Marina Demetrios, MBBS, FAFRM1, Caroline Brand, MBBS, MPH, RACP2,3, Julie Louie, MPhys4 and Fary Khan, MBBS, FAFRM, MD1,5
From the 1Department of Rehabilitation Medicine, Royal Melbourne Hospital, 2Melbourne EpiCentre,
The University of Melbourne, 3Centre for Research Excellence in Patient Safety (CREPS), Monash University,
4Department of Physiotherapy, Royal Melbourne Hospital and 5Department of Medicine, Dentistry and
Health Sciences, The University of Melbourne, Melbourne, Australia
OBJECTIVES: To describe ambulatory rehabilitation programmes (physical and occupational therapy activities and interventions) following botulinum toxin injections for post-stroke spasticity using a stroke rehabilitation taxonomy. To explore the relationship between therapy provided and injected limb/s and treatment goals.
DESIGN: Prospective, observational cohort study.
PARTICIPANTS: Stroke survivors (n = 47) participating in ambulatory rehabilitation programmes following botulinum toxin injections for upper limb, lower limb or upper and lower limb spasticity.
METHODS: Standardized therapy documentation forms were completed prospectively for each occupational and physical therapy session. Main outcomes were the proportion of: total therapy time spent in various therapeutic activities; total sessions during which each intervention was used to facilitate the activities most time was spent in; and goals related to each activity category. Sub-analysis was carried out for participants, based on limb/s injected.
RESULTS: Most time was spent in “upper extremity control” activities as the upper limb was more often injected. A large proportion of therapy time was spent in activities remediating “performance skills or body structure and function impairments”. In the upper and lower limb, and upper limb groups 38.7% and 46.2% of goals, respectively, related to this activity category, but less than 10% in the lower limb group. Little time was spent in community participation and leisure activities, whilst over one-third of lower limb group goals related to this category.
CONCLUSION: Ambulatory rehabilitation programmes following botulinum toxin injections for post-stroke spasticity varied depending on limb/s injected and reflected treatment goals to some extent.
Key words: spasticity; stroke; rehabilitation; occupational therapy; physiotherapy.
J Rehabil Med 2016; 48: 426–434
Correspondence address: Marina Demetrios, Rehabilitation Medicine Physician, Royal Melbourne Hospital, Royal Park Campus, 34–54 Poplar Road, Parkville 3052, VIC, Australia. E-mail: Marina.Demetrios@mh.org.au
Accepted Feb 18, 2016; Epub ahead of print Apr 13, 2016
Introduction
Stroke-related disability is globally increasing with the ageing population (1). Spasticity is a chronic impairment contributing to this and requiring long-term management. Spasticity has been reported in over 40% of stroke survivors (2), with disabling or severe spasticity affecting 4% (3) to 20% (4). Prevalence rates are even higher in those attending outpatient rehabilitation facilities, with severe or symptomatic spasticity in 30–36% (5). It can interfere with activity or provision of care, or cause pain or secondary complications (6, 7).
Botulinum toxin type A (BoNT-A) is widely used for focal muscle overactivity following stroke to manage these limitations. This is usually part of an integrated multidisciplinary rehabilitation programme (8, 9) targeting individualized patient and caregiver goals (10). However, rehabilitation in this setting remains a “black box”. Whilst guidelines recommend multidisciplinary management following BoNT-A (6, 11), the details of therapy content, and optimal therapy types (activities, interventions, therapy approaches), settings and intensities are unclear and highly variable (12). Studies rarely describe details of rehabilitation programmes beyond duration, frequency and generic broad therapy terms (13–15), making replication in a clinical setting difficult. In addition, therapy interventions are often investigated in isolation rather than in the milieu of the complex array of rehabilitation interventions provided in everyday clinical practice during rehabilitation programmes (12).
Extensive work has been done in opening and examining the “black box” of stroke rehabilitation programmes (16) during occupational therapy (OT) (17–19) and physiotherapy (PT) (19, 20). However, no such studies have investigated the therapy components provided following BoNT-A for post-stroke spasticity, although rehabilitation is often routinely provided. These complex rehabilitation interventions are difficult to standardize and define, and measuring what actually works in rehabilitation is a challenge. Standardized approaches to documenting therapy interventions are a step towards bridging this gap. A general model for describing critical attributes of disease management programmes for chronic conditions has been developed, allowing for comparisons across interventions (21). At a more detailed level, use of a taxonomy to characterize the complex array of therapy activities and interventions systematically provides a means of capturing what actually happens in stroke rehabilitation programmes (16, 22) and determining how therapy prescription relates to patients’ goals. Patient factors and specific therapy activities associated with better outcomes can then be identified (22), improving patient selection, service delivery and effectiveness. Stroke rehabilitation in inpatient (17, 18, 20, 23) and day hospital settings (19) has been defined in such a way. This is yet to be described specifically for multidisciplinary rehabilitation programmes following BoNT-A for post-stroke spasticity.
