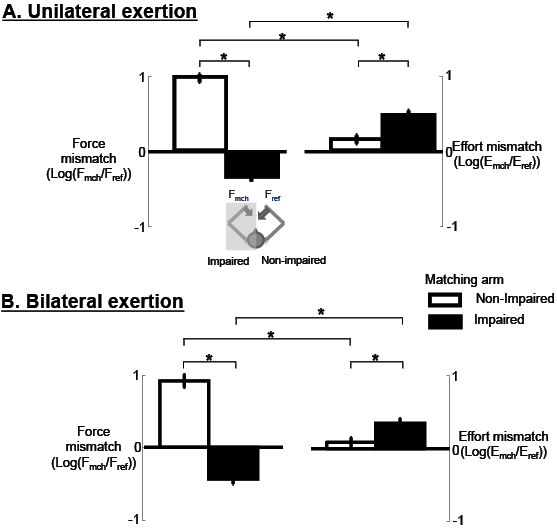
Fig. 2. Mismatches in force and effort for contralateral matching tasks. (A) Unilateral contractions and (B) bilateral exertions. Fref: reference force; Fmch: matching force. Data are means (± 1 standard error (SE)) across subjects, force target levels, and trials of each contralateral matching task. *Statistical significance (p < 0.025).
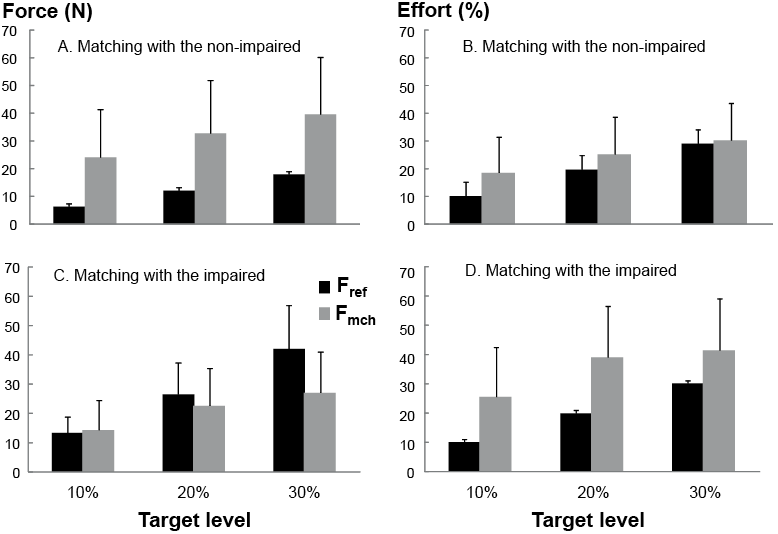
Table II summarizes force and effort for each exertion condition, matching arm and target force level. Log ratios (both FR and ER), indicating mismatches, were significantly different from zero (i.e. mismatches were significant) for most conditions (p < 0.025, Table II). Exceptions included the 10% MVC target level when matching with the impaired arm, in which mean FR was not significantly different from zero (unilateral: –0.2 (SD 1.0), p = 0.271; bilateral: –0.2 (SD 0.6), p = 0.027). The other exceptions were for ER at the 20% and 30% MVC target levels when matching with the non-impaired arm (unilateral 20%: 0.1 (SD 0.5), p = 0.247; unilateral 30%: –0.1 (SD 0.5), p = 0.457; bilateral 20%: 0.0 (SD 0.7), p = 0.987; bilateral 30%: –0.1 (SD 0.5, p = 0.325). In other words, stroke subjects were able to match the magnitude of the reference force magnitude at low levels (10% MVC) with the impaired arm, while they matched the effort of the reference force at moderate levels (20, 30% MVC) with the non-impaired arm.
The following sections provide more detailed breakdowns of the comparison between matching arms and the comparison between exertion types according to post-hoc analyses.
Matching with the non-impaired arm vs matching with the impaired arm
For both unilateral and bilateral exertions, force mismatches were different when the non-impaired arm was the matching arm from when the impaired arm was the matching arm (Fig. 2). Mean FR with the non-impaired arm matching (1.0 (SD 0.6)) was significantly different from FR with the impaired arm matching (–0.3 (SD 0.8), p < 0.001) during unilateral exertions. During bilateral exertions, mean FR with the non-impaired arm matching (0.9 (SD 0.8)) was also significantly different from FR with the impaired arm matching (–0.4 (SD 0.6), p < 0.001). Consideration of only the magnitude of FR also revealed significant differences (p < 0.001) for both unilateral and bilateral exertions. Collectively, these results indicated that there was overestimation (positive FR) when matching with the non-impaired arm, and underestimation (negative FR) when matching with the impaired arm. Both unilateral and bilateral exertion showed a similar pattern of overestimation and underestimation.