Using a standardized taxonomy (16, 22, 23), this study describes the therapeutic activities and interventions utilized within multi-disciplinary ambulatory rehabilitation programmes for stroke survivors receiving BoNT-A for upper and/or lower limb spasticity. Furthermore, how rehabilitation content differs, as categorized by the limb/s injected or the goals selected, is investigated; thus, exploring the “black box” of rehabilitation.
METHODS
Study design and participants
Adult stroke survivors (stroke >3 months previously) treated with BoNT-A for problematic upper and/or lower limb spasticity were recruited from a multidisciplinary, tertiary referral, spasticity management service in Victoria, Australia. Ethics approval was obtained from the relevant ethics committee. This study was part of a larger trial, comparing high intensity and lower intensity (usual care) ambulatory rehabilitation programmes following BoNT-A, as described elsewhere (24). All participants received individualized BoNT-A injections in the affected limb/s, as determined by clinical factors, spasticity patterns and treatment goals (6, 11). Up to 3 individualized, SMART (specific, measurable, achievable, realistic and timed) (25) goals for each treated limb (maximum 6 goals if both limbs were treated) were negotiated between participants, caregivers and therapist (JL) using the goal attainment scaling (GAS) process (26). Baseline data included demographic and clinical characteristics, such as stroke aetiology and localization.
Following BoNT-A injections participants were referred to ambulatory rehabilitation services determined by geographical catchment areas (24). Details of treatment goals were provided to treating therapists. Therapy programmes were individualized and goal-directed following treating team assessments, where interventions and therapy approaches were determined.
Standardized therapy documentation forms. Treating therapists used a stroke rehabilitation intervention classification system (comprising 2 forms; physical and occupational therapy) (17, 20, 22, 23) to document rehabilitation activities and interventions used by therapists during each PT and OT session for the programme duration. Sixteen occupational and 11 physical therapy activities of variable complexity comprise the key structure of the classification system, in addition to 31 interventions classified by targeted body systems (Table I). To complete the documentation grid, therapists recorded the duration of each therapeutic activity in 5-min intervals and codes for the interventions (maximum of 5) used to facilitate performance of these activities. A category for “other” interventions was utilized if needed. Additional information recorded included: the therapist’s discipline and level of experience, i.e. therapist, assistant or student; session duration; and time spent in formal assessment, co-treatment with other disciplines or in a group. Therapists were provided with written and verbal instructions in completing the forms, the relevant reference (22) and definitions of terms obtained from De Jong et al. (22). Therapists could use either form depending on relevance.
To categorize rehabilitation activities and goals the classification system used in studies describing OT stroke rehabilitation (17, 23) was modified for the purposes of this study. The 4 activity categories used in this study were: (i) performance skill or body structure or function impairments (17, 23) (e.g. pre-functional activity, transfers, upper extremity control, wheelchair, pre-gait, sitting balance or trunk control); (ii) gait; (iii) personal care tasks or home management; and (iv) community participation (community mobility, community integration) or leisure (Table I).
|
Table I. Rehabilitation activities and interventions comprising physical and occupational therapy standardized documentation forms |
||
|
PhysT activities |
Pre-functional activitya Bed mobilitya Sittinga Transfersa Sit-to-standa Wheelchair mobilitya |
Pre-gaita Gaitb Advanced gaitb Community mobilityd Intervention not related to functional activity |
|
Categories and examples of PhysT interventions |
Neuromuscular: balance training, postural awareness, motor learning, PNF, NDT, involved upper extremity addressed, CIMT Musculoskeletal: strengthening, mobilization, PROM/stretching, manual therapy, motor control Cardiopulmonary: breathing, aerobic/conditioning exercises Cognitive/perceptual/sensory: cognitive, perceptual, visual and sensory training |
Education: patient, family/caregiver, staff Equipment: prescription/selection, application, fabrication, ordering Modality: electrical stimulation, biofeedback, ultrasound Assistive device: ankle dorsi flex assist, cane, crutches, parallel bars, Swiss ball, walker, wheelchair Area involved: upper extremity, lower extremity, trunk, head and neck |
|
OT activities |
Pre-functional activitya Dressingc Bathingc Groomingc Toiletingc Feeding/Eatingc Transfersa Bed mobilitya Functional mobilityb |
Home managementc Community integrationd Leisure performanced Upper extremity controla Wheelchair managementa Sitting balance/trunk controla Intervention not related to functional activity |
|
Categories and examples of OT interventions |
Neuromuscular: Balance training, postural awareness, motor learning, PNF, NDT, CIMT Adaptive/compensatory: one handed skills, energy conservation, environmental adaption, adaptive equipment Musculoskeletal: strengthening, mobilization/manual therapy, PROM/stretching, oedema control Cardiopulmonary: breathing, aerobic/conditioning exercises Cognitive/perceptual/sensory: cognitive, perceptual, visual and sensory training |
Equipment: prescription/selection, application, fabrication (inc serial casts, splints), ordering Modality: electrical stimulation, biofeedback, hot/cold therapy Education: patient, family/caregiver, staff Assistive device: cane, crutches, parallel bars, Swiss ball, tray table, walker, wheelchair Area involved: upper extremity, trunk |
|
Adapted from De Jong et al., 2004 (22). Activities relating to: aperformance skill or body structure and function; bgait; cpersonal care tasks and home management; and dcommunity participation/leisure. Definitions (22, 23). Pre-functional activities: activities related to or provided in preparation for functional activities; upper extremity control: training/facilitation of normal movement, strength, range of motion, and alignment in the upper extremity. Initiating and completing care of the upper extremity for functional activities; intervention(s) not related to functional activity: interventions/time spent on patient’s behalf, but not in direct contact with patient, e.g. time spent selecting and ordering a splint. OT: occupational therapy; PhysT: physical therapy; PNF: proprioceptive neuromuscular facilitation; NDT: neurodevelopmental therapy; CIMT: constraint induced movement therapy; PROM: passive range of movement. |
||
Concordance between the total number of sessions for which therapy documentation forms were completed and the total number of OT and PT sessions attended according to the hospital computerized health management system was determined.
Participants’ experiences with the rehabilitation programme were assessed using questionnaires. Participants were asked to rate the degree to which the programme addressed and contributed to goal achievement, translation of skills learnt in everyday life and overall satisfaction.
Outcome measurement
Information provided in the therapy documentation forms was used to determine the following:
• Programme and session data: median programme duration (weeks); number and intensity of therapy sessions in total and per discipline (OT and PT); therapy time (h) in total and per discipline, mean session duration (min), and percentage of co-treatment and group sessions.
• Intervention content: percentage of total therapy time spent in each occupational and physical therapy rehabilitation activity and activity category, percentage of participants spending time in each activity and percentage of total sessions during which interventions were used to facilitate the 3 occupational and physical therapy activities that most time was spent in.
• Therapists: discipline, level of expertise.
Participant goals were categorized as described above and the percentage of total goals relating to each category, and upper or lower limb, was determined.
Data analysis
Data was entered into Microsoft Excel database and exported into Stata12 (StataCorp, TX, USA) for analysis. Descriptive statistics were presented as mean and standard deviation (SD) for continuous normally distributed data, median and interquartile range (IQR) for skewed or ordinal data and n (%) for categorical data. p-values < 0.05 indicated statistical significance. Sub-analysis was carried out for groups based on limb/s injected, i.e. upper and lower limb (ULLL), upper limb (UL) and lower limb (LL).
Programme duration (weeks) was calculated by dividing the number of days from first and last session, inclusive, by 7. Therapy intensity was defined as the number of sessions divided by programme duration (sessions per week). The total amount of therapy time for each activity (addition of time spent in the activity by each participant) and activity category was converted to a percentage of total therapy time across all participants and sub-groups.
Clinical trial registration number: Melbourne Health HREC 2010.165.
RESULTS
Of the 59 participants recruited to the larger study (24), between January 2011 to June 2012, therapy documentation forms were completed for 47 participants (median age 60.7 years and 2.0 years post-stroke) (Table II), documenting 285 OT sessions and 640 PT sessions (total 925 sessions). The only significant baseline difference was a shorter median time since stroke (1 (0.5–1.7) year) in the LL group compared with other groups (p = 0.004) (Table II). Whilst this group received more PT sessions in total (p = 0.019), there was no significant difference in programme duration and total number of therapy sessions (Table III).