With respect to ER, similarly, there was a significant difference between matching arm conditions for both unilateral and bilateral exertions (Fig. 2B). Mean ER with the non-impaired arm matching (0.1 (SD 0.6)) was significantly smaller than ER with the impaired arm matching (0.5 (SD 0.8), p < 0.001) during unilateral exertions. During bilateral exertions, mean ER with the non-impaired arm matching (0.1 (SD 0.6)) was also significantly smaller than ER with the impaired arm matching (0.3 (SD 0.6), p = 0.009). These results showed that there was overestimation of effort when matching with the impaired arm and the non-impaired arm in both matching conditions, but overestimation was significantly greater in unilateral exertion than in the bilateral exertion-matching condition (p < 0.025).
Matching performance during unilateral vs bilateral exertions
Unilateral and bilateral exertions showed similar patterns of effort and force mismatches. Force mismatches were not significantly different between unilateral and bilateral exertions, for both when the non-impaired arm was the matching arm (p = 0.336) and when the impaired arm was the matching arm (p = 0.050, Fig 2A). Effort mismatches were also not significantly different between exertion types (matching with the non-impaired arm: p = 0.336; matching with the impaired arm: p = 0.050; Fig. 2B).
Matching force or effort
Both effort and force were mismatched in most conditions, as shown above. The log ratios (FR and ER) allowed further comparisons between force mismatches (FR) and effort mismatches (ER). ER was significantly less than FR when the non-impaired arm was the matching arm (unilateral exertion: p = 0.002; bilateral exertion: p = 0.005). In contrast, when the impaired arm was the matching arm, the ER was not significantly less than FR (unilateral exertion: p = 0.694; bilateral exertion:p = 0.439).
Effects of target force levels on matching
For unilateral exertions, when subjects matched with the non-impaired arm, there was a significant target force level effect on the log ratios (FR: p = 0.003; ER: p = 0.007). There was greater overestimation in the force magnitude at lower levels while the effort was matched at high levels (20, 30% MVC). This effect was not significant when matching with the impaired arm (FR: p = 0.263; ER: p = 0.115). For bilateral exertions, there was a significant target force level effect when matching with the non-impaired arm on ER (p = 0.019), but not on FR (p = 0.101). There was also no significant effect when matching with the impaired arm (FR: p = 0.067; ER: p = 0.035) (Table II, Fig. 3).
|
Table II. Force and effort for each exertion condition, matching arm, and force target level. Values are across-subject means and standard deviation (SD). Target levels are percent of reference arm maximum voluntary contractions (MVC) |
||||||||
|
Exertion |
Matching arm |
Target level (%) |
Fmch |
Fref |
Log (Fmch/Fref) |
Emch |
Eref |
Log (Emch/Eref) |
|
Unilateral |
Non-impaired |
10 |
24.0 (17.3) |
6.3 (2.8) |
1.2 (0.6) |
18.5 (12.8) |
10.1 (1.0) |
0.4 (0.7) |
|
20 |
32.7 (19.1) |
12.1 (5.2) |
0.9 (0.5) |
25.2 (13.3) |
19.7 (0.8) |
0.1 (0.5) |
||
|
30 |
39.6 (20.5) |
17.9 (7.6) |
0.7 (0.5) |
30.2 (13.3) |
29.0 (1.0) |
–0.1 (0.5) |
||
|
Impaired |
10 |
14.3 (10.1) |
13.3 (5.4) |
–0.2 (1.0) |
25.5 (16.9) |
10.1 (0.9) |
0.6 (1.0) |
|
|
20 |
22.6 (12.7) |
26.5 (10.7) |
–0.3 (0.7) |
39.0 (17.4) |
19.9 (1.0) |
0.5 (0.7) |
||
|
30 |
27.0 (13.9) |
42.0 (14.8) |
–0.5 (0.5) |
41.4 (17.6) |
30.1 (0.9) |
0.2 (0.4) |
||
|
Bilateral |
Non-impaired |
10 |
23.9 (16.4) |
6.8 (3.3) |
1.1 (0.9) |
17.9 (10.4) |
10.9 (2.5) |
0.3 (0.7) |
|
20 |
32.7 (22.6) |
12.5 (5.5) |
0.8 (0.9) |
23.7 (12.7) |
20.1 (2.1) |
–0.0 (0.7) |
||
|
30 |
41.0 (26.9) |
18.2 (8.0) |
0.7 (0.8) |
29.7 (13.1) |
29.4 (2.9) |
–0.1 (0.5) |
||
|
Impaired |
10 |
12.8 (7.4) |
14.4 (5.3) |
–0.2 (0.6) |
21.8 (9.3) |
11.0 (0.7) |
0.6 (0.6) |
|
|
20 |
17.4 (9.1) |
27.8 (11.5) |
–0.6 (0.5) |
30.1 (12.8) |
21.0 (1.9) |
0.2 (0.6) |
||
|
30 |
28.6 (15.2) |
42.4 (15.3) |
–0.5 (0.6) |
42.7 (16.0) |
30.7 (1.2) |
0.2 (0.6) |
||
|
Fmch: force of the matching arm; Fref: force of the reference arm; Emch: effort of the matching arm; Eref: effort of the reference arm. Bold log ratios are not significantly different from zero (p > 0.025). |
||||||||
DISCUSSION