|
Table II. Baseline characteristics of all participants and sub-groups based on limb/s injected with botulinum toxin type A |
||||
|
Characteristics |
All (n = 47) |
UL (n = 19) |
LL (n = 13) |
ULLL (n = 15) |
|
Demographic factors |
|
|
|
|
|
Sex male, n (%) |
34 (72.3) |
13 (68.4) |
11 (78.6) |
10 (71.4) |
|
Age, years, median (IQR) |
60.7 (47.5–68.3) |
61.9 (45.9–68.3) |
59.9 (50.7–65.2) |
59.3 (47.5–69.5) |
|
Time since stroke, years, median (IQR) |
2.0 (1.1–4.0) |
2.5 (1.8–7.2) |
1.0 (0.5–1.7)* |
3.0 (1.4–5.4) |
|
Clinical characteristics |
|
|
|
|
|
Stroke aetiology, n (%) |
|
|
|
|
|
Infarct |
34 (73.9) |
10 (55.6) |
12 (85.7) |
12 (85.7) |
|
Haemorrhage/mixed |
12 (25.5) |
8 (44.4) |
2 (14.3) |
2 (14.3) |
|
Stroke localization, n (%) |
|
|
|
|
|
Cortical |
38 (80.9) |
14 (73.7) |
12 (85.7) |
12 (85.7) |
|
Subcortical |
12 (25.5) |
6 (31.6) |
2 (14.3) |
4 (28.6) |
|
Dominant side affected, n (%) |
29 (61.7) |
9 (47.4) |
10 (71.4) |
10 (71.4) |
|
Living arrangements, n (%) |
|
|
|
|
|
Friends/family |
41 (87.2) |
16 (84.2) |
13 (92.9) |
12 (85.7) |
|
Alone |
2 (4.3) |
1 (5.3) |
0 (0) |
1 (7.1) |
|
Other |
4 (8.5) |
2 (10.5) |
1 (7.1) |
1 (7.1) |
|
*p-value > 0.05. IQR: interquartile range. UL: upper limb; LL: lower limb; ULLL: upper and lower limb. |
||||
|
Table III. Programme and session characteristics per participant |
||||
|
|
All (n = 47) |
ULLL (n = 15) |
UL (n = 19) |
LL (n = 13) |
|
Programme duration, weeks, median (IQR) |
10.0 (8.0–12.7) |
10.0 (8.0–12.7) |
9.6 (7.9–10.9) |
12.7 (9.0–14.1) |
|
Total sessions, median (IQR) |
17.0 (10.0–26.0) |
17.0 (10.0–26.0) |
18.0 (9.0–30.0) |
17.0 (13.0–26.0) |
|
Total number of sessions by discipline, median (IQR) |
|
|
|
|
|
OT |
3.0 (0–10.0) |
4.0 (0–9.0) |
8.0 (3.0–18.0)* |
0 (0-0)a |
|
PT |
12 (7.0–18.0) |
12.0 (7.0–16.0) |
10.0 (6.0–17.0) |
17.0 (13.0–25.0) |
|
Sessions per week, median (IQR) |
|
|
|
|
|
All |
1.5 (1.0–2.2) |
1.4 (1.2–2.4) |
1.7 (1.0–2.0) |
1.5 (1.0–2.2) |
|
OT |
0.4 (0–1.3) |
0.3 (0–0.9) |
1.4 (1.0–1.7)* |
0 (0-0) |
|
PT |
1.2 (0.8–1.7) |
1.1 (0.7–1.7) |
1.0 (0.7–1.6) |
1.5 (1.0–2.1) |
|
Total therapy time, h, median (IQR) |
|
|
|
|
|
OT |
2.5 (0–9.0) |
2.5 (0–9.0) |
7.5 (3.0–17.7)* |
0 (0–0) |
|
PT |
11.8 (6.9–18.5) |
11.5 (6.3–15.7) |
9.5 (4.3–16.7) |
18.5 (13.3–24.6)* |
|
OT+PT |
16.3 (9.6–30.8) |
15.8 (9.6–25.7) |
16.3 (7.8–30.4) |
18.5 (13.3–24.8) |
|
Duration of sessions, min, mean (SD) |
|
|
|
|
|
OT |
50.9 (9.6) |
49.9 (9.8) |
53.0 (16.6) |
42.5 (10.6) |
|
PT |
56.8 (6.6) |
56.3 (8.4) |
54.9 (6.3) |
59.7 (2.8) |
|
*p < 0.05. aTwo participants who had lower limb BoNT-A injections had OT. OT: occupational therapy; PT: physiotherapy; IQR: interquartile range; UL: upper limb; LL: lower limb; ULLL: upper and lower limb; SD: standard deviation. |
||||
Concordance between the number of sessions for which therapists’ completed therapy documentation forms and those actually attended by a sample of 22 participants was 68.8%.
Programme and session characteristics
Table III shows programme and session characteristics using data from the therapy documentation forms. The UL group received significantly more OT and the LL group more PT per participant compared with other groups (Table III).
Intervention content
Fig. 1 shows the proportion of total therapy time all participants and sub-groups spent in the various physical and occupational therapy activities. For all participants most time was spent in “upper extremity control”, followed by physical therapy activities including “interventions not related to functional activity”, “gait” and “pre-functional activity” (Fig. 1). Following “upper extremity control”, the next most common OT activities were “intervention not related to functional activity” and “pre-functional activity”. In the UL and ULLL groups most time was spent in “upper extremity control” activities (Fig. 1), whilst 88.5% and 43.5% of goals related to the UL respectively, and the remainder to gait.
Table IV shows the percentage of total therapy time and goals related to the activity categories for all participants and sub-groups. Most time was spent in performance skill or body structure and function activities, with more than one-third of goals for all groups except LL relating to this activity category. There was a higher percentage of total therapy time and goals related to gait activities in those who had the LL injected. Minimal time was spent in community participation and leisure activities (Fig. 1 and Table IV). A small proportion of goals, except in the LL group (mostly related to community mobility), related to this activity category. Whilst little time was spent in personal care activities and home management, a large proportion of goals in all but the LL group were associated with these tasks (Table IV).
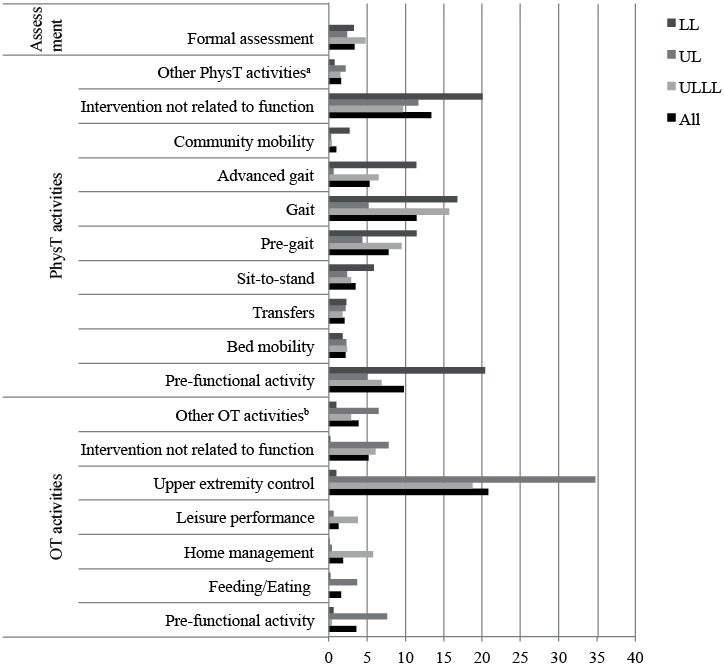
Fig. 1. Percentage of total therapy timec spent in various physical and occupational therapy rehabilitation activities for all participants and sub-groups. Activities performed for ≤ 2% of total therapy time in all groups classified as: aOther PhysT activities: sitting and wheelchair mobility. bOther OT activities: bathing, dressing, grooming, toileting, transfers, functional mobility, bed mobility, community integration, wheelchair management and sitting balance/trunk control. cTotal therapy time defined as total time spent in physiotherapy and OT across each group. PhysT: physical therapy; OT: occupational therapy; LL: lower limb; UL: upper limb; ULLL: upper and lower limb.
|
Table IV. Percentage of total therapy time and goals related to each of the activity categories for all participants and sub-groups |
|||||||||||
|
Activity and goal categories |
All n = 47 |
|
ULLL n = 15 |
|
UL n = 19 |
|
LL n = 13 |
||||
|
% time |
% goals |
|
% time |
% goals |
|
% time |
% goals |
|
% time |
% goals |
|
|
Performance skill/body structure & function |
53.6 |
34.5 |
|
45.9 |
38.7 |
|
64.4 |
46.2 |
|
45.2 |
8.8 |
|
Gait |
16.8 |
33.8 |
|
22.2 |
40.3 |
|
5.8 |
11.5 |
|
28.2 |
55.9 |
|
Personal care tasks and home management |
5.1 |
19.6 |
|
6.8 |
19.4 |
|
7.0 |
32.7 |
|
0.4 |
0.0 |
|
Community participation/leisure |
2.4 |
12.2 |
|
4.4 |
1.6 |
|
0.9 |
9.6 |
|
2.7 |
35.5 |
|
Percentage of total therapy time spent in “intervention not related to functional activity” and formal assessment not included. UL: upper limb; LL: lower limb; ULLL: upper and lower limb. |
|||||||||||
Table V shows the percentage of participants participating in occupational and physical therapy activities for the group and sub-groups. More than 50% of all participants participated in “upper extremity control” activities, as the UL was injected more often.
|
Table V. Proportion of participants participating in occupational and physical therapy activities |
||||
|
|
All n = 47 |
ULLL n = 15 |
UL n = 19 |
LL n = 13 |
|
OT activities |
|
|
|
|
|
Pre-functional activity |
15 (31.9) |
2 (13.3) |
11 (57.9)* |
2 (15.4) |
|
Bathing |
1 (2.1) |
0 (0.0) |
1 (5.3) |
0 (0.0) |
|
Dressing |
5 (10.6) |
1 (6.7) |
3 (15.8) |
1 (7.7) |
|
Grooming |
2 (4.3) |
0 (0.0) |
2 (10.5) |
0 (0.0) |
|
Toileting |
3 (6.4) |
2 (13.3) |
1 (5.3) |
0 (0.0) |
|
Feeding/eating |
7 (14.9) |
0 (0.0) |
6 (31.6)* |
1 (7.7) |
|
Transfers |
7 (14.9) |
1 (6.7) |
4 (21.1) |
2 (15.4) |
|
Bed mobility |
4 (8.5) |
0 (0.0) |
3 (15.8) |
1 (7.7) |
|
Functional mobility |
11 (23.4) |
1 (6.7) |
7 (36.8) |
3 (23.1) |
|
Home management |
6 (12.8) |
2 (13.3) |
3 (15.8) |
1 (7.7) |
|
Community integration |
1 (2.1) |
1 (6.7) |
0 (0.0) |
0 (0.0) |
|
Leisure performance |
1 (2.1) |
1 (6.7) |
0 (0.0) |
0 (0.0) |
|
Upper extremity control |
25 (53.2) |
8 (53.3) |
16 (84.2)* |
1 (7.7) |
|
Wheelchair management |
3 (6.4) |
2 (13.3) |
1 (5.3) |
0 (0.0) |
|
Sitting balance/trunk control |
13 (27.7) |
4 (26.7) |
7 (36.8) |
2 (15.4) |
|
Intervention not related to functional activity |
17 (36.2) |
7 (46.7) |
9 (47.4) |
1 (7.7)* |
|
PhysT activities |
|
|
|
|
|
Pre-functional activity |
34 (72.3) |
10 (66.7) |
13 (68.4) |
11 (84.6) |
|
Bed mobility |
22 (46.8) |
6 (40.0) |
8 (42.1) |
8 (61.5) |
|
Sitting |
22 (46.8) |
5 (33.3) |
9 (47.4) |
7 (53.8) |
|
Transfers |
23 (48.9) |
8 (53.3) |
9 (47.4) |
5 (38.5) |
|
Sit-to-stand |
32 (68.1) |
12 (80.0) |
10 (52.6) |
10 (76.9) |
|
Wheelchair mobility |
4 (8.5) |
1 (6.7) |
1 (5.3) |
0 (0.0) |
|
Pre-gait |