The main findings of the present study include: (i) there are both force and effort mismatches in most conditions; (ii) there are asymmetries in force perception. When the non-impaired arm is the matching arm, effort and force overestimation occur, but effort is matched better than force. When the impaired arm is the matching arm, force underestimation and effort overestimation occur, but force is matched better than effort; (iii) there is no difference in matching performance between unilateral and bilateral exertion-matching tasks. Our findings of mismatches of both force and effort using a standard force-matching paradigm are clearly different from the results of previous studies in which both healthy subjects and stroke survivors without spasticity were tested. In these studies, a reference force is reproduced by a contralateral homologous muscle based on the “sense of effort”, regardless of the cause of strength asymmetry, such as fatigue (11, 23), partial curarization (13), changes in muscle length (10) or stroke (14, 24). One major difference between the current study and a recent study with stroke subjects is different inclusion/exclusion criteria. Only stroke survivors with spastic hemiparesis were recruited in this study, while stroke survivors with spastic hypertonia were excluded in previous studies of stroke subjects (14, 24). The strikingly contrasting results between this and the previous stroke studies suggest that spasticity may be the primary factor that force and effort misperception occurs during both reference and matching force production. Misperception of force and effort is possibly related to involuntary activation of synergistic muscles mediated by diffuse bulbospinal projections (4–6), as discussed in the Introduction.
When a reference force is produced by a spastic muscle, both intended voluntary force by the spastic muscle and involuntary activation of spontaneous motor units and other synergistic spastic muscles are registered in the central nervous system (CNS). In attempt to match the intended voluntary force of the spastic muscle based on the “sense of effort”, a higher magnitude of matching force is produced on the non-impaired limb. That explains why effort was better matched, particularly at moderate levels (20, 30% MVC) than force when the non-impaired limb was the matching arm in this study. On the other hand, when the impaired side is the matching arm, we observed a higher normalized matching force, i.e. “effort”. It is likely that the CNS has to increase “effort” to accommodate related spastic synergistic activation on the impaired limb when activating the spastic elbow flexors in producing the matching force. Force from involuntary synergistic activation of forearm muscles may also contribute to measured elbow flexion force. As a result, the absolute magnitude of matching force was greater based on the “sense of effort”. Therefore, force was better matched than effort when the impaired arm was the matching arm at higher levels (20, 30% MVC targets).
Our main findings suggest that there is spasticity-related involuntary activation during intended activation of a spastic muscle, and that this type of synergistic activation is not perceived individually, but as a whole. Understanding the role of spasticity in involuntary activation and force perception could help clinicians better understand a number of clinical phenomena. For example, stroke survivors cannot accurately perceive the amount of force produced by part of a synergistic movement. This may be presented as an abnormally high level of grip force for a fragile object. The elbow joint may rest at an abnormal flexed position secondary to spontaneous activation of motor units of spastic flexors that help generate anti-gravity force to maintain such a position (2, 7, 35). This altered resting joint position may assist a stroke survivor in carrying an object for a longer period of time. As such, spastic hypertonia of elbow flexors may, at times, be misperceived as strength by stroke survivors.
In conclusion, both force and effort misperceptions occur in most conditions in both unilateral and bilateral exertions in stroke survivors with spasticity. In contrast to the findings of effort matching in stroke survivors without spasticity, current findings strongly suggest that spasticity is likely to contribute to force and effort misperception during voluntary activation in chronic stroke. A limitation of the study is that there is no electromyographic demonstration of synergistic activation of spastic muscles, although this has been reported previously (4). A further limitation is the lack of exploration of individual data. Though well-documented, results from stroke survivors without spasticity will better support the argument in this paper.
Acknowledgement
This study was supported in part by an National Institutes of Health (NIH) grant R01NS060774.
REFERENCES