37 (78.7) |
13 (86.7) |
11 (57.9) |
12 (92.3) |
|
Gait |
39 (83.0) |
14 (93.3) |
12 (63.2)* |
13 (100) |
|
Advanced gait |
20 (42.6) |
6 (40.0) |
4 (21.1) |
10 (76.9)* |
|
Community mobility |
11 (23.4) |
3 (20.0) |
1 (5.3) |
7 (53.8)* |
|
Intervention not related to functional activity |
33 (70.2) |
12 (80.0) |
10 (52.6) |
11 (84.6) |
|
Formal assessment |
25 (53.2) |
9 (60.0) |
10 (52.6) |
7 (53.8) |
|
*p < 0.05. OT: occupational therapy; PhysT: physical therapy; UL: upper limb; LL: lower limb; ULLL: upper and lower limb. |
||||
Tables VI and VII show the percentage of total sessions during which interventions were used to facilitate the 3 occupational and physical therapy activities, respectively, in which most time was spent.
|
Table VI. Occupational therapy interventions used to facilitate the most common activities: percentage of total sessionsa |
|||
|
OT interventions |
OT activities |
||
|
Pre-functional activity n% |
Upper extremity control n% |
Intervention not related to functional activity n% |
|
|
Neuromuscular |
|
|
|
|
Balance training |
0.1 |
0.0 |
1.0 |
|
Postural awareness |
0.8 |
1.2 |
1.0 |
|
Motor learning |
1.5 |
2.8 |
0.0 |
|
NDT/Bobath |
1.7 |
4.2 |
0.0 |
|
Adaptive/compensatory |
|
|
|
|
One-handed skills |
1.0 |
0.5 |
0.0 |
|
Musculoskeletal |
|
|
|
|
Strengthening |
2.2 |
5.1 |
1.4 |
|
Mobilization, manual therapy |
2.1 |
3.6 |
1.1 |
|
PROM/stretching |
4.9 |
8.8 |
0.9 |
|
Educational |
|
|
|
|
Patient |
1.2 |
3.5 |
1.8 |
|
Other |
|
|
|
|
Exercise prescription |
2.8 |
5.8 |
0.6 |
|
Data are n% of total therapy sessions. Only interventions for which n ≥ 1.0% for at least one activity are included. aTotal sessions: OT and PT sessions. OT: occupational therapy; PT: physiotherapy; NDT: neurodevelopmental therapy; PROM: passive range of movement. |
|||
|
Table VII. Physical therapy interventions used to facilitate the most common activities: percentage of total sessionsa |
|||
|
PhysT interventions |
PhysT activities |
||
|
Pre-functional activity n% |
Gait n% |
Intervention not related to functional activity n% |
|
|
Neuromuscular |
|
|
|
|
Balance training |
3.6 |
8.1 |
3.1 |
|
Postural awareness |
4.1 |
6.3 |
1.6 |
|
Motor learning |
3.9 |
13.3 |
2.2 |
|
PNF |
1.3 |
0.1 |
0.0 |
|
NDT |
9.5 |
8.6 |
1.5 |
|
Involved upper extremity addressed |
7.0 |
1.3 |
4.1 |
|
Musculoskeletal |
|
|
|
|
Strengthening |
13.4 |
5.8 |
9.4 |
|
Mobilization |
2.9 |
0.3 |
4.4 |
|
PROM/stretching |
8.0 |
1.1 |
4.9 |
|
Manual therapy |
1.8 |
0.1 |
0.3 |
|
Motor control |
8.3 |
5.2 |
2.2 |
|
Cardiopulmonary |
|
|
|
|
Aerobic exercise |
0.2 |
4.4 |
8.3 |
|
Educational |
|
|
|
|
Patient |
0.6 |
2.5 |
5.0 |
|
Family/caregiver |
0.6 |
0.6 |
3.4 |
|
Modality interventions |
|
|
|
|
Electrical stimulation |
1.6 |
0.0 |
0.9 |
|
Assistive devices |
|
|
|
|
Hemi-rail |
0.0 |
1.0 |
0.0 |
|
Walker 4-wheel |
0.0 |
1.6 |
0.0 |
|
Other |
|
|
|
|
Other interventionsb |
0.4 |
1.5 |
9.4 |
|
Data are n% of total therapy sessions. Only interventions for which n ≥ 1.0% for at least one activity are included. aTotal sessions: OT and PT sessions. bOther interventions: exercise prescription, hydrotherapy, exercise bike, wobble board, pilates. OT: occupational therapy; PT: physical therapy; PhysT: physical therapy; PNF: proprioceptive neuromuscular facilitation; NDT: neurodevelopmental therapy; PROM: passive range of movement. |
|||
Overall, 87.3% of participants and/or their caregiver/s received some educational intervention during at least 1 session (range 1–21) relating to any of the rehabilitation activities.
Delivery personnel
Of all participants 93.6% received PT and 61.7% received OT. The majority of participants in the ULLL and UL group received OT (n = 11/15 (73.3%) and n = 16/19 (84.2%), respectively) compared with the LL group (n = 3/13 (23.1%), p = 0.002). All participants who had LL BoNT-A injections had PT. Over 25% of all participants were also seen by other disciplines including orthotists, podiatrists, exercise physiologists, social workers and dieticians.
Qualified occupational therapists and physiotherapists delivered the majority of rehabilitation sessions. Allied health assistants provided a small number of sessions (ranging from 1 to 12 sessions) in OT (n = 2/47 participants) and PT (n = 11/47 participants). Group sessions accounted for just over 20% (n = 186/925) of all sessions, involving 17 participants. Co-treatment with 2 disciplines (OT and PT) occurred in almost 9% (n = 82/925) of sessions, all of which were group sessions, and involved over 20% (n = 10/47) of participants.
Environment/therapy settings
The majority of participants (n = 46/47) attended government-funded, community-based rehabilitation services, whilst 1 saw a private therapist. Rehabilitation programmes were centre-based, except for 2 participants who received home-based therapy. All sessions were face-to-face contact.
Participant experience
Of the participants who completed questionnaires (n = 35/47), most reported that the programme addressed their goals (71.4%) and helped to contribute to their goals being achieved (74.3%) to a great deal or extreme amount. Over 70% reported being able to use skills learnt during the programme in everyday life to an extreme amount or great deal, up to 6 months but only 20% did so beyond this time frame. Overall, 91.4% reported being very satisfied or satisfied with the rehabilitation programme.
DISCUSSION
Therapeutic activities and interventions (physical and occupational therapy) comprising individualized ambulatory rehabilitation programmes following BoNT-A for spasticity in stroke survivors were explored using a standardized taxonomy (22). As far as we are aware this is the first study to explore current rehabilitation practices for post-stroke spasticity in such detail.
The 47 chronic (> 1 year) stroke survivors had a similar median age to those in other studies (18, 23). Whilst the LL group had a significantly shorter median time since stroke at baseline, they did not receive more therapy compared with other groups. Long-term motor improvement has been shown in those with chronic stroke following rehabilitation (27). In order to determine effectiveness, the therapeutic components of rehabilitation programmes need to be identified.
Programme characteristics varied depending on the limb injected. The UL group received significantly more OT and the LL group more PT. These results may reflect the tendency in Australia for occupational therapists to treat the UL and physiotherapists to treat mainly the LL; however, there is crossover. Therapy allocation may also be influenced by availability of services and resource allocation.
The rehabilitation activities that most therapy time was spent in reflected the limb/s injected, and goals to some extent; particularly in the UL group in relation to upper extremity control and those who had the LL injected where therapy and goals focused on gait activities. However, minimal therapy time was spent in personal care tasks, despite a moderate proportion of goals being related to such tasks in those receiving UL injections. Perhaps this is because these skills are often addressed in inpatient rehabilitation programmes (17, 23). In addition, little therapy time was spent addressing community participation and leisure activities, contrary to what may be expected for ambulatory rehabilitation programmes. Focal spasticity may not have resulted in significant limitation in community participation in the study cohort, except in the LL sub-group where over one-third of goals related to community mobility. As the majority of sessions were centre- rather than home-based, this factor in conjunction with resource limitations may have restricted the therapists’ focus on community activities. Nonetheless, translation of skills learnt in rehabilitation to the home and community environment is important in improving activity level and participation. Further studies are needed to explore how rehabilitation activities relate to goal categories (10) in this group, as diverting resources to relevant therapeutic activities may improve outcomes such as goal achievement.
A large proportion of therapy time in all groups was spent in remediating performance skills deficits or body structure and function impairments, particularly upper extremity control, with “passive range of motion” being the intervention most frequently used to facilitate such activities, similar to other studies of OT in stroke rehabilitation (18, 23). More than 1/3 of goals in all but the LL group related to performance skills or body structure and function impairmens. The importance of addressing performance skill deficits and motor preparation may be due to the window of opportunity after BoNT-A to normalize motor patterns, which have previously been inhibited by spasticity, before translation into functional activities, particularly as optimal reduction in spasticity occurs before the maximum change in function (28). Research into how therapeutic activities vary over the course of rehabilitation programmes following BoNT-A and their relationship to long-term functional outcomes is warranted.
The therapy documentation forms used in this study (16, 22, 23) combine documentation of therapeutic activities and corresponding interventions allowing the multidimensional nature and complexity of therapy to be described, whereas other tools record activities only (19). Further studies to assess the relevance of the current taxonomy to describing similar rehabilitation programmes would be beneficial. The forms, however, have inherent limitations. Firstly, they do not allow for recording of rest or inactive time, so do not reflect how intensely participants participated in therapy. This data is important in determining therapy effectiveness, as greater intensity of therapy has been suggested to result in increased recovery of motor function to a varying degree (29–31) and greater UL goal achievement following BoNT-A (24) after stroke. Stroke inpatients have been found to spend approximately one-third of therapy time in rest or inactivity (32), so it is also important to capture this time in ambulatory settings. In addition, the forms rely on therapists’ estimating activity time rather than an objective measure of accuracy. Therapists have been found to be inaccurate in their estimations of the time patients spend engaged in active task practice during therapy sessions (33, 34), overestimating active time by 28% and underestimating rest time by 36% (33). Objective measures for recording therapy activity time include simple counting of repetitions of tasks or exercises (35) or using activity monitors, such as accelerometers (36, 37), which also capture activity level out of therapy time in community-dwelling patients. This would be useful in determining the contribution of formal therapy time vs home-based activity to patient outcomes.
Although the therapists who completed the therapy documentation forms received instructions and written definitions of terms, reporting reliability was not assessed due to resource limitations. Thus, interventions and activities may have been incorrectly classified. “Intervention not related to functional activity” was defined as “activities not in direct contact with participants”; however, therapists recorded interventions that involved contact with participants in this section, but were not related to any of the functional activities, e.g. strengthening, aerobic/conditioning exercises. Video-recording of sessions and completion of forms by trained external therapists or an observer therapist (23, 38) would have enabled comparisons of accuracy of documentation. Information obtained from these methods may also have assisted in determining whether the forms capture details and multidimensionality of the rehabilitation programmes and applicability to this outpatient group. This would assist in determining whether adequate information is obtained to enable effectiveness research.
Lastly, particular rehabilitation activities may have related to more than 1 activity, e.g. “upper extremity control” may have been addressed during dressing or grooming tasks. However, it is not possible to categorize more than 1 activity per 5-min period (17). Hence, time spent in particular activities may have been underestimated.
The small number of rehabilitation centres and sample size limit the generalizability of the study findings. Due to small numbers, time spent in rehabilitation activities was examined as a percentage of the total therapy time added up for all participants and each sub-group rather than per participant, and sub-group analysis was not included for the rehabilitation interventions used. A much larger sample size would allow for analysis of all rehabilitation activities and interventions used in order to determine patterns of therapy approaches. As this study was conducted in the “real-life” clinical setting with limited funding, factors that influence therapy delivery, such as organizational and cultural differences between the centres, and availability of resources, were not addressed.
This study demonstrates that a stroke rehabilitation taxonomy (22) assists in describing therapeutic interventions and activities comprising ambulatory rehabilitation programmes following BoNT-A for spasticity in stroke survivors. Future, larger, multi-centred studies may use this approach to determine the relationship between activities and interventions and treatment goals, to identify which facets of therapy improve outcomes. Evidence-based spasticity management guidelines could then include detailed guidance on effective rehabilitation interventions after BoNT-A treatment to improve clinical practice and service delivery. Thus, healthcare providers, and other funding bodies, would be better informed about the benefits of costly rehabilitation programmes in promoting greater community independence (16).
ACKNOWLEDGEMENTS
We are grateful to all participants and therapists who completed the therapy documentation forms. We thank Louisa Ng for input into study design, Alex Gorelik for assistance with statistics and Fig. 1, Ian Baguley for valuable advice and Mary Galea for comments during the editing phase. This study was partially funded by the Australasian Faculty of Rehabilitation Medicine, Ipsen Open Research Fellowship. The funder has had no influence on the interpretation of data and the final conclusions drawn.
Conflicts of interest. Marina Demetrios has been on the Advisory Board for Ipsen and received sponsorship to attend meetings from makers of BoNT-A (Allergan and Ipsen). Julie Louie has received honoraria from Ipsen. No author has a personal financial interest in BoNT-A or in any of the methods used in this research.
REFERENCES
